Professional Documents
Culture Documents
Journal
Uploaded by
M.kishore KumarOriginal Description:
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Journal
Uploaded by
M.kishore KumarCopyright:
Available Formats
Journal of Ethnopharmacology 128 (2010) 1519
Contents lists available at ScienceDirect
Journal of Ethnopharmacology
j our nal homepage: www. el sevi er . com/ l ocat e/ j et hphar m
Screening of analgesic and anti-inammatory activities of Citrullus colocynthis
from southern Tunisia
Belsem Marzouk
a
, Zohra Marzouk
b
, Ehsen Haloui
b
, Nadia Fenina
b
,
Abderrahman Bouraoui
c
, Mahjoub Aouni
a,
a
Laboratoire des Maladies Transmissibles et Substances Biologiquement Actives, Facult de Pharmacie, Monastir, Rue AVICENNE, 5000 Monastir, Tunisia
b
Unit de Pharmaco-conomie et Dveloppement des Mdicaments, Laboratoires de Biologie Vgtale et Laboratoire de Pharmacologie, Facult de Pharmacie, Monastir, Tunisia
c
Unit URSAM, Laboratoire de Pharmacologie, Facult de Pharmacie, 5000 Monastir, Tunisia
a r t i c l e i n f o
Article history:
Received 24 June 2009
Received in revised form 3 November 2009
Accepted 26 November 2009
Available online 3 December 2009
Keywords:
Citrullus colocynthis Schrad.
Aqueous extract
Acute toxicity
Analgesic activity
Anti-inammatory activity
Tunisian medicinal plant
a b s t r a c t
Ethnopharmacological relevance: Inammations and immune-related diseases such as rheumatoid arthri-
tis are growing global concerns. Most of the drugs from plants which have become important in modern
medicine had a folklore origin and are traditional in systems of medicine. Citrullus colocynthis Schrad.
(cucurbitaceae), endemic in Southern Tunisia, is used in folk medicine to treat many inammation
diseases.
Aim of study: To evaluate the acute toxicity of different parts of Citrullus colocynthis and then to screen
the analgesic and anti-inammatory activities of aqueous extracts fromroots and stems of the plant and
from fruits and seeds at different maturation stages.
Materials and methods: After identication and acute toxicity assay Citrullus colocynthis Schrad. aqueous
extracts were screened for analgesic and anti-inammatory activities using, respectively, the acetic acid
writhing test in mice and the carrageenan-induced paw edema assay in rats.
Results: All extracts displayedanalgesic andanti-inammatoryactivities at different doses without induc-
ing acute toxicity. Topic results were obtained with immature fruits followed by seeds. The stem and
root extracts were shown to possess the less signicant inhibitory activity against analgesic and anti-
inammatory models.
Conclusions: Based on this study, we conrmed that Citrullus colocynthis Schrad. is a potentially useful
drug suitable for further evaluation for rheumatoid arthritis, and its folk medicinal use as an analgesic
and anti-inammatory agents is validated.
2009 Elsevier Ireland Ltd. All rights reserved.
1. Introduction
For the most part, modern science has neglected botanical
source of knowledge and the pharmacological potential of old
remedies have not been systematically evaluated. Nevertheless
many species used in traditional medicine have the potential
to provide pharmacologically active natural products. Citrullus
colocynthis Schrad. is a wild native plant growing in arid areas
(Pottier-Alapetite, 1981). This cucurbitaceae is widely used in
Tunisian folk medicine and it possesses therapeutic activities
against a wide range of ailments including inammatory disorders,
arthritis and gout (Le Flock, 1983; Boukef, 1986; Marzouk et al.,
2009). Nevertheless, a human overdose of plant immature fruits is
hazard. Intoxication is manifested by colitis, gastro-intestinal irri-
Corresponding author. Tel.: +216 73 461 000; fax: +216 73 461 830.
