Professional Documents
Culture Documents
Benzene 2
Uploaded by
claimstudent3515Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Benzene 2
Uploaded by
claimstudent3515Copyright:
Available Formats
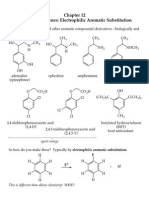
Aromatic compounds
GCE
BENZENE
Structure Primary analysis revealed benzene had an... empirical formula of CH and a molecular formula of C6H6 Draw out some suitable structures which fit the molecular formula C6H6
Q.1
Kekule
Kekul suggested a PLANAR, CYCLIC structure with ALTERNATING DOUBLE AND SINGLE BONDS
However it did not readily undergo electrophilic addition - no true C=C bond
X X X X
only one 1,2 disubstituted product existed i.e you didnt get two isomers like these..
all six CC bond lengths were similar. Double bonds are shorter than single ones
NOT
the ring was thermodynamically more stable than expected - see below
To explain the above, it was suggested that the structure oscillated between the two Kekul forms but was represented by neither of them - a resonance hybrid.
www.XtremePapers.net
KNOCKHARDY SCIENCE 2008
GCE
Aromatic compounds
Delocalised The theory suggested that instead of three localised (in one position) double system bonds, the six (pi) electrons making up those bonds were delocalised (not in
any one particular position) around the ring by overlapping the p orbitals. There would be no double bonds to be added to and all bond lengths would be equal. It also gave a planar structure.
6 single bonds one way to overlap adjacent p orbitals another possibility delocalised pi orbital system
C C C
C C C C C
C C C C C
C C C
This structure was particularly stable and resisted any attempt to break it down through normal electrophilic addition. However, overall substitution of any of the hydrogen atoms would not affect the delocalised system.
Thermodynamic evidence for stability When unsaturated hydrocarbons are reduced to the corresponding saturated compound, energy is released. If the experiment is carried out in a bomb calorimeter the amount of heat liberated per mole (enthalpy of hydrogenation) can be measured.
When cyclohexene (one C=C bond) is reduced to cyclohexane, 120kJ of energy is released per mole. C6H10(l) + H2(g) > C6H12(l)
theoretical H = - 360 kJ mol 1
152 kJ mol 1 more stable than expected
Theoretically, if benzene contained three separate C=C bonds it would release 360kJ per mole when reduced to cyclohexane C6H6(l) + 3H2(g) > C6H12(l)
H = - 120 kJ mol 1
experimental H = - 208 kJ mol 1
benzene releases 208kJ per mole when reduced putting it lower down the energy scale it is 152kJ per mole more stable than expected. this value is known as the resonance energy.
www.XtremePapers.net
KNOCKHARDY SCIENCE 2008
Aromatic compounds
GCE
REACTIONS OF THE BENZENE (AROMATIC) RING
Nitration Converts benzene into nitrobenzene, C6H5NO2 The nitration of benzene is the first step in an historically important chain of reactions. These lead to the formation of dyes, and explosives. reagents conditions equation mechanism + NO2 conc. nitric acid and conc. sulphuric acid (catalyst) reflux at 55C C6H6 + HNO3 > C6H5NO2 + H2O
NO2
NO2 + H+
an electron pair leaves the delocalised system to form a bond to the electrophile this disrupts the stable delocalised system and forms an unstable intermediate. to restore stability, the pair of electrons in the C-H bond moves back into the ring. overall there is substitution of hydrogen ... ELECTROPHILIC SUBSTITUTION
electrophile
NO2+ , nitronium ion or nitryl cation it is generated in an acid-base reaction as follows... 2H2SO4 + acid proton donor HNO3 base 2HSO4 + H3O+ + NO2+
proton acceptor
Importance
Nitrobenzene is the start of an important chain of reactions.
