Professional Documents
Culture Documents
1 Course Ravan Deem Ter Notes
Uploaded by
Jonnathan Ariel CespedesOriginal Description:
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
1 Course Ravan Deem Ter Notes
Uploaded by
Jonnathan Ariel CespedesCopyright:
Available Formats
1
Broadening in chromatography (Notes adapted from Harris Quantitative Chemical Analysis)
V. L. Colvin, chemistry 330
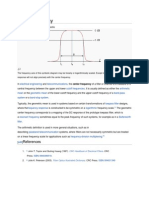
We use the term plate height or H as a catchall term for the width of a chromatographic
band. The smaller the plate height, the narrower the band. A very small plate height is therefore
a desirable feature of a chromatographic column. It relates to the theoretical number of plates,
N, in a column through:
H
L
N =
Where L is the total length of the column. For a fixed L, if we have extremely small plate
heights then N is large which signals a great, narrow chromatography band.
Conceptually the plate height, or H, is a measure of the band spread in the column. If
you have a very thin H, then the peak is not very spread out in space. A large H, the peak is
really spreading. I like to remember what H is by imagining chopping up the column to capture
a band in space. H is proportional to the thickness of the slice that would contain most of the
analyte actually 36% to be (nearly) exact. Eventually it runs through the column it reaches
the end and travels the distance L at its elution time t
r
. The plate height here is the theoretical
plate height in this case.
Lets think about H from a practical, experimental point of view. If you had a column,
a flow rate, a target analyte and you measured the chromatogram you could measure the peak
width which you captured by measuring the standard deviation, o, of the peak - and the elution
time t
r
. You would also know the column length. You can calculate H then:
L
H
2
o
=
Plate height uses the variance, o
2
, in this equation rather than the standard deviation, o,
because variances are additive. Thus we can describe variances from multiple sources, and as
will be done next plate heights from different sources. Typically in a gas chromatography
separation H values for good columns are about 100 to 1000 microns while for liquid
chromatography systems they are only 10 microns.
We can consider plate height also theoretically. While abstract, the insights this provides
helps us design better columns and improve the selection of columns and running conditions.
The Van Deemter equation rolls many band broadening sources into one compact form:
x
x
Cu
u
B
A H + + =
Where u
x
is the linear flow rate and A, B and C are constants. This equation tells us that there
are mechanisms of band broadening that depend linearly and inversely on flow rate, and some
that are independent. In the next section I give you some standard equations for the most critical
sources of broadening without full derivations so you can examine how choices you might
make in running a column will influence your plate height. Remember you almost always
want H to be as small as possible to make for an efficient separation with highly resolved peaks.
2
1. Sources that lead to broadening independent of flow rate (A constants)
(A) You never apply your sample in a step function. There is some width in time/volume to the
initial application of sample, which is characterized by a At for the injection. This is usually
referred to as a plate height contribution from injection or sometimes application:
units length
L
H
2
inj
inj
o
=
There is a similar issue that arises from the detection process. No detector samples instantly;
samples are usually collected over some time period At for the sampling. Sometimes these
broadening issues are given to you as a volume (e.g. you injected 300 microliters onto a column).
To convert to a spread in time you need the flow rate in terms of volume per minute. Simply
multiplying the sampling or injection volume by this flow rate gives you the spread in time.
(B) Another contribution here is the possibility of multiple flow paths. Important primarily in
liquid chromatography, this leads to an intrinsic spread in the data. It becomes less severe when
bead sizes are smaller.
2. Sources that lead to broadening that depend inversely on the velocity. Diffusion is a large
factor in column broadening. Molecules randomly diffuse in space due to the concentration
gradient they move into areas of lower concentration. How quickly they do this depends on the
molecules diffusion constant in the mobile phase (hence D
m
in the equation below). You can
look up diffusion constants for molecules; the units are usually like acceleration m
2
/sec.
x
m
x
m
m
dif
diffusion
u
D
L
u
L
D
L
t D
L
H
=
|
.
|
\
|
=
= =
2
2
2
2
o
Where velocity along the direction of elution (x) is u
x
. Note: we calculated the time, t, from the
velocity and the overall column length. This equation tells us that to have small plate height we
want a velocity like a firehose, not a trickle.