E-mail address: mahjoub.ouni@fphm.rnu.tn (M. Aouni).
tations, delirium, hypothermia and cerebral congestions (Meyer,
1989; Bellakhdhar, 1999).
In Tunisia, as in other Mediterranean countries (Al-Rawi and
Chaakravarty, 1964), the parts of plants most often used for medic-
inal purposes are fruits and/or seeds, though other parts of the
plants can be used, for example roots to treat urinary infection
(Nadkami, 1954) or leaves (Batanouny, 1999). Traditional heal-
ers seem to not pay attention to the plants degree of maturity.
The literature rarely mentions if seeds are present in preparations
involving ground fruit/pulp. Modes of preparation and administra-
tion vary, even for similar indications. Common preparations use
fresh, warmed or dried plant material (often ground), as well as
extracts used mostly in a liquid form. Extracts are prepared either
in water or in aqueous mixtures containing more lipophilic com-
pounds (hot milk extractions, water/olive oil at various ratios) at
a temperature ranged from tepid to boiling. Ground plant material
can be mixed with honey for ingestion or topical gynaecological
application or with other plants for poultices (for example with
0378-8741/$ see front matter 2009 Elsevier Ireland Ltd. All rights reserved.
doi:10.1016/j.jep.2009.11.027
16 B. Marzouk et al. / Journal of Ethnopharmacology 128 (2010) 1519
Lawsonia inermis and Capparis spinosa). Methods of administration
are topical, rectal or vaginal (fruit), enema, cervico-vaginal douche,
andby ingestion(Boukef, 1986; Marzouk et al., 2009). Extreme cau-
tion should be exercised with ingestion, due to the plants drastic
laxative properties, and with contact with leaves, due to risks of
syncope (for all mammals including domestic animals) (Marzouk
et al., 2009). Use is contraindicated during pregnancy as the plant is
abortifacient (Pottier-Alapetite, 1981; Delazar et al., 2006). Many of
todays traditional medicinal uses of the plant are found through-
out history (see the Ebers papyrus in ca. 1550BCE Egypt) (Riddle,
1999) over a large geographical zone fromMauritania toIndia, even
extending outside of the plant endemic zone, to Europe (Adams et
al., 2009).
The present investigation was undertaken to establish the acute
toxicity study and to evaluate the analgesic and anti-inammatory
activities of the reconstituted lyophilized extracts from vegeta-
tive organs (roots, stems and leaves) and two different maturation
states of reproductive organs (fruits and seeds).
2. Materials and methods
2.1. Sampling and identication
Citrullus colocynthis Schrad. plants were collectedinAugust near
Medenine, Tunisia in the municipality of Sidi Makhlouf (33
33N,
10
27W). The identication was performed according to the ora
of Tunisia (Pottier-Alapetite, 1981) and a voucher specimen (C.C-
01.01) deposited in the biological laboratory of the Faculty of
Pharmacy of Monastir.
2.2. Extraction protocol
The extraction was performed on 100g of each fresh organ:
roots, stems, leaves and two different stages of maturation of fruits
and seeds (immature and ripe). Yields of prepared extracts are
gravimetrically determined. For preparing the aqueous extract,
100g of each fresh organ were ground with a mixer and added
to 500ml of distilled water. The mixture was allowed to reux
for 30min, after which the solution was allowed to cool (4h at
4
C). The mixture was then ltered using lter paper (Whatman
no.1) under the vacuumof a water pump. The ltrate obtained was
lyophilized, yielding the lyophilized aqueous extract.
2.3. Animals
Male adult Wistar rats weighing 160180g and Swiss albinos
mice (weighing 1825g) of both sex were obtained from Pas-
teur institute (Tunis, Tunisia). They were housed in polypropylene
cages and were left for 2 days for acclimatization to animal room
maintained under controlled condition (a 12h lightdark cycle at
222
C) on standard pellet diet and water ad libitum. Before the
day of assay, only the Wistar rats were fasted overnight with the
free access to water. Housing conditions and in vivo experiments
approved according to the guidelines established by the European
Union on Animal Care (CFE Council (86/609)). The rats were used
for the anti-inammatory evaluation of the aqueous extracts while
the mice were used for the analgesic investigation and for the acute
toxicity testing.