NO2 NH2
+ Cl N2
OH
nitrobenzene
phenylamine (aniline)
benzene diazonium chloride
phenol
www.XtremePapers.net
KNOCKHARDY SCIENCE 2008
4 Halogenation
GCE
Aromatic compounds
Converts benzene into chlorobenzene, C6H5Cl chlorine and a halogen carrier (the catalyst) reflux in the presence of a halogen carrier such as iron, iron(III) chloride, iron(III) bromide, aluminium chloride C6H6 + Cl2
+ Cl
reagents conditions
equation mechanism
>
C6H5Cl
HCl
Cl
Cl + H +
PROBLEM
Chlorine is non polar so is not a good electrophile. A catalyst (HALOGEN CARRIER) is required to polarise the halogen. anhydrous aluminium chloride can act as the catalyst the Al in AlCl3 has only 6 electrons in its outer shell; - LEWIS ACID it increases the polarisation of the Cl-Cl bond this makes the charge on C more positive and the following occurs
Cl2
AlCl3
Cl
AlCl4
Cl Cl Cl
Cl+
Cl
Cl
Al
Al
Cl
Cl+
Cl
Cl
The aluminium chloride acts as a LEWIS ACID as the aluminium atom has only 6 electrons in its outer shell. It accepts a pair of electrons from one of the atoms in the chlorine molecule.
All the atoms in the new species have complete outer shells
The new species has 6 electrons in its outer shell. It has one less electron than a chlorine atom so has a + charge.
the H+ ion removed from the benzene ring reacts with the AlCl4 ion to regenerate the AlCl3 H+ + AlCl4 > AlCl3 + HCl
Comparison with alkenes
The conditions are much tougher than with alkenes because the delocalised system makes benzene more stable and less reactive.
www.XtremePapers.net
KNOCKHARDY SCIENCE 2008
Aromatic compounds
GCE
Friedel Crafts Reactions General A catalyst is used to increase the positive nature of the electrophile and make it better at attacking benzene rings. AlCl3 acts as a Lewis Acid and helps break the CCl bond.
Alkylation
substitutes an alkyl (e.g. methyl, ethyl) group reagents conditions electrophile equation mechanism a haloalkane (RX) and anhydrous aluminium chloride AlCl3 room temperature; dry inert solvent (ether) a carbocation ion R+ (e.g. CH3+) C6H6 + C2H5Cl > C6H5C2H5 + HCl
CH 2 CH 3
CH 2 CH 3
CH 2 CH 3
catalyst
anhydrous aluminium chloride acts as the catalyst the Al in AlCl3 has only 6 electrons in its outer shell; - LEWIS ACID it increases the polarisation of the C-Cl bond in the haloalkane this makes the charge on C more positive and the following occurs RCl + AlCl3 AlCl4 + R+
Q.2
Which haloalkane would you use to make... propylbenzene C6H5CH3
www.XtremePapers.net
KNOCKHARDY SCIENCE 2008
6 Industrial
GCE
Aromatic compounds
Alkenes are used instead of haloalkanes but an acid must also be present Phenylethane, C6H5C2H5 is made by this method reagents electrophile equation ethene, anhydrous AlCl3 , conc. HCl C2H5+ (an ethyl carbocation) > C6H5C2H5 (ethyl benzene) C6H6 + C2H4
mechanism a) the HCl reacts with the alkene to generate a carbocation
H H C H C H H
+
Cl
H H
H C
H C H Cl
b) electrophilic substitution takes place as the C2H5+ attacks the ring
CH 2 CH 3
CH 2 CH 3
CH 2 CH 3
use
ethyl benzene is dehydrogenated to produce phenylethene (styrene); this is then used to make poly(phenylethene) - also known as polystyrene Why is ethene used industrially rather than chloroethane ?