3. Sources that lead to broadening that are proportional to the velocity. Another source of
broadening is equilibration. If you dont let the solute/analyte sit long enough to partition
according to the equilibrium constant (e.g. partition coefficient) between the mobile and
stationary phase then you have a distribution of partition coefficients and hence, a broad
chromatogram. This term is sometimes called the mass transfer term:
x
s
trans
transfer
u
D k
d k
L
H
+
~ =
2 '
2 '
2
) 1 (
o
In this expression we use D
s
which is the diffusion constant in the stationary phase, k the
capacity factor of the analyte, and d is the thickness of the stationary phase.
On the course website you will find and excel sheet entitled VanDeemter.xls. In this excel sheet
you can run different conditions for the Van Deemter equation and compare how hard, or easy, it
may be to minimize the plate height for a given separation.
You might also like
- Fenton90b Nonlinear Wave TheoriesDocument18 pagesFenton90b Nonlinear Wave TheoriesMed Ali MaatougNo ratings yet
- Theories of ChromatographyDocument13 pagesTheories of ChromatographyZarish Iftikhar100% (1)
- Theory of HPLC Band BroadeningDocument15 pagesTheory of HPLC Band Broadeningpenelopaaa22No ratings yet
- Introduction To Chromatography TheoryDocument18 pagesIntroduction To Chromatography TheoryAkash Marathakam33% (3)
- 5.1.CHM 3401 - Chromatography 1Document45 pages5.1.CHM 3401 - Chromatography 1anisghani100% (1)
- Van Deemter EquationDocument19 pagesVan Deemter EquationMohammad Sabir HussainNo ratings yet
- The Van Deemter Equation by FahadDocument7 pagesThe Van Deemter Equation by FahadNauman RanaNo ratings yet
- Column Efficiency TestingDocument3 pagesColumn Efficiency TestingvamsiNo ratings yet
- Phase (Which May Be A Gas, A Liquid or A Supercritical Fluid) - The Mobile Phase IsDocument5 pagesPhase (Which May Be A Gas, A Liquid or A Supercritical Fluid) - The Mobile Phase IsasjawolverineNo ratings yet
- Jce-1983-393 - Modernization of The Van Deemter EquationDocument6 pagesJce-1983-393 - Modernization of The Van Deemter EquationSolomon EricksonNo ratings yet
- The Van Deemter EquationDocument15 pagesThe Van Deemter EquationLailan_Fauzana_2560No ratings yet
- Instrumental Analysis II Band Broadening and Column EfficiencyDocument23 pagesInstrumental Analysis II Band Broadening and Column EfficiencySalix MattNo ratings yet
- Theories of ChromatographyDocument21 pagesTheories of Chromatographysupport marenNo ratings yet
- The Chromatographic Process ExplainedDocument40 pagesThe Chromatographic Process ExplainedFarida NurNo ratings yet
- Theoretical Plate: Unit OperationsDocument17 pagesTheoretical Plate: Unit OperationsMehari AsratNo ratings yet
- 1 LectureDocument25 pages1 LectureChemistry DepartmentNo ratings yet
- Lecture 2, Fundamentals HPLCDocument36 pagesLecture 2, Fundamentals HPLCChevuru ManushaNo ratings yet
- Application of MachineDocument21 pagesApplication of MachineSengatan LebahNo ratings yet
- Introduction to Chromatographic SeparationsDocument45 pagesIntroduction to Chromatographic SeparationsIvy JoyceNo ratings yet
- VAN DEEMTER EQUATION OPTIMIZATIONDocument9 pagesVAN DEEMTER EQUATION OPTIMIZATIONMuhammad AhmedNo ratings yet
- Van Deemter EquationDocument12 pagesVan Deemter EquationSANJEET YADAVNo ratings yet
- Chromatographic Theory: Prof. Derick CarbooDocument45 pagesChromatographic Theory: Prof. Derick CarbooMaame Ama FrempongNo ratings yet
- CHromatography Chapter 26Document45 pagesCHromatography Chapter 26Steffi YapNo ratings yet
- Balance Equations and Conservation of MassDocument7 pagesBalance Equations and Conservation of MassTiago FerreiraNo ratings yet
- An Introduction To Chromatographic SeparationsDocument45 pagesAn Introduction To Chromatographic Separationsliska ramdanawatiNo ratings yet
- Column Efficiency Is Affected by TheDocument8 pagesColumn Efficiency Is Affected by TheSana QureshiNo ratings yet
