Professional Documents
Culture Documents
Scheme Quiz Klon Set 2
Uploaded by
Supia NazmaOriginal Description:
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Scheme Quiz Klon Set 2
Uploaded by
Supia NazmaCopyright:
Available Formats
KOLEJ MATRIKULASI SELANGOR
SESSION 2015/2016 SEMESTER 1
SK016 CHEMISTRY
QUIZ 1.0 : MATTER (SCHEME SET 2)
NAME :
PRACTICUM :
DATE :
MARKS :
Objective Questions:
/ 15 =
[5 marks]
1. Which two of the ions below have the same number of electrons?(ans:A)
,
a.
and
b.
and
c.
and
d.
and
e.
and
2. Two isotopes of a given element will have the same number of ____, but a different number of ____ in
their nucleus.
protons, electrons c. protons, neutrons
electrons, protons d. neutrons, protons
3. Silver has two stable isotopes with masses of 106.90509 amu and 108.9047 amu. The average molar mass
of silver is 107.868 amu. What is the percent abundance of each isotope?
50.0% Ag-107 and 50.0% Ag-109
c.
55.4% Ag-107 and 44.6% Ag-109
b. 51.8% Ag-107 and 48.2% Ag-109
d.
48.2% Ag-107 and 51.8% Ag-109
4. What is the definition of molality?
a.
mass of solute per liter of solvent
b.
mass of solute per kg of solvent
c.
moles of solute per kg of solvent
d.
moles of solute in one liter of solvent
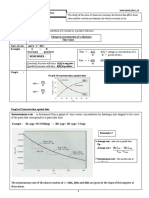
5. Concentrated sodium hydroxide is 19.4 M and has a density of 1.54 g/mL. What is the molality of
concentrated NaOH?
a.
m is no
c. secret
25.4 mto success.It is the result of preparation,hard work,learning
12.6
There
b.
19.8 m
d. 29.9 m
from failure.
Colin Powell
PART
ANSWER SCHEME
Reduction: Cr2O72- + 14H+ + 6e -
1(a)
2Cr3+ + 7H2O
MARKS
1
Oxidation: 6Fe2+ 6Fe3+ + 6e -
Overall: Cr2O72- + 14H+
2Cr3+ + 6Fe3+ + 7H2O
(b)(i)
nAl =
= 1.259 mol Al (available)
nCl2 =
= 0.549 mol Cl2 (available)
2 mol Al
3 mol Cl2
1.259 mol Al 1.259 x 3/2 @
= 1.889 mol Cl2 needed
Cl2 is the limiting reactant
(b)(ii)
3 mol Cl2
1
1
2 mol AlCl3
0.549 mol Cl2 0.549 x 2/3
= 0.366 mol AlCl3
Mass of AlCl3 = 0.366 x 133.5
1
1
= 48.86 g
There is no secret to success.It is the result of preparation,hard work,learning
from failure.
Colin Powell
10
There is no secret to success.It is the result of preparation,hard work,learning
from failure.
Colin Powell
You might also like
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryFrom EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryRating: 3.5 out of 5 stars3.5/5 (231)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)From EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Rating: 4.5 out of 5 stars4.5/5 (120)
- Grit: The Power of Passion and PerseveranceFrom EverandGrit: The Power of Passion and PerseveranceRating: 4 out of 5 stars4/5 (588)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaFrom EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaRating: 4.5 out of 5 stars4.5/5 (266)
- The Little Book of Hygge: Danish Secrets to Happy LivingFrom EverandThe Little Book of Hygge: Danish Secrets to Happy LivingRating: 3.5 out of 5 stars3.5/5 (399)
- Never Split the Difference: Negotiating As If Your Life Depended On ItFrom EverandNever Split the Difference: Negotiating As If Your Life Depended On ItRating: 4.5 out of 5 stars4.5/5 (838)
- Shoe Dog: A Memoir by the Creator of NikeFrom EverandShoe Dog: A Memoir by the Creator of NikeRating: 4.5 out of 5 stars4.5/5 (537)
- The Emperor of All Maladies: A Biography of CancerFrom EverandThe Emperor of All Maladies: A Biography of CancerRating: 4.5 out of 5 stars4.5/5 (271)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeFrom EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeRating: 4 out of 5 stars4/5 (5794)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyFrom EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyRating: 3.5 out of 5 stars3.5/5 (2259)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersFrom EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersRating: 4.5 out of 5 stars4.5/5 (344)
