Professional Documents
Culture Documents
Elements of Modern Style Article
Uploaded by
mamazookeeprCopyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Elements of Modern Style Article
Uploaded by
mamazookeeprCopyright:
Available Formats
Elements of [Modern] Style
Ytterby there are two secret pasts. Two centuries ago, the sleepy village- now dotted with vacation homes belonging to wealthy residents of nearby Stockholm- was a restless mining settlement, shipping out high-grade feldspar for the royal porcelain factories of Europe and quartz to line the blast furnaces springing up across England. It is also the birthplace of some of natures most wondrous and least appreciated chemical elements. The latter story began in 1787, when an amateur geologist named Carl Arrhenius was visiting a mine in Ytterby. He discovered an unusually heavy black rock among the gray outcroppings and, being a man of healthy scientific curiosity, sent a sample for analysis to Johan Gadolin, a prominent chemist at the Royal Academy of Turku in Finland. In 1794 Gadolin concluded that the specimen contained an entirely new element, later named yttrium. By 1879 chemists had isolated six additional elements from the same rock, bringing the grand total in the newly invented periodic table to 70. Three of those elementsytterbium, erbium, and terbium- were simply given additional variants on the name Ytterby, while the other three were named holmium (for Stockholm), scandium, and thulium (both from the Latin for Scandanavia), in the nationalistic fashion then in favor. After a long, lucrative run, the Ytterby quarry was closed in 1933. In many ways, though, the towns influence looms larger than ever. The elements discovered there, known collectively as rare earths, today form the backbone of the modern wired and wireless world- even though you have probably never heard of them. th The name rare earths made sense to the 19 century mind; rare because it seemed at first that they came only from Scandanavia, and earths because they occurred in an earthly oxide form from which it was exceptionally hard to obtain the pure metal. Today it is clear that the rare earths are hardly rare. The most th common of them, cerium, ranks 25 in abundance in the earths crust, one place ahead of homely copper. Yttrium is twice as abundant as lead; all of the rareearth metals (with the exception of radioactive promethium) are more common than
by Hugh Aldersey-Williams
Six valuable rare earths, shown in powdered oxide form. Neodumium, a key component of electronic devices, is at front.
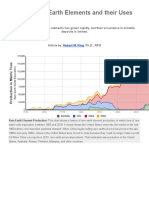
silver. The earths part is also misleading. These elements are actually metals, and quite marvelous ones at that. The warm glow of terbium is essential to high-efficiency compact-fluorescent bulbs. Europium is widely exploited to make vivid displays for laptop computers and smart phones. Rare earths also pop up in more unexpected places like baseball bats, European currency, and night-vision goggles. With their growing popularity comes new value, and even political notoriety. Terbium and europium recently overtook silver in price, reaching $40 an ounce. The growing demand for rare earths has become the subject of numerous government reports and a bill that passed in the House of Representatives. The reason these elements are causing such a stir is not their scarcity but their inaccessibility. Rare earths tend to occur in hard rock such as granites, where they lump together in a uniform way that makes them difficult to extract. Separating out the desired elements demands a toxic and dangerous process, and China has the best infrastructure for doing so economically. China holds about 36 percent of the worlds 110 million tons of recoverable rare-earth ores, with the rest scattered worldwide, principally in the United States, India, Australia, and Russia. Yet China currently produces as much as 97 percent of the worlds rare earth oxides, according to the US Government Accountability Office. Pekka Pyykk, a professor of chemistry at the University of Helsinki, puts it this way:
Elements of [Modern] Style
Not all the deposits are in China, but the processing capacity right now is. Supply would not matter if not for demand, and the demand comes from the unusual electrical properties of the rare earths- or lanthanides, as chemists prefer to call them, because they mostly follow lanthanum in the periodic table of elements. The lanthanides share similar chemical properties because they all react similarly, mostly with their three outer electrons. (An atoms arrangement of electrons is what determines most of its physical and chemical attributes.) Like copper, iron, cobalt, and other more familiar metals, lanthanides form many colored compounds. The magic happens when those outer electrons change energy states and release visible light. But the rare earths are especially valuable for their property of fluorescence: They can absorb light or ultraviolet rays and re-emit the energy as an eerie glow of certain colors specific to each element. The brilliant emission of red and green is the reason why lanthanides are indispensable components of todays television sets and compact fluorescent bulbs. From a technological perspective, a more intriguing trait of the rare earths is that some of them are highly magnetic. Alloyed with other metals, they make extraordinarily strong and compact magnets: perfect for computer hard drives, cordless power tools, microphones, and headphones. An iPod takes a triple sip of rare earths: to store digital music, to recreate it in earbuds, and to display what is playing. An
by Hugh Aldersey-Williams
Life on Lanthanides: Toyota Prius
Toyota has sold 2 million Priuses globally since the cars introduction in 1997, including a million in the United States. Unfortunately, while the worlds most popular hybrid sips gasoline, it devours rare earths. The Prius gets its juice from a about 20 pounds of lanthanum, which shuttles electrons between the batterys positive and negative terminals. Powerful magnets in the Priuss electric motors incorporate neodymium, whose cost shot up 150 percent in the first third of 2011. The price of lanthanum has increased sevenfold, to $60 a pound, over the past year, and in 2010 China briefly suspended rare-earth shipments to Japan in a diplomatic dispute. Toyota is working with the electric car company Tesla Motors to install rare-earth-free motors in future vehicles.