Animals were divided into drug-treated test and saline-treated
control groups of six or eight animals per group.
2.4. Acute toxicity
For acute toxicity, mice were divided into groups of eight ani-
mals each. One group served as a control and received 0.9% NaCl
alone (10ml/kg) given intraperitoneally (i.p.), while the remaining
groups were treated with increasing doses of the aqueous extract:
50,100, 250, 500, 750, 1000, 1500, 2000, 3000and4000mg/kg (i.p.),
respectively.
The mortality rate within a 48h period was determined and the
LD
50
was estimated according to the method described by Miller
and Tainter (1944).
According to the results of acute toxicity test, doses were chosen
for pharmacological evaluations. After the last observationthe mice
were killed and the liver, lungs, heart, spleen and kidneys were
withdrawn, weighed and stoked for next evaluations.
2.5. Analgesic activity
Analgesic activity was performed according to the method of
Koster et al. (1959) and assessed by the acetic acid abdominal
constriction test (writhing test)a chemical visceral pain model.
Swiss albinos mice were selected 1 day prior to each test and
were divided into groups of six mice each. One group served as
the control and was pretreated under cutaneously with 10ml/kg
of saline. Another group was pretreated with the reference drug,
acetyl salicylate of lysine (ASL), 200mg/kg, by the same route.
The remaining groups were injected intraperitoneally (i.p.) with
10ml/kg of 1% acetic acid solution 30min after the administration
of different extracts at the doses of 2, 4 and 8mg/kg except imma-
turefruits towhichwedecreasedoses to0.1mg/kg. After acetic acid
administration, the number of writhes was counted during 30min.
Antinociceptive activity was expressed as inhibition percent of
the usual number of writhes observed in control animals. The
percentages of inhibition were calculated according to the follow-
ing formula: % inhibition=((number of writhes)
control
(number of
writhes)
treated group
) 100/(number of writhes)
control
.
2.6. Anti-inammatory activity
The anti-inammatory activity was assessed on the basis of
inhibition of paw edema induced by the injection of carrageenan
(an edematogenic agent) into the subplantar region of the right
hind paw of the rat (Winter et al., 1962). Male Wistar rats were
divided into different groups of 8 animals. The control group
received 2.5ml/kg of saline, the standard group received the ref-
erence drug (acetyl salicylate of lysine (ASL), 300mg/kg) and the
test groups received different organ extracts of Citrullus colocynthis
at a dose of 1 and 4mg/kg. Thirteen minutes after intraperitoneal
administrationof different substances, 0.05ml of 1%of carrageenan
suspension was injected to all animals in the right hind paw.
The paw volume, up to tibiotarsal articulation, was measured
using a plethysmometer. The measures were determinedat 0h(V
0
:
before edematogenic agent injection) and 1, 2, 3, 4, 5, 6 and 24h
intervals later (V
T
). The difference between V
T
(1, 2, 3, 4, 5, 6 and
24h) and V
0
was taken as the edema value. The percentages of inhi-
bitionwere calculatedaccording to the following formula: %inhibi-
tion=((V
T
V
0
)
control
(V
T
V
0
)
treated group
) 100/(V
T
V
0
)
control
.
2.7. Statistical analysis
Data obtained from animal experiments were expressed as
meanS.E.M. and as percentage. Results were statistically evalu-
ated by ANOVAand using Students t-test. p0.05 were considered
signicant.
3. Results
3.1. Toxicity studies
Swiss albinos mice were observed during 48h and morbid-
ity and/or mortality were recorded, if happens, for each group at
B. Marzouk et al. / Journal of Ethnopharmacology 128 (2010) 1519 17
Table 1
Extraction yields (w/w, %) and LD
50
of Citrullus colocynthis Schrad. organ aqueous extracts.
