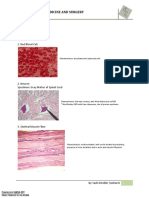
Professional Documents
Culture Documents
Collagen Levels of Structure
Uploaded by
truti0 ratings0% found this document useful (0 votes)
33 views0 pagesCollagen is the most abundant protein in the human body, accounting for around 30% of the total amount of protein. Collagen is a family of around 20 related proteins, which form triple helices through three polypeptide chains winding around each other in a rope-like structure. The tough collagen fibres formed by Type 1 collagen give tendons or ligaments their high tensile strength, but other types of collagen form smaller, or more branched structures in the extracellular matrix.
Original Description:
Copyright
© Attribution Non-Commercial (BY-NC)
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCollagen is the most abundant protein in the human body, accounting for around 30% of the total amount of protein. Collagen is a family of around 20 related proteins, which form triple helices through three polypeptide chains winding around each other in a rope-like structure. The tough collagen fibres formed by Type 1 collagen give tendons or ligaments their high tensile strength, but other types of collagen form smaller, or more branched structures in the extracellular matrix.
Copyright:
Attribution Non-Commercial (BY-NC)
Available Formats
Download as PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
33 views0 pagesCollagen Levels of Structure
Uploaded by
trutiCollagen is the most abundant protein in the human body, accounting for around 30% of the total amount of protein. Collagen is a family of around 20 related proteins, which form triple helices through three polypeptide chains winding around each other in a rope-like structure. The tough collagen fibres formed by Type 1 collagen give tendons or ligaments their high tensile strength, but other types of collagen form smaller, or more branched structures in the extracellular matrix.
Copyright:
Attribution Non-Commercial (BY-NC)
Available Formats
Download as PDF, TXT or read online from Scribd
You are on page 1of 0
page 1/6
JPK Instruments AG - all rights reserved
www.jpk.com
Rachel Owen
tel.: +49 30 5331 12081
NanoWizard, CellHesion, TAO, BioMAT, NanoTracker and ForceRobot
are trademarks or registered trademarks of JPK Instruments AG
Collagen: levels of structure and alignment
Collagen structure and function
Collagen is the most abundant protein in the human body,
accounting for around 30% of the total amount of protein.
Collagen is a structural support for most tissues in the
body as the extracellular matrix, and is particularly
abundant in connective tissue. Skin, for example, is
around 75% collagen, and collagen therefore has a vital
role in many processes such as wound healing. Collagen
production or mineralisation are the basis for the formation
of cartilage, tendons or bones. Cells in all the other tissues
of the body are also surrounded by finer collagen
structures through the extracellular matrix, so collagen has
a vital role also in cell proliferation, migration and
differentiation.
Collagen is a family of around 20 related proteins, which
form triple helices through three polypeptide chains
winding around each other in a rope-like structure. These
triple-stranded nanometre-sized protofibrils can bind
together to form different types of higher level structures.
The tough collagen fibres formed by Type 1 collagen give
tendons or ligaments their high tensile strength, but other
types of collagen form smaller, or more branched
structures in the extracellular matrix. Collagen is involved
in the structure or function of many tissues, so there are
many diseases associated with collagen malfunction. A
genetic defect called Alport syndrome affecting Type IV
collagen, for example, causes malfunction of the glomeruli
in the kidneys, as well as eye and ear problems, and
generally leads to kidney failure.
The structure of Type 1 collagen fibrils is sketched
schematically in Figure 1. Three polypeptide chains wind
together to form a stiff helical structure. These collagen
molecules then align along the helix axis and group as a
bundle to form the collagen fibrils. These collagen fibrils
can also align laterally to form bundles on a higher order of
structure and make up the tough micron-sized collagen
fibres found in ligaments. A characteristic feature of the
collagen fibrils is their banded structure. The diameter of
the fibril changes slightly along the length, with a highly
reproducible D-band repeat of approximately 67nm.
Fig 1. Schematic diagram of Type I collagen fibril structure.
Helical collagen molecules form from three polypeptide chains,
and these associate laterally to form collagen fibrils with a
characteristic banded structure.
Aside from its natural function in the body, collagen has
also been used in technical and medical applications as a
model physiological surface for cell culture, implant
biocompatibility and directed cell growth.