iron alloy containing terbium and dysprosium has a particularly useful property: It expands and contracts efficiently in the presence of a magnetic field. Sensors, actuators, and injectors commonly use such materials, for instance to regulate the flow of gasoline into an auto motive engine. Compact, powerful magnets are key components for efficient generators and electric motors, and this makes rare earths star players in green technology. (Molycorp Minerals- the biggest rare-earth mining operation in the United Statesoptimistically calls them the green elements.) Lanthanide-flecked supermagnets are used to generate electricity in wind turbines. A turbine capable of generating 2.5 megawatts of power typically uses 700 pounds of neodymium. High-speed maglev trains make extensive use of such magnets to levitate the cars above their tracks. The Toyota Prius is practically a rolling exhibit of rare earths, containing eight different elements, for a total of 25 pounds of rare earthiness.
Life on Lanthanides: Baseball Bats
Are you the weakest player on your company softball team? One perfectly legal way to boost your performance is to use a metal bat infused with the rare-earth element scandium. When mixed with aluminum, the silvery-white element shrinks and refines the grains of the metal, making an alloy that is highly resistant to cracking. Just a little bit lets you build a stronger bat without sacrificing resilience and durability, says Dewey Chauvin, director of bat engineering at Easton Sports. You can add more features to the bat, like a longer barrel and hence a larger sweet spot, without incurring weight. But scandium production is so low that the element- which is also used in bicycle frames and lacrosse sticks- can cost hundreds of dollars a pound, limiting its use to top-of-the-line bats priced at $150 or more.
Elements of [Modern] Style
Life on Lanthanides: Smart Phones
The era of ever-shrinking consumer electronics would be virtually impossible without rare earths. Neodymium is one of the most important elements here because of its powerful magnetic properties. In modern cell phones, neodymium is a key ingredient in the wafer-thin permanent magnet that converts electric signals to sound waves in speakers and headsets. It is also used in the small motors that vibrate when you turn your ringer off. Other rare earths are crucial for the vivid displays in smart phones, televisions, and laptops. Europium and terbium emit red and green light, respectively, and their market value rose more than 75 percent in the first four months of 2011. Expect demand to sharpen as color touch screens become more common with the spread of tablets and e-readers. But the commercial versatility of the lanthanides extends far beyond the world of electronics. Cerium is a prime example. It was discovered in 1803 by Swedish chemist Jns Jacob Berzelius, who named it after the asteroid Ceres, discovered two years earlier. (Ceres was then regarded as a new planet and so was much celebrated.) In 1885 the Viennese chemist Carl Auer von Welsbach found that a pinch of cerium oxide, together with thorium oxide, usefully increases the brightness of a gas lamp. Soon after, Auer discovered that a mixture of cerium and iron readily generates sparks. The alloy, which he modestly called Auermetall, is still used to ignite the flame in cigarette lighters. Ceriums applications kept multiplying. A fine powder of cerium oxide, known as opticians rouge, is an excellent abrasive for polishing glass. Cerium compounds form nontoxic red pigments that are used in toys and household products. In medicine, cerium compounds were once administered for everything from tubercular coughing to seasickness; today those treatments are mostly regarded as quackery, but the element still earns its place in hospitals as an antiseptic wash for severe burns. You may have cerium oxide in your kitchen if your oven is a self-cleaning model; it helps break down burned-on leftovers. You certainly have cerium oxide in your car, assuming it is less than 35 years old. The
by Hugh Aldersey-Williams
compound works to convert carbon monoxide emissions in automotive exhaust to less harmful carbon dioxide. As a fine powder mixed into diesel fuel, cerium oxide can also clean up the sooty fumes produced by trucks and buses. Industry consumes 55,000 tons of cerium every year. The element, pulled from a type of weathered granite called monazite, fetches about $60 a pound. Useful and yet so unfamiliar: the average person is still largely in the dark about rare earths, even though many of them are literally casting light into the world. Cerium is a crucial element in the intense, white-light arc lamps used by moviemakers to illuminate the action on-set. Glass doped with cerium oxide effectively absorbs ultraviolet light. That makes it useful for UV-blocking windows and sunglasses, as well as for protecting X-ray equipment from radiation damage. Two other rare-earth metals, neodymium and praseodymium, work their magic at the other end of the spectrum. They impeded infrared light and so are incorporated into welders goggles to protect workers eyes from the heat. These oxides also absorb yellow and green, so glass made with them has a characteristic mauve tinge. More broadly, rare earths add color to our world because of their rich and varied optical properties. The glazes on earthenware dishes and vases commonly contain oxides of tin (white), copper (blue), or iron (amber). But rare-earth oxides can dissolve in the glaze to create more exotic hues, such as pink and lime green, with a sharp, electric appearance that artists love. They can be transparent, which really brings ceramics alive, says David Pier, a ceramicist in Chapel Hill, North Carolina. Adding erbium oxide, for example, gives a subtle tone-