Immature seeds Ripe seeds Immature fruits Ripe fruits Leaves Stems Roots
Extraction yields 2.94 2.21 2.76 3.04 12.89 11.05 4.56
LD
50
(mg/kg) 744.1 884.2 553.7 487.6 95.8 3903.2 833.3
the end of observation period. Due to death index, the LD
50
of
all extracts were determined (Table 1). This value is in relation
with organs but also with the maturation states. The LD
50
were
ranged from 95.8 to 3903.2mg/kg. Stems were evaluated as the
less toxic part of Citrullus colocynthis (LD
50
=3903.2mg/kg). For the
fruit extracts, immature fruits hadthe lowest LD
50
. Immature seeds
were as toxic as mature ones. Leaves seem to be the most toxic
organ with a LD
50
equal to 95.8mg/kg; all the animals treated with
this extract presented feebleness, hypothermia and insufciency
respiratory. Looking to the higher toxicity of this vegetative organ
(leaves), it was discardedfromthe analgesic andanti-inammatory
investigations.
3.2. Analgesic activity
The inhibitionpercentages of writhing for all extracts are shown
in Table 2. The reference drug inhibited 61.88% of the number of
writhing elicited by acetic acid. The analgesic effect was tested
for concentrations ranging from 2 to 8mg/kg, except immature
fruits; to which we decrease the concentration to 0.1mg/kg. The
administration of all tested extracts induced a dose-dependent
antinociceptive activity. The immature fruits and seeds possess the
highest analgesic properties; the most active of them were imma-
ture fruits as well as at 0.1mg/kg (93.52%). Stem extracts were as
active as those of roots. The lowest activity was observed for ripe
fruits.
Table 2
Analgesic activity of different Citrullus colocynthis Schrad. aqueous extracts and ref-
erence drug (ASL).
Extract Concentration
(mg/kg)
Number of writhes Inhibition of
writhing (%)
Control 74.33 4.64
Immature seeds 2 2.67 0.51
***
96.41
4 2.00 0.63
***
97.31
8 0.50 0.54
***
99.33
Ripe seeds 2 4.50 0.54
***
93.95
4 2.67 0.51
***
96.41
8 1.00 0.63
***
98.65
Immature fruits 0.1 3.33 0.51
***
93.52
0.5 2.67 0.81
***
96.41
2 1.67 0.51
***
97.76
4 1.33 0.51
***
98.21
8 0.33 0.51
***
99.55
Ripe fruits 2 66.50 2.25
***
10.59
4 48.83 1.94
***
30.42
8 33.50 1.37
***
54.93
Stems 2 55.83 1.47
***
55.53
4 21.17 1.16
***
71.52
8 8.83 0.75
***
88.12
Roots 2 7.33 0.51
***
90.13
4 5.17 0.75
***
93.05
8 3.67 0.51
***
95.07
Reference drug (ASL) 200 28.33 2.06
***
61.88
Values are expressed as meanS.E.M. (N=6); ASL: acetyl salicylate of lysine.
***
p0.001 signicant from control.
3.3. Anti-inammatory effect
In carrageenan-induced rat paw edema, all extracts produced
a reduction of the edema throughout the entire period of obser-
vation. The intraperitoneal administration of the aqueous extracts
of reproductive organs (fruits and seeds) reduced signicantly
the paw edema induced by the noxious agents. This inhibition is
less remarkable after the two vegetative organ (roots and stems)
aqueous extract injections. For this preliminary assessment of
the anti-inammatory activity, the obtained results demonstrate
that the reduction of the paw edema vary in a dose-dependent
fashion with a maximum attend at 4mg/kg. On the contrary,
at 1mg/kg, only a moderate (vegetative organs) and appreciably
(reproductive organs) activities were noted. With all extracts, 3h
after carrageenan injection, the anti-inammatory activity insti-
gate unambiguous increasingly to attend the maximum at 6 and
24h, after a little decrease at 5h. In terms of plant organs, imma-
ture organs (fruits andseeds) showedbetter activity thanripe ones.