Naturally formed collagen tissue
The classic banded fibril structures can be seen clearly in
rat tail collagen, which demonstrates the high degree of
structure and regularity possible in collagen fibres. Figure
2 shows height and vertical deflection images of rat tail
collagen fibres, for a 2 x 2 micron scan area. In this image,
many of the fibres are lying parallel to each other, although
a few are crossed over in the lower left hand corner. The
cross-section image below shows a section along the
central fibre axis. The red markers are 1345 nm apart,
which corresponds to 20 D-band repeats. This gives a
value for this sample of 67.3 nm for the repeat period.
Changes in the repeat period due to various mutations can
be important to measure accurately.
Polypeptide chain
Triple-stranded collagen
molecule
Collagen fibril
1.5 nm
10-300 nm
67 nm
~ 300 nm
page 2/6
JPK Instruments AG - all rights reserved
www.jpk.com
Rachel Owen
tel.: +49 30 5331 12081
NanoWizard, CellHesion, TAO, BioMAT, NanoTracker and ForceRobot
are trademarks or registered trademarks of JPK Instruments AG
Fig 2. Height and vertical deflection images of a 2 x 2 micron scan
area on a rat tail collagen sample. The cross-section shows 20
repeats of the D-banding marked with red lines, giving and
average period of 67.3 nm.
Figure 3 shows smaller (600 x 600 nm) area scans of the
same sample, again height and vertical deflection signals,
to show the higher order structure that is visible. The third
image is part of the height scan after it has been high-pass
filtered to remove the background curvature of the fibre.
This shows the smaller features more clearly, and the
cross-section image shows the spacing of the small
structures on the surface. The height scale for the cross-
section is not absolute, since the background fibre
curvature has been removed. The lateral size of the
features is around 7 nm. Example features are marked
with red lines with a separation of 6.7 nm.
Fig 3. Height and vertical deflection images of a 600 x 600 nm
area of a rat collagen fibril. Besides the transverse banding, there
is also structure visible across the fibre, as shown in the cross-
section in the lower part.
Deposited collagen fibrils substrates for
bone cell studies
Isolated collagen fibrils from a bovine tendon sample
(deposited at low density on APTES-coated glass) are
shown in Figure 4. The images are courtesy of Prof. M.
Horton, Bone and Mineral Center of University College
London and the London Centre for Nanotechnology. The
optical image (top) shows the AFM cantilever and the
larger collagen fibrils are also visible using optical phase
contrast. The two large AFM images show height and
vertical deflection signals for a 100 x 100 micron scan
area. Most of the fibrils form smoothly curving structures.
A smaller (5 x 5 micron) scan is also shown as a 3D height
image, where the fibril periodicity can be seen.
Height
Vertical deflection
Height cross-section
Height Vertical deflection
Height cross-section (background curvature removed)
page 3/6
JPK Instruments AG - all rights reserved
www.jpk.com
Rachel Owen
tel.: +49 30 5331 12081
NanoWizard, CellHesion, TAO, BioMAT, NanoTracker and ForceRobot
are trademarks or registered trademarks of JPK Instruments AG
Fig 4. Bovine tendon collagen fibrils on APTES-glass, images
courtesy of Prof. M. Horton, Bone and Mineral Center UCL and
London Centre for Nanotechnology. Optical phase contrast (top),
100 x 100 micron scan area height and deflection AFM images
(centre), and 5 x 5 micron 3D height image (bottom).
Collagen structures within natural bone
Fig 5. Bone collagen structure revealed by osteoclasts removing
collagen and minerals from a slice of cortical bone. Images
courtesy of Bozec and Horton [2], UCL and London Centre for
Nan technology. 4 x 4 micron scan area for both large AFM
images (521 nm z-range height, 300mV z-range deflection image).
3-D height projection zoom into osteocyte lacuna.