The Mountain Pass mine in California restarted operations earlier this year, providing a rare-earth alternative to China.
Elements of [Modern] Style
more pink lemonade than Pepto-Bismol, as Pier describes it. Colorful fluorescence is the calling card of another rare-earth element with another odd name: europium. Elements were frequently named for the town or region where they were discovered, sometimes in a very provincial manner, as was the case with yttrium. Europium runs against the trend. It was the last of the natural lanthanides to be found, in 1901. In that year, Frenchman Eugne-Anatole Demaray isolated the element as an impurity in samples of another rare earth, samarium (named, indirectly, after Russian mine official V. E. SamarskyBykhovets). In a fit of magnanimity, Demaray decided to name his element not after Paris, where he worked, or after France, but in honor of all Europe. all the rare earths, europium is perhaps the most visible. It is used in the phosphors of display screens, where it emits red and blue light. A little europium is also added to mercury vapor streetlamps to whiten their otherwise cold blue light. It even has found its way into the wallet. Officials at the European Central Bank evidently decided it would be fitting if europiumbased compounds were added to euro banknotes as fluorescent security markings. When the notes are inspected under a banks ultraviolet scanner, pictures of a bridge and the European continent light up in green. The relationship between europium and the euro was not announced; it was revealed only when Freek Suijver and Andries Meijerink, two curious chemists at the University of Utrecht in the Netherlands, put the notes in their spectrometer. They had a good laugh but were unable to determine who concocted this sophisticated inside joke. I learned that the use of europium was intentional, Meijerink says, but I am sorry to say that I have no inside information on who, where, and how it was decided to use the europium luminescence. The importance of the rare earths is no secret in the industrialized world, however. One year ago, when China announced it would limit exports, the commodity price of all the rare earths almost instantly shot up. In just six months the price of samarium oxide quadrupled, to more than $50 a pound in May
by Hugh Aldersey-Williams
of this year, owing to its property as a permanent supermagnet when alloyed with cobalt. Samariums ability to stay strongly magnetic in extreme temperatures makes it a favorite of the military for use in precision-guided weapons. In April, China took steps to keep prices high by extending a national ban on rare-earth exploration and the opening of new mines. For all the dire headlines, the latest rareearth frenzy may be little more than a bubble. Not so long ago, China aggressively pursued cheaper means of isolating rare earths, benefiting from the absence of costly environmental regulations, which is how the country acquired its current near-monopoly. But its dominance could well be temporary. Because of its cheap prices, the competitors closed down, says Pekka Pyykk. In the present situation, new capacity will undoubtedly be restarted. Mountain Pass, California, is one of the places where American rare earths may rise again. Here, Molycorp used to meet much of the worlds demand for these elements. The company shuttered its operations in 2002, but earlier this year it reopened its mine. Another American company, U.S. Rare Earths, owns mineral rights to lanthanide resources in Montana and Idaho; according to a report by the Government Accountability Office, these are in early exploratory stages of development. Mines in these areas may take up to 15 years to bring online, the report states, largely due to the time it takes to comply with multiple state and federal regulations. A visit to other parts of the periodic table could also turn up substitutes for some of the lanthanides. In January, Toyota- spooked by rising prices and supply concerns- announced that it is developing a new type of propulsion, called an induction motor, for future electric and hybrid vehicles. The technology notably does not rely on rare-earth elements. For now, the world is hooked on the lanthanides, and China is the only place that has the processing capacity up and running. As for Ytterby, that mine at least is unlikely to reopen. It is now a historic landmark, located just off the intersection of Terbium and Mine roads. A small sign commemorates
Elements of [Modern] Style
it on the quayside, noting the sites prodigious contribution to chemistry.