Q.3
Acylation
substitutes an acyl (e.g. ethanoyl) group the aluminium chloride catalyst acts in the same was as with alkylation reagents conditions electrophile product equation mechanism
O C CH 3 O C CH 3 O C CH 3
an acyl chloride (RCOCl) and anhydrous AlCl3 reflux 50C; dry inert solvent (ether) RC+= O ( e.g. CH3C+O ) > C6H5COCH3 + HCl carbonyl compound (aldehyde or ketone) C6H6 + CH3COCl
www.XtremePapers.net
KNOCKHARDY SCIENCE 2008
Aromatic compounds
GCE
FURTHER SUBSTITUTION OF BENZENE
Theory It is possible to substitute more than one functional group. But, the functional group already on the ring affects... how easy it can be done where the next substituent goes
Z Z
Group
ELECTRON RELEASING OH, CH3 Increases Easier 2,4,and 6
ELECTRON WITHDRAWING NO2 Decreases Harder 3 and 5
Example(s) Electron density of ring Ease of substitution Position of substitution
Examples
Substitution of nitrobenzene is... more difficult than with benzene - NO2 is electron withdrawing produces a 1,3 disubstituted product - NO2 directs to the 3 (meta) position
NO2 NO 2
NO 2
Substitution of methylbenzene is... easier than with benzene produces a mixture of isomers
CH 3
- CH3 is electron releasing - CH3 directs to 2 (ortho) and 4 (para) positions
CH 3 NO2 CH 3
NO2
OH Br
OH Br
Some groups make substitution so much easier that multiple substitution takes place
www.XtremePapers.net
KNOCKHARDY SCIENCE 2008
Br
GCE
Aromatic compounds
Q.4
Draw structures for the monosubstituted organic product(s) of the following reactions.
c. HNO3 / c. H2SO4 >
C2H5Cl / AlCl3 >
NO2
C2H5Cl / AlCl3 >
CH3
CH3Cl / AlCl3 >
NO2
Br2 / FeBr3 >
OH
c. HNO3 / c. H2SO4 >
CH3
Cl2 / FeCl3 >
www.XtremePapers.net
KNOCKHARDY SCIENCE 2008
Aromatic compounds
GCE
9 CH3
METHYLBENZENE
Introduction methylbenzene used to be called toluene it has a methyl group attached directly to the benzene ring it has the formula C6H5CH3
Preparation formed from benzene using Friedel-Crafts alkylation reagents conditions electrophile equation chloromethane and anhydrous aluminium chloride AlCl3 room temperature; dry inert solvent (ether) CH3+ C6H6 + CH3Cl > C6H5CH3 + HCl
Reactions
there are two parts to the methylbenzene molecule
CH3
the alkyl side chain undergoes free radical substitution
the aromatic ring
undergoes electrophilic substitution
Q.5
For each of the reactions below... state the reagents and conditions required state the type of mechanism taking place write a balanced equation state any other possible organic products, giving brief reasons for their formation explain how the chlorinated product responds to treatment with NaOH(aq)
CH3 CH3 Cl
a)
CH 3
CH2 Cl
b)
www.XtremePapers.net
KNOCKHARDY SCIENCE 2008
You might also like
- Schaum's Easy Outline of Organic Chemistry, Second EditionFrom EverandSchaum's Easy Outline of Organic Chemistry, Second EditionRating: 3.5 out of 5 stars3.5/5 (2)
- Experiment 1 OrgChem Lab 2Document5 pagesExperiment 1 OrgChem Lab 2John Harvey Santos100% (2)
- Benzene: Draw Out Suitable Structures Which Fit The Molecular Formula C HDocument8 pagesBenzene: Draw Out Suitable Structures Which Fit The Molecular Formula C HmuhajireenNo ratings yet
- Romatic Ydrocarbons: CH CH CH C HDocument7 pagesRomatic Ydrocarbons: CH CH CH C HMukesh BishtNo ratings yet
- 3 ArenesDocument7 pages3 ArenesAnonymous 8VJhV1eI2yNo ratings yet
- 4.1.1 ArenesDocument8 pages4.1.1 ArenesFin BrickmanNo ratings yet