Optical
(phase contrast)
Vertical
deflection
Height
(3-D projection)
Height
Vertical deflection
Height (3-d projection)
Height
page 4/6
JPK Instruments AG - all rights reserved
www.jpk.com
Rachel Owen
tel.: +49 30 5331 12081
NanoWizard, CellHesion, TAO, BioMAT, NanoTracker and ForceRobot
are trademarks or registered trademarks of JPK Instruments AG
Despite the apparently stability of bone, in fact the bone in
the skeleton of a living human is constantly being
remodelled. Osteoblasts and osteoclasts are cells that are
responsible for the formation and resorption of bone, and
together they maintain a dynamic equilibrium in the
skeleton of healthy adults [1,2]. Collagen has a vital role in
these processes, since it forms the structural protein
framework of bone, and also has a role in signalling
between the osteoblasts and osteoclasts.
Figure 5 shows AFM images of a slice of cortical bone
where the bone collagen structure has been revealed by
osteoclasts removing collagen and minerals. Laterally
associated collagen fibres with clear D-banding are seen in
the topography and error images. An osteocyte lacuna is
seen within the bone (ringed). Images courtesy of Bozec
and Horton, UCL and London Centre for Nanotechnology
[2]. The upper AFM images show a 4 x 4 micron area with
a 521 nm z-range for the height image and a 300 mV z-
range for the deflection image. The lower image shows a
3-D height projection zoom into the osteocyte lacuna, the
resorption pit formed by removal of collagen and minerals.
Using the AFM, the fibril arrangement, periodicity and
diameter can be measured statistically, giving the
possibility to study subtle differences between healthy or
diseased tissues [2].
Deposited collagen film for implants
Various methods have been developed to deposit collagen
onto materials to form biocompatible surfaces, or for
directed cell growth. The collagen can be adsorbed
relatively straightforwardly to surfaces such as glass, but it
is more difficult to attach collagen stably to synthetic
materials such as silicone. These kinds of composite
structures can be used to promote wound healing and
improve biocompatibility of materials for implants,
particularly vascular grafts [3,4]. The crosslinked collagen
can be treated with molecules such as heparin to reduce
thrombosis activation, or fibroblast growth factor to
promote endothelial cell seeding [3].
The images in Figure 6 show a 10 x 10 micron overview of
a sample of collagen-coated silicone film (sample courtesy
of M. J. B. Wissink, University of Twente, The
Netherlands). The sample was imaged under phosphate
buffered saline (PBS) solution (height and amplitude
images for intermittent contact mode in liquid). The larger
collagen fibrils can be seen lying in a smoother matrix.
The collagen is very soft in liquid, since it contains a high
proportion of water.
Fig 6. Intermittent contact mode image in liquid (10 x 10 microns)
of a collagen-coated silicone film. Sample courtesy of M. J. B.
Wissink, University of Twente, The Netherlands.
On smaller images, the details of the D-banding structure
can be seen. Height, amplitude and phase images of the
same sample are shown in Figure 7 for a smaller scan
area (2 x 2 microns). The D-banding can be seen in all
three images for the collagen fibres, which are lying
diagonally across the image in the background matrix.
Although the whole collagen layer is very soft, the 67 nm
banding can be seen clearly.
Height
Amplitude
page 5/6
JPK Instruments AG - all rights reserved
www.jpk.com
Rachel Owen
tel.: +49 30 5331 12081
NanoWizard, CellHesion, TAO, BioMAT, NanoTracker and ForceRobot
are trademarks or registered trademarks of JPK Instruments AG
Fig 8. 2 x 2 micron intermittent contact mode scan of collagen-
coated silicone in buffer (sample courtesy of M.J.B. Wissink,
University of Twente, The Netherlands).
Aligned collagen layers and directed cell
growth
Collagen molecules can be assembled and adsorbed onto
mica supports to coat the surface evenly in a thin (~ 3 nm)
flat layer [5]. Around five individual collagen molecules
associate form microfibrils, with a lateral size of around 3-5
nm. A monolayer of these microfibrils can then be
adsorbed to the surface to form a nanostructured,
biologically active surface. These microfibrils are also
likely to be an intermediate stage in the formation of the
larger collagen fibres seen in natural tissue.
The first level of organisation is to align the collagen fibrils
so that there is an overall orientation in the surface layer,
which can be achieved through adsorbing under conditions
of hydrodynamic flow [5]. Within a certain time after
deposition, the orientation can also be manipulated using
the AFM tip [6]. Figure 9 shows a 750 x 750 nm
intermittent contact mode height image of unfixed collagen
in buffer (z-range 5.5 nm). The sample is courtesy of A.