by Hugh Aldersey-Williams
Life on Lanthanides: Wind Turbines
Carbon-free electricity gets a big boost from rare earths. Behind the rotors of a wind turbine, wire coils move through a magnetic field to generate current. To maximize efficiency, manufacturers like General Electric often use high-intensity magnets that incorporate rare-earth elements. Some turbines contain 700 pounds of neodymium. Without rare earths, youd have to use larger, heavier magnets, and youd have to build a stronger, more expensive tower to support them, says materials scientist Alex King of the U.S. Department of Energys Ames Laboratory. Last year GE received a $2 million federal grant to develop magnets with only one-fifth the rare-earth content of todays offerings.
You might also like
- Rare EarthDocument10 pagesRare Earthjagadish.kvNo ratings yet
- HTTP WWW - Technologyreview.com Printer Friendly ArticleDocument3 pagesHTTP WWW - Technologyreview.com Printer Friendly ArticleAurelio SartoriNo ratings yet
- The Metal of Ages: A Brief History of CopperDocument5 pagesThe Metal of Ages: A Brief History of CopperDouglay McTay0% (1)
- Written Report in GenChemistryDocument6 pagesWritten Report in GenChemistryAlexisNo ratings yet
- How yttrium is extracted and used in superconductors and lasersDocument11 pagesHow yttrium is extracted and used in superconductors and lasersBagas AdiansyahNo ratings yet
- Modern gold mine extraction process and geochemically scarce metals productionDocument63 pagesModern gold mine extraction process and geochemically scarce metals productionKevinNo ratings yet
- REE Rare EarthsDocument11 pagesREE Rare EarthsRicardo JesusNo ratings yet
- Planetary Metal&Herbal CorrespondencesDocument30 pagesPlanetary Metal&Herbal CorrespondencesCor ScorpiiNo ratings yet
- REE - Rare Earth Elements: Home MetalsDocument10 pagesREE - Rare Earth Elements: Home MetalsMorkizgaNo ratings yet
- Members: Saz, Frederico Toh, Marvin Unahan, Shierwin Jee Borba, Godfrey Ventero, Patrick Siacor, Breezy Mae EyleDocument44 pagesMembers: Saz, Frederico Toh, Marvin Unahan, Shierwin Jee Borba, Godfrey Ventero, Patrick Siacor, Breezy Mae EylemalynNo ratings yet
- Need To FinishDocument6 pagesNeed To FinishCely Marie VillarazaNo ratings yet
- Background of The StudyDocument18 pagesBackground of The StudyCely Marie VillarazaNo ratings yet
- Transition Metals II: (General Chemistry)Document11 pagesTransition Metals II: (General Chemistry)Erica Bianca MirandaNo ratings yet
- Chemistry ProjectDocument4 pagesChemistry ProjectThet HtarNo ratings yet
- Handbook of Extractive Metallurgy: CopperDocument9 pagesHandbook of Extractive Metallurgy: Copper1394203031No ratings yet
- Business of Rare Earth ElementsDocument6 pagesBusiness of Rare Earth ElementsAnanthNo ratings yet
- Stone-Age People Knew Nothing of Metal. Colorful: Hematite Iron Ore Were Found, They Would Be GroundDocument2 pagesStone-Age People Knew Nothing of Metal. Colorful: Hematite Iron Ore Were Found, They Would Be Groundlink2u_007No ratings yet
- Gold Iron Copper OxygenDocument3 pagesGold Iron Copper OxygenfranshuNo ratings yet
- Material WorldDocument188 pagesMaterial WorldYahaya HassanNo ratings yet
- Everything You Need to Know About TungstenDocument24 pagesEverything You Need to Know About TungstenGottumukkala Venkateswara RaoNo ratings yet