- Van Deemter EquationDocument19 pagesVan Deemter EquationAsif AliNo ratings yet
- Column Efficiency and Van Deemter Equation 27 OctDocument19 pagesColumn Efficiency and Van Deemter Equation 27 OctAdvocate MataleNo ratings yet
- The Rate Theory of ChromatographyDocument2 pagesThe Rate Theory of ChromatographySana BatoolNo ratings yet
- Basic HPLC Theory FornstedtDocument24 pagesBasic HPLC Theory FornstedtFadhlanFachrozaHasibuanNo ratings yet
- Band Broadening Theory (Van Deemter Equation)Document5 pagesBand Broadening Theory (Van Deemter Equation)Ruiz ManuelNo ratings yet
- And In: Relative Contributions of Molecular Diffusion Anastomosis Gas Chromatographic ColumnsDocument6 pagesAnd In: Relative Contributions of Molecular Diffusion Anastomosis Gas Chromatographic Columnsnbson217No ratings yet
- Halász Equation - 01Document11 pagesHalász Equation - 01Je RivasNo ratings yet
- Basic Chromatography Notes 1Document27 pagesBasic Chromatography Notes 1Aufa InsyirahNo ratings yet
- Hurst ExponentDocument8 pagesHurst Exponentharrison9No ratings yet
- Gas Chromatography 1Document14 pagesGas Chromatography 1Arjun MaharajNo ratings yet
- HPLC Separation Efficiency and OptimizationDocument9 pagesHPLC Separation Efficiency and OptimizationSecret ManNo ratings yet
- Introduction To ChromatographyDocument24 pagesIntroduction To ChromatographyAnonymous dY7XNz100% (1)
- Rose - 2007 - An Introduction To Ultrasonic Guided WavesDocument18 pagesRose - 2007 - An Introduction To Ultrasonic Guided Wavespaivensolidsnake0% (1)
- Nche 211 Unit 6 2022Document97 pagesNche 211 Unit 6 2022Nicholus Opincai JercilioNo ratings yet
- Chapter 26 - Gas Chromatography1Document19 pagesChapter 26 - Gas Chromatography1PARIKNo ratings yet
- Statistical Wireless Channel ModelDocument5 pagesStatistical Wireless Channel Modelanc62No ratings yet
- The Lecture Contains: Correlation Functions Ideas About Eddy Size Intensity of Turbulence or Degree of TurbulenceDocument19 pagesThe Lecture Contains: Correlation Functions Ideas About Eddy Size Intensity of Turbulence or Degree of Turbulencekoushikthephoenix1No ratings yet
- ENSC 283 Introduction To Fluid Mechanics: Chapter 7 - Flow Past Immersed BodiesDocument16 pagesENSC 283 Introduction To Fluid Mechanics: Chapter 7 - Flow Past Immersed BodiesArjun CpNo ratings yet
- Separation Methods Based On Distributions in Discrete Stages (9/26/12)Document18 pagesSeparation Methods Based On Distributions in Discrete Stages (9/26/12)raghu_iictNo ratings yet
- A. Van Deemter Equation in Chromatography. SolutionDocument4 pagesA. Van Deemter Equation in Chromatography. SolutionSourav PandaNo ratings yet
- Application of DifferentiationDocument4 pagesApplication of DifferentiationnishagoyalNo ratings yet
- Chromatography 1Document63 pagesChromatography 1umarNo ratings yet
- Review of Fundamental Problems and Solutions in Fractal Surface GrowthDocument26 pagesReview of Fundamental Problems and Solutions in Fractal Surface GrowthChristian LauNo ratings yet
- Chromatographic MethodsDocument28 pagesChromatographic Methodsbnar jNo ratings yet
- Packed distillation columns optimizationDocument7 pagesPacked distillation columns optimizationramsrivatsan1992No ratings yet
- Halász Equation - 02Document10 pagesHalász Equation - 02Je RivasNo ratings yet
- Derivation of High-Order Advection - Diffusion Schemes: Pavel TkalichDocument16 pagesDerivation of High-Order Advection - Diffusion Schemes: Pavel TkalichIssa IssaNo ratings yet
- labirinthFloSci Bib28 06Document10 pageslabirinthFloSci Bib28 06amiboyzNo ratings yet
- Theoretical Plate - An Overview - ScienceDirect TopicsDocument19 pagesTheoretical Plate - An Overview - ScienceDirect TopicsChibuike CharlesNo ratings yet
- Center Frequency Filter GuideDocument3 pagesCenter Frequency Filter GuideNurul FakhriahNo ratings yet
- New Tube Count Approach Heat ExchangersDocument5 pagesNew Tube Count Approach Heat ExchangersSalim YilmazNo ratings yet