Taubenberger, D.J. Mller Cellular Machines group,
Technical University Dresden. The fibres are all oriented in
the same direction, and the 67 nm D-repeat can be seen
on the individual fibres. In this case there is no large-scale
alignment of the D-repeat between adjacent fibrils.
Fig 9. Intermittent contact mode image of unfixed collagen in
buffer (750 x 750 nm, z-range 5.5 nm). Sample courtesy of A.
Taubenberger, D.J. Mller Cellular Machines group, TU Dresden.
Height
Amplitude
Phase
Height
page 6/6
JPK Instruments AG - all rights reserved
www.jpk.com
Rachel Owen
tel.: +49 30 5331 12081
NanoWizard, CellHesion, TAO, BioMAT, NanoTracker and ForceRobot
are trademarks or registered trademarks of JPK Instruments AG
A further level of organisation can be achieved if the D-
repeat banding of the collagen microfibrils is also aligned,
as in natural tissue. The alignment or non-alignment of the
D-repeats is sensitive to the ionic composition of the
deposition buffer, and alignment is seen in solutions
mimicking cytoplasmic or extracellular environments of
eukaryotic cells [6]. In the height and vertical deflection
images shown in Figure 10, the molecules have been
deposited so that there is an overall alignment of the D-
repeat bands of adjacent collagen fibrils. The images in
Figure 10 are courtesy of A. Taubenberger, D.J. Mller
Cellular Machines group, TU Dresden. In this sample, the
collagen does not completely cover the surface and gaps
can be seen where the glass surface is visible beneath.
Fig 10. Aligned unfixed collagen fibrils in liquid. Images courtesy
of A. Taubenberger, D.J. Mller Cellular Machines group, TU
Dresden.
These surfaces have some of the nanostructural properties
of natural collagen fibres, laid down as a thin coating. The
alignment of the D-repeats also produces a bioactive
surface. Fibroblast cells cultivated on the collagen where
the fibrils are oriented, but not aligned, show no particular
orientation of their shape or growth direction. When the D-
repeats are also aligned, however, the fibroblasts show
highly oriented motility along the axis direction, and have
an elongated shape in this direction. The texture of the
surface caused by large-scale orientation of the fibrils
alone was not sufficient to control cell motility, and this
suggests a biological function for the D-band alignment in
guiding cell growth [7].
Conclusions
The AFM is a powerful tool for observing the levels of
alignment and organisation in collagen structures. Natural
collagen structures, isolated fibrils and novel biomaterials
can all be studied under physiological conditions. These
measurements can illuminate fundamental biological
questions, as well as test new biocompatible or specifically
bioactive composites.
Literature
[1] Charras G.T., Lehenkari P. and Horton M.A. "Atomic force
microscopy can be used to mechanically stimulate
osteoblasts and evaluate cellular strain distributions"
Ultramicroscopy 86: 269-278 (2000).
[2] Bozec L., de Groot J., Odlyha M., Nicholls B., Nesbitt S.,
Flanagan A and Horton M.A. "Atomic force microscopy of
collagen structure in bone and dentine revealed by osteoclast
resorption" Ultramicroscopy, 105(1-4):79-89 (2005).
[3] Wissink M.J.B., Beernink R., Poot A.A., Engbers G.H.M.,
Beugeling T., van Aken W.G. and Feijen J. "Improved
endothelialization of vascular grafts by local release of growth
factor from heparinized-collagen matrices" J. Controlled
Release 64: 103-114 (2000).
[4] Wissink M.J.B., Beernink R., Pieper J.S., Poot A.A., Engbers
G.H.M., Beugeling T., van Aken W.G. and Feijen J.
"Immobilization of heparin to EDC/NHS-crosslinked collagen.
Characterization and in vitro evaluation." Biomaterials 22:
151-163 (2001).
[5] Jiang F., Hrber H., Howard J. and Mller D.J. "Assembly of
collagen into microribbons: effects of pH and electrolytes" J.
Struct. Biol. 148(3): 268-278 (2004).
[6] Jiang F., Khairy K., Poole K., Howard J., Mller D.J.