- Draw Out Some Suitable Structures Which Fit The Molecular Formula C HDocument4 pagesDraw Out Some Suitable Structures Which Fit The Molecular Formula C HSean s ChipangaNo ratings yet
- 16 Organ PDFDocument3 pages16 Organ PDFAya ZhNo ratings yet
- 13239639-đã chuyển đổiDocument9 pages13239639-đã chuyển đổiBùi Quang HuyNo ratings yet
- Carboxylic Acid and Their DerivativesDocument13 pagesCarboxylic Acid and Their DerivativesaqidahNo ratings yet
- CH 18 SummaryDocument5 pagesCH 18 Summaryalstjq1003No ratings yet
- Hsslive XI CH 9 Chemistry Notes by AkDocument18 pagesHsslive XI CH 9 Chemistry Notes by AkkundrapupNo ratings yet
- Hydrocarbon NotesDocument187 pagesHydrocarbon Notessamay gujratiNo ratings yet
- Halogenoalkanes and Haloarenes: Nucleophilic Substitution ReactionsDocument30 pagesHalogenoalkanes and Haloarenes: Nucleophilic Substitution ReactionsEarNo ratings yet
- Hydrocarbon Compounds: AromaticDocument49 pagesHydrocarbon Compounds: AromaticUMMU MARDHIAH ABDUL HALIMNo ratings yet
- Organic Chemistry - Reactions and MechanismsDocument120 pagesOrganic Chemistry - Reactions and MechanismsLoveena Steadman100% (8)
- Topic 2Document32 pagesTopic 2KAI YANG LIMNo ratings yet
- Organic Chemistry Review: Structural and Functional GroupsDocument4 pagesOrganic Chemistry Review: Structural and Functional GroupsRanie MagpocNo ratings yet
- Organic Chemistry PDFDocument13 pagesOrganic Chemistry PDFPrem KumarNo ratings yet
- Introduction To Orgnic ChemistryDocument27 pagesIntroduction To Orgnic ChemistryladybugNo ratings yet
- Introduction To FGs and AlkanesDocument28 pagesIntroduction To FGs and AlkanesAbdullah AmjadNo ratings yet
- Al KynesDocument19 pagesAl KynesAnkit JaipuriaNo ratings yet
- Aromatic Compounds: NamingDocument13 pagesAromatic Compounds: NamingTanzimNo ratings yet
- St. Andrew's Junior College H2 Chemistry 2013 Organic Chemistry Lecture Notes 5 ArenesDocument26 pagesSt. Andrew's Junior College H2 Chemistry 2013 Organic Chemistry Lecture Notes 5 ArenesArvin LiangdyNo ratings yet
- ArenesDocument2 pagesArenesSam SmithNo ratings yet
- Reaction KineticsDocument12 pagesReaction KineticsTheresaHangDuongNo ratings yet
- CH - CH - BR 1° Halide 2° Halide 3° Halide: H H C CH CL CHDocument8 pagesCH - CH - BR 1° Halide 2° Halide 3° Halide: H H C CH CL CHShahbaz NazirNo ratings yet
- Important Factors:: Lecture Notes Chem 51B S. KingDocument20 pagesImportant Factors:: Lecture Notes Chem 51B S. KingHuấnĐìnhNguyễnNo ratings yet
- chm207 chp5Document92 pageschm207 chp5Muhd Mirza HizamiNo ratings yet
- Reasoning Questions in Organic Chemistry Text ExerciseDocument14 pagesReasoning Questions in Organic Chemistry Text ExerciseEr Purushottam PalNo ratings yet
- Bailey Alkanes PowerpointDocument31 pagesBailey Alkanes PowerpointBritney PattersonNo ratings yet
- 32 Organ PDFDocument3 pages32 Organ PDFThuvarakaNo ratings yet
- Alkene Chemistry Properties and Formation-1Document40 pagesAlkene Chemistry Properties and Formation-1Alvis MwangiNo ratings yet
- Reactions of AlkanesDocument6 pagesReactions of AlkanesHamzaNo ratings yet
- Organic Reaction MechanismsDocument27 pagesOrganic Reaction MechanismsAhmed SideegNo ratings yet
- OrganicDocument93 pagesOrganicPatel MswaziNo ratings yet
- 3 Functional Groups Alcohols:: Lecture Notes Chem 51B S. KingDocument27 pages3 Functional Groups Alcohols:: Lecture Notes Chem 51B S. KingHuấnĐìnhNguyễnNo ratings yet