- Metals and their properties in materials scienceDocument62 pagesMetals and their properties in materials scienceEdgar GadzikwaNo ratings yet
- Classification of MaterialsDocument113 pagesClassification of Materialsabhinavgiri17No ratings yet
- Byron Capital Markets Rare Earths Industry ReportDocument20 pagesByron Capital Markets Rare Earths Industry Reportgeorg1967No ratings yet
- Copper: Properties, Production and ApplicationsDocument35 pagesCopper: Properties, Production and Applicationsjagan005No ratings yet
- General Chemistry Script and FlowDocument7 pagesGeneral Chemistry Script and FlowdecastroghislaneNo ratings yet
- 2.6.a-Reshaping Our LivesDocument14 pages2.6.a-Reshaping Our Livestwinkledreampoppies50% (2)
- Classification of MaterialsDocument113 pagesClassification of Materialsabhinavgiri17No ratings yet
- Characteristics of Iron OreDocument4 pagesCharacteristics of Iron OreNathan SwamiNo ratings yet
- Vasilyev Metals and ManDocument285 pagesVasilyev Metals and Manatto_11100% (1)
- Chemical Elements Crossword 1Document2 pagesChemical Elements Crossword 1David LockartNo ratings yet
- Group Jupiter 1Document21 pagesGroup Jupiter 1zin GuevarraNo ratings yet
- Invest in Transitional MetalsDocument12 pagesInvest in Transitional MetalsTristan WagnerNo ratings yet
- Invest in Transitional MetalsDocument12 pagesInvest in Transitional MetalsTristan WagnerNo ratings yet
- Unit 2B.1Document16 pagesUnit 2B.1PualeilehuaNo ratings yet
- 2.copper Latin Name:cuprum: 3.gold Latin Name:aurum Symbol:Au When:1848 Where:California Who:James Wilson MarshallDocument4 pages2.copper Latin Name:cuprum: 3.gold Latin Name:aurum Symbol:Au When:1848 Where:California Who:James Wilson MarshallPatricia B. LedesmaNo ratings yet
- Carbon Magic: by David Lincoln AUG 2018Document37 pagesCarbon Magic: by David Lincoln AUG 2018David LincolnNo ratings yet
- CopperDocument18 pagesCopperaimanhazimNo ratings yet
- 10 Copper Facts - Https://egyptiansorcery - ComDocument2 pages10 Copper Facts - Https://egyptiansorcery - ComAravenaNo ratings yet
- Iron and The EnvironmentDocument15 pagesIron and The EnvironmentIzzy ConceptsNo ratings yet
- Native Elements Group - Properties and UsesDocument5 pagesNative Elements Group - Properties and UsesBroadsageNo ratings yet
- MS4SSA IntroductiontoMetalsDocument62 pagesMS4SSA IntroductiontoMetalsSrikanth SrikantiNo ratings yet
- Chapter 2 RRLDocument31 pagesChapter 2 RRLIkamuzu OturanNo ratings yet
- The Importance of Iron in Our Daily LifeDocument38 pagesThe Importance of Iron in Our Daily Lifelamia97100% (1)
- 5 Electricity and Chemistry: ElectrolysisDocument12 pages5 Electricity and Chemistry: Electrolysistotatote034No ratings yet
- Nickel OccurrenceDocument16 pagesNickel OccurrenceArfinsa AinurzanaNo ratings yet
- A Closer Look at Silicon - Chemistry Book for Elementary | Children's Chemistry BooksFrom EverandA Closer Look at Silicon - Chemistry Book for Elementary | Children's Chemistry BooksNo ratings yet
- IridiumDocument6 pagesIridiumCh V S RajuNo ratings yet
- Copper Design ManualDocument22 pagesCopper Design Manualamr_scorpion_engNo ratings yet
- METALSDocument62 pagesMETALSEstefany MarilagNo ratings yet
- History of MetalDocument5 pagesHistory of Metalrbnlth.joNo ratings yet
- Mars and IronDocument3 pagesMars and IronRimuel92No ratings yet