"Creating nanoscopic collagen matrices using atomic force
microscopy" Microsc. Res. Tech. 64: 435-440 (2004).
[7] Poole K., Khairy K., Friedrichs J., Franz C., Cisneros D.A.,
Howard J. and Mller D.J. "Molecular-scale topographic cues
induce the orientation and direct movement of fibroblasts on
two-dimensional collagen surfaces" J. Mol. Biol. 349: 380-
386 (2005).
Height
Vertical deflection
You might also like
- 1 s2.0 S0305440314000879 Main PDFDocument7 pages1 s2.0 S0305440314000879 Main PDFtrutiNo ratings yet
- Verbos Conjugados PDFDocument2 pagesVerbos Conjugados PDFBrayan FavelaNo ratings yet
- FTP PDFDocument11 pagesFTP PDFtrutiNo ratings yet
- Walker 2008 Cranial SexDocument12 pagesWalker 2008 Cranial SextrutiNo ratings yet
- Heady Business JMA 2004-LibreDocument25 pagesHeady Business JMA 2004-LibretrutiNo ratings yet
- Nakamura Pels Proof - PDFDocument38 pagesNakamura Pels Proof - PDFtrutiNo ratings yet
- Fetch ObjectDocument16 pagesFetch ObjecttrutiNo ratings yet
- 704 FTPDocument9 pages704 FTPtrutiNo ratings yet
- And Gabriele: Ilium, Calcium, PhosDocument5 pagesAnd Gabriele: Ilium, Calcium, PhostrutiNo ratings yet
- Evaluation of Dental Non-Metric Traits in A Medieval Population From Ibiza (Spain)Document13 pagesEvaluation of Dental Non-Metric Traits in A Medieval Population From Ibiza (Spain)trutiNo ratings yet
- Dabbs and Moore JansenDocument4 pagesDabbs and Moore JansentrutiNo ratings yet
- Herxheim CannibalsDocument15 pagesHerxheim CannibalsGergely GortvaNo ratings yet
- Correlation Compression The Proximal External Degeneration: OF Gross OFDocument10 pagesCorrelation Compression The Proximal External Degeneration: OF Gross OFtrutiNo ratings yet
- Klales Et Al 2012 Phenice-2Document11 pagesKlales Et Al 2012 Phenice-2truti100% (1)
- Bioarchaeology In: Oxford Bibliographies Online - Anthropology (2013)Document31 pagesBioarchaeology In: Oxford Bibliographies Online - Anthropology (2013)truti100% (1)
- Osteology Method Statementrevised 2012Document64 pagesOsteology Method Statementrevised 2012Michelle Regan Feiszli100% (1)
- Sexing The Pubis': A Newly Developed Visual Method ofDocument5 pagesSexing The Pubis': A Newly Developed Visual Method oftrutiNo ratings yet
- New Perspectives in Forensic Anthropology: Dennis C. Dirkmaat, Luis L. Cabo, Stephen D. Ousley, and Steven A. SymesDocument20 pagesNew Perspectives in Forensic Anthropology: Dennis C. Dirkmaat, Luis L. Cabo, Stephen D. Ousley, and Steven A. SymestrutiNo ratings yet
- Chapter Nine: Physical Anthropology AnalysisDocument67 pagesChapter Nine: Physical Anthropology AnalysistrutiNo ratings yet
- New Perspectives in Forensic Anthropology: Dennis C. Dirkmaat, Luis L. Cabo, Stephen D. Ousley, and Steven A. SymesDocument20 pagesNew Perspectives in Forensic Anthropology: Dennis C. Dirkmaat, Luis L. Cabo, Stephen D. Ousley, and Steven A. SymestrutiNo ratings yet
- And Gabriele: Ilium, Calcium, PhosDocument5 pagesAnd Gabriele: Ilium, Calcium, PhostrutiNo ratings yet
- A New Calibration of The XRD Technique For The Study of Archaeological Burned Human RemainsDocument8 pagesA New Calibration of The XRD Technique For The Study of Archaeological Burned Human RemainstrutiNo ratings yet
- Carlos Lorenzo & Juan Luis ArsuagaDocument22 pagesCarlos Lorenzo & Juan Luis ArsuagatrutiNo ratings yet
- 3.-Human Osteological MethodsDocument30 pages3.-Human Osteological Methodstruti100% (1)
- Effect of Heating Process On Micro Structure Level of Cortical Bone Prepared For Compositional AnalysisDocument9 pagesEffect of Heating Process On Micro Structure Level of Cortical Bone Prepared For Compositional AnalysistrutiNo ratings yet