- Al KanesDocument9 pagesAl KanesJue MayaNo ratings yet
- Benzene StructureDocument30 pagesBenzene StructureAnuki PereraNo ratings yet
- Alkanes and CycloalkanesDocument11 pagesAlkanes and CycloalkanesDinesh BijalwanNo ratings yet
- Chapter 5 Alkyl HalidesDocument32 pagesChapter 5 Alkyl HalidesMohd HanafiahNo ratings yet
- CH22Document23 pagesCH22David LiNo ratings yet
- 5.4.1 Arenes635464Document6 pages5.4.1 Arenes635464ArchitNo ratings yet
- BIFUNCTIONAL CHEMISTRY REACTIONSDocument36 pagesBIFUNCTIONAL CHEMISTRY REACTIONSchidambaramrNo ratings yet
- Bailey Alkanes PowerpointDocument31 pagesBailey Alkanes PowerpointBritney PattersonNo ratings yet
- Functional Groups and Hydrocarbons: Lecture #2 Melchor Cerdania Dept. of Chemistry, Silliman UniversityDocument45 pagesFunctional Groups and Hydrocarbons: Lecture #2 Melchor Cerdania Dept. of Chemistry, Silliman UniversityGlayzell AltrinnaNo ratings yet
- Physical and Chemical Properties of AlkynesDocument83 pagesPhysical and Chemical Properties of AlkynesAira Villarin100% (2)
- 11 Cbse Chemistry Organic ChemistryDocument22 pages11 Cbse Chemistry Organic ChemistryKrish KakkarNo ratings yet
- Hydrocarbons PDFDocument12 pagesHydrocarbons PDFMayank ShahabadeeNo ratings yet
- Cyclo Al KanesDocument3 pagesCyclo Al KanesIbraim ZekirNo ratings yet
- Carboxylic Acid and Their DerivatesDocument10 pagesCarboxylic Acid and Their Derivatesvita iftitahiyahNo ratings yet
- 11 Chemistry Notes Ch13 HydrocarbonsDocument23 pages11 Chemistry Notes Ch13 HydrocarbonsSahil ChaudharyNo ratings yet
- CARBONYL CONDENSATION REACTIONS 2 (10 Mei 2013)Document34 pagesCARBONYL CONDENSATION REACTIONS 2 (10 Mei 2013)Mammy Nya AllyaNo ratings yet
- AlkeneDocument12 pagesAlkeneAditya NandaNo ratings yet
- Organic Chemistry - Alkanes: Hydrocarbons (Compounds Containing Only C and H)Document6 pagesOrganic Chemistry - Alkanes: Hydrocarbons (Compounds Containing Only C and H)Jojo LeongNo ratings yet
- Physical Organic Chemistry—Ii: Specially Invited Lectures Presented at the Second IUPAC Conference on Physical Organic Chemistry Held at Noordwijkerhout, Netherlands, 29 April–2 May 1974From EverandPhysical Organic Chemistry—Ii: Specially Invited Lectures Presented at the Second IUPAC Conference on Physical Organic Chemistry Held at Noordwijkerhout, Netherlands, 29 April–2 May 1974Th. J. De BoerNo ratings yet
- Practice Makes Perfect in Chemistry: Oxidation-ReductionFrom EverandPractice Makes Perfect in Chemistry: Oxidation-ReductionRating: 5 out of 5 stars5/5 (1)
- Practice Makes Perfect in Chemistry: Acids, Bases, and Salts with AnswersFrom EverandPractice Makes Perfect in Chemistry: Acids, Bases, and Salts with AnswersNo ratings yet
- Practice Makes Perfect in Chemistry: Organic ChemistryFrom EverandPractice Makes Perfect in Chemistry: Organic ChemistryRating: 3 out of 5 stars3/5 (1)
- Selected Constants: Oxidation–Reduction Potentials of Inorganic Substances in Aqueous SolutionFrom EverandSelected Constants: Oxidation–Reduction Potentials of Inorganic Substances in Aqueous SolutionNo ratings yet
- Short StatureDocument32 pagesShort Statureclaimstudent3515100% (1)
- Dr. Shazia Aurangzeb Assistant Professor in Paeds KTHDocument42 pagesDr. Shazia Aurangzeb Assistant Professor in Paeds KTHclaimstudent3515No ratings yet
- A. Flutter, AFDocument80 pagesA. Flutter, AFclaimstudent3515No ratings yet