- Prospecting and Mining Basics of Gold and Base MetalsDocument93 pagesProspecting and Mining Basics of Gold and Base MetalsKaharuddin HawingNo ratings yet
- How rocks and minerals affect our daily lives in surprising waysDocument8 pagesHow rocks and minerals affect our daily lives in surprising waysallogyNo ratings yet
- Ele City: (Investigatory Project)Document8 pagesEle City: (Investigatory Project)Liyana ChuaNo ratings yet
- Earth Science Quarter 1 - Module 2Document4 pagesEarth Science Quarter 1 - Module 2Shelly Ryn SaligumbaNo ratings yet
- Chemistry Midterm EssayDocument5 pagesChemistry Midterm EssayIrakli NatroshviliNo ratings yet
- Mining Obscure MetalsDocument3 pagesMining Obscure MetalsRXillusionistNo ratings yet
- Earth's Natural Wealth Running Out in 15 YearsDocument6 pagesEarth's Natural Wealth Running Out in 15 YearsBrando MezNo ratings yet
- Electrical CordsDocument34 pagesElectrical CordsShiela Mae BigataNo ratings yet
- Hydrate Lab: Never Place A Hot Crucible On A Balance! When Cool, Determine The Mass of The Crucible andDocument1 pageHydrate Lab: Never Place A Hot Crucible On A Balance! When Cool, Determine The Mass of The Crucible andmamazookeeprNo ratings yet
- Formula Sheet 1Document1 pageFormula Sheet 1mamazookeeprNo ratings yet
- Chemical Formula QuizDocument3 pagesChemical Formula QuizmamazookeeprNo ratings yet
- Elements of Modern Style ArticleDocument5 pagesElements of Modern Style ArticlemamazookeeprNo ratings yet
- Practice Symbol Quiz AnswersDocument1 pagePractice Symbol Quiz AnswersmamazookeeprNo ratings yet
- Formula Sheet 2Document1 pageFormula Sheet 2mamazookeeprNo ratings yet
- Trends ReviewDocument2 pagesTrends ReviewmamazookeeprNo ratings yet
- Build A Trend LabDocument2 pagesBuild A Trend LabmamazookeeprNo ratings yet
- Unit 4 Test 2009Document2 pagesUnit 4 Test 2009mamazookeeprNo ratings yet
- Shorthand ConfigurationsDocument1 pageShorthand ConfigurationsmamazookeeprNo ratings yet
- Elements of Modern Style QuestionsDocument2 pagesElements of Modern Style QuestionsmamazookeeprNo ratings yet
- Unit 4 ReviewDocument2 pagesUnit 4 ReviewmamazookeeprNo ratings yet
- Lab - Which Side Are You OnDocument3 pagesLab - Which Side Are You OnmamazookeeprNo ratings yet
- Practice Symbol QuizDocument1 pagePractice Symbol QuizmamazookeeprNo ratings yet
- Periodic+Table XDocument38 pagesPeriodic+Table XmamazookeeprNo ratings yet
- Unit 3 Practice TestDocument3 pagesUnit 3 Practice TestmamazookeeprNo ratings yet
- Atoms and The PTDocument16 pagesAtoms and The PTmamazookeeprNo ratings yet
- Electron Configuration SheetDocument2 pagesElectron Configuration SheetmamazookeeprNo ratings yet
- Unit 2 Re TestDocument2 pagesUnit 2 Re TestmamazookeeprNo ratings yet
- Answers To Example Problems From Class Regular ConfigurationsDocument1 pageAnswers To Example Problems From Class Regular ConfigurationsmamazookeeprNo ratings yet
- Rainbow of ColorsDocument2 pagesRainbow of ColorsmamazookeeprNo ratings yet
- Lab Report Format: Title Source Name Partner Date PurposeDocument2 pagesLab Report Format: Title Source Name Partner Date PurposemamazookeeprNo ratings yet
- Subatomic Particle WorksheetDocument1 pageSubatomic Particle WorksheetmamazookeeprNo ratings yet
- Light Equation SheetDocument1 pageLight Equation SheetmamazookeeprNo ratings yet
- Mole SheetDocument1 pageMole SheetmamazookeeprNo ratings yet