- Midwest Anthropology Students Journal Accepting SubmissionsDocument1 pageMidwest Anthropology Students Journal Accepting SubmissionstrutiNo ratings yet
- 2 Manifold IAPO Bulletin 2012-2-1Document19 pages2 Manifold IAPO Bulletin 2012-2-1trutiNo ratings yet
- Oa 2282Document7 pagesOa 2282trutiNo ratings yet
- Chapter Nine: Physical Anthropology AnalysisDocument67 pagesChapter Nine: Physical Anthropology AnalysistrutiNo ratings yet
- Shoe Dog: A Memoir by the Creator of NikeFrom EverandShoe Dog: A Memoir by the Creator of NikeRating: 4.5 out of 5 stars4.5/5 (537)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeFrom EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeRating: 4 out of 5 stars4/5 (5794)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceFrom EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceRating: 4 out of 5 stars4/5 (890)
- The Yellow House: A Memoir (2019 National Book Award Winner)From EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Rating: 4 out of 5 stars4/5 (98)
- The Little Book of Hygge: Danish Secrets to Happy LivingFrom EverandThe Little Book of Hygge: Danish Secrets to Happy LivingRating: 3.5 out of 5 stars3.5/5 (399)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryFrom EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryRating: 3.5 out of 5 stars3.5/5 (231)
- Never Split the Difference: Negotiating As If Your Life Depended On ItFrom EverandNever Split the Difference: Negotiating As If Your Life Depended On ItRating: 4.5 out of 5 stars4.5/5 (838)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureFrom EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureRating: 4.5 out of 5 stars4.5/5 (474)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersFrom EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersRating: 4.5 out of 5 stars4.5/5 (344)
- Grit: The Power of Passion and PerseveranceFrom EverandGrit: The Power of Passion and PerseveranceRating: 4 out of 5 stars4/5 (587)
- On Fire: The (Burning) Case for a Green New DealFrom EverandOn Fire: The (Burning) Case for a Green New DealRating: 4 out of 5 stars4/5 (73)
- The Emperor of All Maladies: A Biography of CancerFrom EverandThe Emperor of All Maladies: A Biography of CancerRating: 4.5 out of 5 stars4.5/5 (271)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaFrom EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaRating: 4.5 out of 5 stars4.5/5 (265)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreFrom EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreRating: 4 out of 5 stars4/5 (1090)
- Team of Rivals: The Political Genius of Abraham LincolnFrom EverandTeam of Rivals: The Political Genius of Abraham LincolnRating: 4.5 out of 5 stars4.5/5 (234)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyFrom EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyRating: 3.5 out of 5 stars3.5/5 (2219)
- The Unwinding: An Inner History of the New AmericaFrom EverandThe Unwinding: An Inner History of the New AmericaRating: 4 out of 5 stars4/5 (45)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)From EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Rating: 4.5 out of 5 stars4.5/5 (119)
- Her Body and Other Parties: StoriesFrom EverandHer Body and Other Parties: StoriesRating: 4 out of 5 stars4/5 (821)
- Cancer Cell AssayDocument8 pagesCancer Cell AssayPracren TamneNo ratings yet
- Essentials of Single-Cell Analysis: Fan-Gang Tseng Tuhin Subhra Santra EditorsDocument415 pagesEssentials of Single-Cell Analysis: Fan-Gang Tseng Tuhin Subhra Santra EditorsShouvik ChaudhuriNo ratings yet
- Lab1 Histology Bios E1b 2016cDocument13 pagesLab1 Histology Bios E1b 2016cwekrjbhweiuNo ratings yet