- Two Concepts of FreedomDocument28 pagesTwo Concepts of Freedomclaimstudent3515No ratings yet
- Fine Needle Aspiration Cyto (FNAC): Procedure, Uses, RisksDocument90 pagesFine Needle Aspiration Cyto (FNAC): Procedure, Uses, Risksclaimstudent3515No ratings yet
- First Steps For Managing An Outbreak of Acute Diarrhoea: Don'T ForgetDocument8 pagesFirst Steps For Managing An Outbreak of Acute Diarrhoea: Don'T Forgetclaimstudent3515No ratings yet
- Fine Needle Aspiration Cyto (FNAC): Procedure, Uses, RisksDocument90 pagesFine Needle Aspiration Cyto (FNAC): Procedure, Uses, Risksclaimstudent3515No ratings yet
- EEG Spike-Wave Pattern in Childhood EpilepsyDocument4 pagesEEG Spike-Wave Pattern in Childhood EpilepsyjumasriNo ratings yet
- Handling Specimens for PathologyDocument23 pagesHandling Specimens for Pathologyclaimstudent3515No ratings yet
- 5 - Ventricular Tachyarrhythmias DoneDocument79 pages5 - Ventricular Tachyarrhythmias Doneclaimstudent3515No ratings yet
- Acuteabdomenseminar 160617111757 PDFDocument38 pagesAcuteabdomenseminar 160617111757 PDFclaimstudent3515No ratings yet
- 12 - Pitutary DiseasesDocument38 pages12 - Pitutary Diseasesclaimstudent3515No ratings yet
- Whoqol: User ManualDocument106 pagesWhoqol: User Manualclaimstudent3515No ratings yet
- Learning Professionalism PerspectivesDocument8 pagesLearning Professionalism Perspectivesclaimstudent3515No ratings yet
- Measuring Health: Methods for Assessing Quality of Life and Health OutcomesDocument44 pagesMeasuring Health: Methods for Assessing Quality of Life and Health Outcomesclaimstudent3515No ratings yet
- Cerebral Palsy in Pakistani Children: A Hospital Based SurveyDocument8 pagesCerebral Palsy in Pakistani Children: A Hospital Based Surveyclaimstudent3515No ratings yet
- Zeeshan QureshiDocument4 pagesZeeshan Qureshiclaimstudent3515No ratings yet
- PedsQL4 0PCDocument2 pagesPedsQL4 0PCRuth FebrinaNo ratings yet
- Improving Bedside Teaching Findings From A Focus.9Document8 pagesImproving Bedside Teaching Findings From A Focus.9claimstudent3515No ratings yet
- Thyroid CADocument10 pagesThyroid CAclaimstudent3515No ratings yet
- Graphical Analysis and Errors - MBLDocument7 pagesGraphical Analysis and Errors - MBLclaimstudent3515No ratings yet
- Calcium For Mbbs StudentsDocument17 pagesCalcium For Mbbs Studentsclaimstudent3515No ratings yet
- Lec24 Conservation LawsDocument20 pagesLec24 Conservation LawsReddyvari VenugopalNo ratings yet
- Bachelor of Medicine and Bachelor of Surgery (MB BS)Document24 pagesBachelor of Medicine and Bachelor of Surgery (MB BS)claimstudent3515No ratings yet
- Lesson 33: Horizontal & Vertical Circular ProblemsDocument5 pagesLesson 33: Horizontal & Vertical Circular Problemsclaimstudent3515No ratings yet
- MutationsDocument13 pagesMutationsclaimstudent3515No ratings yet
- Which Stats TestDocument2 pagesWhich Stats Testaby251188No ratings yet
- Lesson 33: Horizontal & Vertical Circular ProblemsDocument5 pagesLesson 33: Horizontal & Vertical Circular Problemsclaimstudent3515No ratings yet
- Magnetic Field FactsheetDocument4 pagesMagnetic Field Factsheetclaimstudent3515No ratings yet
- Lec24 Conservation LawsDocument20 pagesLec24 Conservation LawsReddyvari VenugopalNo ratings yet
- Kalinga University Organo-transition & bioinorganic QuizDocument13 pagesKalinga University Organo-transition & bioinorganic QuizLokesh BhoiNo ratings yet