- Team of Rivals: The Political Genius of Abraham LincolnFrom EverandTeam of Rivals: The Political Genius of Abraham LincolnRating: 4.5 out of 5 stars4.5/5 (234)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreFrom EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreRating: 4 out of 5 stars4/5 (1090)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceFrom EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceRating: 4 out of 5 stars4/5 (895)
- Her Body and Other Parties: StoriesFrom EverandHer Body and Other Parties: StoriesRating: 4 out of 5 stars4/5 (821)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureFrom EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureRating: 4.5 out of 5 stars4.5/5 (474)
- The Unwinding: An Inner History of the New AmericaFrom EverandThe Unwinding: An Inner History of the New AmericaRating: 4 out of 5 stars4/5 (45)
- The Yellow House: A Memoir (2019 National Book Award Winner)From EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Rating: 4 out of 5 stars4/5 (98)
- On Fire: The (Burning) Case for a Green New DealFrom EverandOn Fire: The (Burning) Case for a Green New DealRating: 4 out of 5 stars4/5 (73)
- Exercise Born HaberDocument17 pagesExercise Born HaberSupia NazmaNo ratings yet
- Quiz PHASE EQUILIBRIA (Set 2)Document4 pagesQuiz PHASE EQUILIBRIA (Set 2)Supia NazmaNo ratings yet
- 2 Metallic BondsDocument13 pages2 Metallic BondsSupia NazmaNo ratings yet
- Instantaneous Rate: Is Determined From A Graph of Time Versus Concentration by Drawing A Line Tangent To The CurveDocument13 pagesInstantaneous Rate: Is Determined From A Graph of Time Versus Concentration by Drawing A Line Tangent To The CurveSupia NazmaNo ratings yet
- Collision Theory States That For A Reaction To OccurDocument9 pagesCollision Theory States That For A Reaction To OccurSupia NazmaNo ratings yet
- Quiz PHASE EQUILIBRIA (Set 3)Document4 pagesQuiz PHASE EQUILIBRIA (Set 3)Supia NazmaNo ratings yet
- Chapter 1: Matter 1.1 Atoms and Molecules: Packed in A Small NucleusDocument35 pagesChapter 1: Matter 1.1 Atoms and Molecules: Packed in A Small NucleusSupia NazmaNo ratings yet
- The Density of An Aqueous Solution Containing 18Document1 pageThe Density of An Aqueous Solution Containing 18Supia NazmaNo ratings yet
- Quiz 5.1 Gas (Set 1)Document2 pagesQuiz 5.1 Gas (Set 1)Supia NazmaNo ratings yet
- Quiz C8 Set 4Document2 pagesQuiz C8 Set 4Supia NazmaNo ratings yet
- Thermochemistry: 1. Concept of EnthalphyDocument2 pagesThermochemistry: 1. Concept of EnthalphySupia NazmaNo ratings yet
- Quiz 1.1 2021 LectureDocument4 pagesQuiz 1.1 2021 LectureSupia NazmaNo ratings yet
- Quiz 5.3 Solid (Set 3)Document2 pagesQuiz 5.3 Solid (Set 3)Supia NazmaNo ratings yet
- Quiz States of Matter (Set 4)Document4 pagesQuiz States of Matter (Set 4)Supia NazmaNo ratings yet
- Exercise Kuliah TermoDocument9 pagesExercise Kuliah TermoSupia NazmaNo ratings yet
- Quiz C6 Set 2Document2 pagesQuiz C6 Set 2Supia NazmaNo ratings yet
- Exercise MassSpecDocument1 pageExercise MassSpecSupia NazmaNo ratings yet
- Extra Exercises - Measurement of ConcentrationDocument1 pageExtra Exercises - Measurement of ConcentrationSupia NazmaNo ratings yet
- Quiz C6 Set 4Document2 pagesQuiz C6 Set 4Supia NazmaNo ratings yet
- Exercise Born HaberDocument17 pagesExercise Born HaberSupia NazmaNo ratings yet
- Exercise 1: DH DH DH For The Following ReactionDocument4 pagesExercise 1: DH DH DH For The Following ReactionSupia NazmaNo ratings yet
- Quiz C6 Set 1Document2 pagesQuiz C6 Set 1Supia NazmaNo ratings yet
- Quiz C5 STATES OF MATTER (Set 5)Document2 pagesQuiz C5 STATES OF MATTER (Set 5)Supia NazmaNo ratings yet
- Quiz States of Matter (Set 4)Document4 pagesQuiz States of Matter (Set 4)Supia NazmaNo ratings yet
- Quiz C6 Set 3Document1 pageQuiz C6 Set 3Supia NazmaNo ratings yet
- Quiz 5.1 Gas (Set 1)Document2 pagesQuiz 5.1 Gas (Set 1)Supia NazmaNo ratings yet
- Quiz 5.2 Liquid (Set 2)Document2 pagesQuiz 5.2 Liquid (Set 2)Supia NazmaNo ratings yet
- Tutorial 1.1 (PG 1-2)Document3 pagesTutorial 1.1 (PG 1-2)Supia NazmaNo ratings yet
- Gases (B)Document115 pagesGases (B)Supia NazmaNo ratings yet
- Liquid StateDocument46 pagesLiquid StateSupia NazmaNo ratings yet