- Average Weighted Atomic MassDocument2 pagesAverage Weighted Atomic MassmamazookeeprNo ratings yet
- Atomic History Old XDocument32 pagesAtomic History Old XmamazookeeprNo ratings yet
- Sample Lab ReportDocument3 pagesSample Lab Reportmamazookeepr63% (8)
- Unit Analysis ProblemsDocument1 pageUnit Analysis ProblemsmamazookeeprNo ratings yet
- Popcorn LabDocument1 pagePopcorn LabmamazookeeprNo ratings yet
- Lab Report 5Document9 pagesLab Report 5Leo Marcelo Villalba100% (1)
- Temp CycloneDocument19 pagesTemp CycloneDarshan KanganeNo ratings yet
- Water in MalaysisDocument44 pagesWater in MalaysisAlvaro HueteNo ratings yet
- Electrical Standards and ConventionsDocument23 pagesElectrical Standards and ConventionsAngelika ParedesNo ratings yet
- CV Lineas de TransmisiónDocument12 pagesCV Lineas de TransmisiónJoshueNo ratings yet
- Parcel 26 Zone (C)Document17 pagesParcel 26 Zone (C)Dessouki SamakNo ratings yet
- Daftar PustakaDocument7 pagesDaftar PustakaUlul AlbabNo ratings yet
- Appendix: 12.1 Inventory of DistributionsDocument6 pagesAppendix: 12.1 Inventory of DistributionsAhmed Kadem ArabNo ratings yet
- Trenching Soil TypesDocument1 pageTrenching Soil TypesJerome MansuetoNo ratings yet
- Turbo Generator Project ReportDocument40 pagesTurbo Generator Project Reportthegopal100% (2)
- Chapter 14mDocument24 pagesChapter 14mamirNo ratings yet
- Balance Between Optical and Bulk Fill Features: Angie Segatto Direct Posteriors - Products - CasesDocument17 pagesBalance Between Optical and Bulk Fill Features: Angie Segatto Direct Posteriors - Products - Casesmaroun ghalebNo ratings yet
- Light and Optics Chapter SummaryDocument83 pagesLight and Optics Chapter SummaryWhye Jun CheongNo ratings yet
- UT Book ArticlesDocument135 pagesUT Book ArticlesAnonymous uXdS9Y7100% (1)
- CapsuleDocument7 pagesCapsuleNouman Khan KhanNo ratings yet
- TeslaDocument9 pagesTeslaTirchila Traian100% (1)
- Everexceed Deep Cycle Gel RangeDocument0 pagesEverexceed Deep Cycle Gel RangeMikhail solizNo ratings yet
- Aerodyn Aeronaut and Fligh MechDocument664 pagesAerodyn Aeronaut and Fligh MechAerospaceAngel100% (5)
- CEMKS3 E7 GM4 1 ItDocument3 pagesCEMKS3 E7 GM4 1 ItHan Yuan YapNo ratings yet
- CU-2022 B.sc. (Honours) Mathematics Semester-1 Paper-CC-1 QPDocument4 pagesCU-2022 B.sc. (Honours) Mathematics Semester-1 Paper-CC-1 QPMy MathNo ratings yet
- 31-XX29-6Document1 page31-XX29-6rohitkush100% (1)
- 2008-Teacher 20080324 1509 2Document20 pages2008-Teacher 20080324 1509 2Mateo PremarionNo ratings yet
- Divine Creation - BigbangDocument3 pagesDivine Creation - BigbangCess AlfonsoNo ratings yet
- TG40E80 PDF, TG40E80 Description, TG40E80 Datasheets, TG40E80 View - ALLDATASHEETDocument2 pagesTG40E80 PDF, TG40E80 Description, TG40E80 Datasheets, TG40E80 View - ALLDATASHEETInas Ratnaning ZafirahNo ratings yet
- Practicaldistillationcontrol 140307141500 Phpapp01Document549 pagesPracticaldistillationcontrol 140307141500 Phpapp01syedmuhammadtariqueNo ratings yet
- Intergreen TimeDocument2 pagesIntergreen TimeMd Didarul AlamNo ratings yet
- Pumps and Systems MagazineDocument68 pagesPumps and Systems MagazineMohamed Abdel BasitNo ratings yet
- Computer SylDocument87 pagesComputer Sylambuj2011No ratings yet
- HYSYS Ver.8 Tutorial MaterialDocument27 pagesHYSYS Ver.8 Tutorial Materialyoonchankim091150% (2)
- 729797Document216 pages729797aomareltayebNo ratings yet