- Cytoskeleton and Cellular StructuresDocument6 pagesCytoskeleton and Cellular StructuresArif NabeelNo ratings yet
- 3D Extracellular Matrix Mimics: Fundamental Concepts and Role of Materials Chemistry To in Uence Stem Cell FateDocument27 pages3D Extracellular Matrix Mimics: Fundamental Concepts and Role of Materials Chemistry To in Uence Stem Cell Fateshinichi kudoNo ratings yet
- The Dental Plaque Biofilm MatrixDocument25 pagesThe Dental Plaque Biofilm MatrixTasha FarahNo ratings yet
- Unit 1 CELLDocument262 pagesUnit 1 CELLKripa SusanNo ratings yet
- Evaluating The Effects of Injected Calcium Hydroxylapatite On Changes in Human Skin Elastin and Proteoglycan FormationDocument5 pagesEvaluating The Effects of Injected Calcium Hydroxylapatite On Changes in Human Skin Elastin and Proteoglycan FormationChris LicínioNo ratings yet
- Biofilm FormationDocument12 pagesBiofilm FormationFhaliesha ZakurNo ratings yet
- Growth Factors and Wound Healing 1997Document374 pagesGrowth Factors and Wound Healing 1997Vo Ha Phuong NguyenNo ratings yet
- Muscle Fascia and Force Transmission: Peter P. Purslow, PHDDocument7 pagesMuscle Fascia and Force Transmission: Peter P. Purslow, PHDBangun MakmunNo ratings yet
- BIOCHEMISTRYDocument10 pagesBIOCHEMISTRYShaira Kaye AlimariNo ratings yet
- Histo 1st ShiftDocument25 pagesHisto 1st ShiftIvy CruzNo ratings yet
- Cell Adhesion MoleculesDocument90 pagesCell Adhesion MoleculesPooja Bappanna100% (1)
- Matrix Metalloproteinases (MMPS) :: & Proteins Technical GuideDocument20 pagesMatrix Metalloproteinases (MMPS) :: & Proteins Technical GuidedrmacrohardNo ratings yet
- Biomechanics of Articular Cartilage Lubrication MechanismsDocument30 pagesBiomechanics of Articular Cartilage Lubrication MechanismsLibbyNo ratings yet
- Structure and Function of The Musculoskeletal SystemDocument9 pagesStructure and Function of The Musculoskeletal Systemeka pionitaNo ratings yet
- Test Bank For Biological Science 3rd Canadian Edition Scott Freeman DownloadDocument13 pagesTest Bank For Biological Science 3rd Canadian Edition Scott Freeman Downloadjuliekimbmcnygjrkx100% (29)
- Membranes For Periodontal RegenerationDocument6 pagesMembranes For Periodontal RegenerationinventionjournalsNo ratings yet
- Rohn 2018Document59 pagesRohn 2018LýDiệuNo ratings yet
- Ohns D29527 en F - M3 - 2016 09 29 - 094835 - 1475507735280Document24 pagesOhns D29527 en F - M3 - 2016 09 29 - 094835 - 1475507735280Δημήτριος ΙωαννίδηςNo ratings yet
- Differences between acute and chronic woundsDocument6 pagesDifferences between acute and chronic woundsputriseptinaNo ratings yet
- 003 Part 1 FRCOphth Examination Application Pack 2012Document28 pages003 Part 1 FRCOphth Examination Application Pack 2012arian tejaratNo ratings yet
- Practitioners HandbookDocument134 pagesPractitioners HandbookAnonymous dpxYTE0% (1)
- Molecular Pathogenesis of Oral Submucous Fibrosis - A Collagen Metabolic DisorderDocument8 pagesMolecular Pathogenesis of Oral Submucous Fibrosis - A Collagen Metabolic Disorderporkodi sudhaNo ratings yet
- BCH 4107 - Plant BiochemistryDocument13 pagesBCH 4107 - Plant BiochemistryOLUWASEGUN K AfolabiNo ratings yet
- Chapter 3 - Protocols For Decellularization of Human Amniotic MembraneDocument11 pagesChapter 3 - Protocols For Decellularization of Human Amniotic MembraneravhcolNo ratings yet
- Abstract Vetigel (Reparation)Document2 pagesAbstract Vetigel (Reparation)bayuNo ratings yet
- Final!Document14 pagesFinal!JoshuaNo ratings yet
- The Mechanism of Wound HealingDocument17 pagesThe Mechanism of Wound HealingHanarisha Putri AzkiaNo ratings yet