- Major Organic Reactions ChapterDocument63 pagesMajor Organic Reactions ChapterdagmawiNo ratings yet
- Ass 2012Document6 pagesAss 2012api-252561013No ratings yet
- Benzene and Aromatic Compounds-fazli-In ClassDocument57 pagesBenzene and Aromatic Compounds-fazli-In Classjokowi123No ratings yet
- AROMATIC SUBSTITUTION REACTIONSDocument54 pagesAROMATIC SUBSTITUTION REACTIONSTariq ZiaNo ratings yet
- Hydrocarbons Derivatives - Alkyl Halide - Aryl Halide PDFDocument15 pagesHydrocarbons Derivatives - Alkyl Halide - Aryl Halide PDFAhmed HammadNo ratings yet
- Halogenation Reactions GuideDocument3 pagesHalogenation Reactions GuideAleem AhmedNo ratings yet
- Pradeep Chemistry Class 11 Vol 2 2023-2024Document843 pagesPradeep Chemistry Class 11 Vol 2 2023-2024laptopemail70No ratings yet
- Alkyl Halides and Amines Mcqs KeyDocument3 pagesAlkyl Halides and Amines Mcqs KeySameer HussainNo ratings yet
- BSC3 Syllabus PDFDocument71 pagesBSC3 Syllabus PDFSiva KiranNo ratings yet
- MSC Chemistry SyllabusDocument96 pagesMSC Chemistry SyllabusA.kNo ratings yet
- 4 CBSQDocument11 pages4 CBSQShauryaNo ratings yet
- Chapter 12Document23 pagesChapter 12Hải NguyễnNo ratings yet
- Chapter 16Document51 pagesChapter 16ادهم ادهمNo ratings yet
- Fiitjee Aits PaperDocument23 pagesFiitjee Aits Paperabhimanyu singhNo ratings yet
- Ligand substitution mechanisms and rate laws in octahedral complexesDocument42 pagesLigand substitution mechanisms and rate laws in octahedral complexesLUIS CARLOS ROMERO ZAPATA100% (1)
- CHE 04 - EN OrganicChemistry IIDocument60 pagesCHE 04 - EN OrganicChemistry IIMadiha RubabNo ratings yet
- Sanjay Gupta: AlcoholsDocument22 pagesSanjay Gupta: AlcoholsSanjay GuptaNo ratings yet
- Hydrocarbons NotesDocument15 pagesHydrocarbons Notesarjunrkumar2024No ratings yet
- Mike's Videos - Organic Chemistry Lesson OutlineDocument64 pagesMike's Videos - Organic Chemistry Lesson Outlinekjk khkNo ratings yet
- MSC Chemistry SyllabusDocument55 pagesMSC Chemistry Syllabusaniruddh pratapNo ratings yet
- Classfcation of Rearrangement PDFDocument6 pagesClassfcation of Rearrangement PDFUsama Tahir0% (1)
- 02 - Alkyl Halide - Aryl Halide (Questions)Document48 pages02 - Alkyl Halide - Aryl Halide (Questions)Sidhiprada PradhanNo ratings yet
- Organic SL With AnswersDocument38 pagesOrganic SL With Answerswakoaisha2No ratings yet
- Advanced Organic Chemistry - I (MPC 102T) Unit-I: Basic Aspects of Organic Chemistry Types of Reaction Mechanisms and Methods of Determining ThemDocument29 pagesAdvanced Organic Chemistry - I (MPC 102T) Unit-I: Basic Aspects of Organic Chemistry Types of Reaction Mechanisms and Methods of Determining ThemEbaieyong SamuelNo ratings yet
- Haloalkanes and Haloarenes: Short Notes Classification of Halogen DerivativeDocument7 pagesHaloalkanes and Haloarenes: Short Notes Classification of Halogen DerivativeSriram LycanNo ratings yet
- Aromatic Chemistry Flash CardsDocument10 pagesAromatic Chemistry Flash CardsSherey FathimathNo ratings yet
- (Paper: 01: Inorganic & Physical Chemistry) : Veer Narmad South Gujarat University, SuratDocument9 pages(Paper: 01: Inorganic & Physical Chemistry) : Veer Narmad South Gujarat University, SuratMihir GamitNo ratings yet
- Chapter 16: Benzene - Electrophilic Aromatic Substitution: Chem231 Study Notes On McmurryDocument20 pagesChapter 16: Benzene - Electrophilic Aromatic Substitution: Chem231 Study Notes On McmurrykjjkimkmkNo ratings yet