The inhibition percentages were ranged, respectively, from 90.00%
to 98.84% and from 88.33% to 98.06% for immature organs, and
ranged from 78.33% to 97.69% and from 65.00% to 97.90%, respec-
tively, for ripe fruits and seeds. The highest activity was found for
immature fruits. Stem aqueous extracts were either as strongly
anti-inammatory as root aqueous extract (16h) or a close second
after 24h fromthe carrageenan injection. Standard drug decreased
paw edema by a maximum of 74.40% after 4h (Table 3).
4. Discussion
This is the rst study evaluating the in vivo acute toxicity, the
antinociceptive and the anti-inammatory activities of extracts
from different Citrullus colocynthis organs.
Based on the LD
50
calculated, the acute administration doses
of all organ aqueous extracts are estimated (1 and 4mg/kg). These
doses whichareveryslighter thantheLD
50
weredemonstratedef-
cient. The leaf toxicity and its symptoms are conrmed by peoples
from Sidi Makhlouf municipality since they suffer from feebleness
and insufciency respiratory every time they were considerably
projected to this organ. This expressed toxicity was absolutely not
conformed to the traditional use of this plant organ (Batanouny,
1999). This contradiction may be attributed to the used doses and
preparations as folkloric medicine. Bruneton (1999) showed that
the leaf toxicity appear on sheep at a dose of 250mg/kg (>obtained
LD
50
=95.8mg/kg). So plant toxicity is in relation with many condi-
tions likethegeographical distributionandthecollect seasonwhich
considerablyinuencedthe plant composition, andthenits toxicity
and its pharmacological activity. In addition, it is known that the
metabolism and the pharmacological activities are very different
according to the animal models (Bertrand, 1976; Rico, 1978).
In acetic acid-induced writhing in mice, all tested samples
extracts reduced signicantly the number of writhing which are
associated with the release of endogenous substances including
serotonin, histamine, prostaglandin and bradykinin (Collier et al.,
1968). The results obtained in this test thus suggest that while
Citrullus colocynthis Schrad. organs possess peripheral analgesic
properties, this particular activity is probably linked to their anti-
inammatory effects.
Carrageenan has been widely used as a noxious agent able to
induce experimental inammation for the screening of compounds
possessing anti-inammatory activity. This phlogistic agent, when
1
8
B
.
M
a
r
z
o
u
k
e
t
a
l
.
/
J
o
u
r
n
a
l
o
f
E
t
h
n
o
p
h
a
r
m
a
c
o
l
o
g
y
1
2
8
(
2
0
1
0
)
1
5
1
9
Table 3
Effects of different Citrullus colocynthis Schrad. organ aqueous extracts and reference drug on carrageenan-induced paw edema.
Extract Dose (mg/kg) Mean swelling thickness (10
2
) S.E.M. (% inhibition)
1h 2h 3h 4h 5h 6h 24h
Control 1 15.00 1.76 24.00 2.66 49.00 4.77 57.50 5.56 59.50 5.25 64.50 8.20 32.50 3.51
Immature seeds 1 14.50 2.89
ns
(3.33) 19.25 2.21
**
(19.79) 33.25 2.21
***
(32.14) 32.50 2.38
***
(43.48) 39.00 3.91
***
(34.45) 29.25 3.86
***
(54.65) 14.25 0.96
***
(56.15)
4 1.75 0.96
***
(88.33) 2.00 0.82
***
(91.66) 3.50 0.58
***
(92.86) 1.50 1.29
***
(97.40) 2.50 1.29
***
(95.80) 1.25 0.50
***
(98.06) 0.75 0.95
***
(97.69)
Ripe seeds 1 15.00 0.82
ns
(0.00) 23.75 1.25
ns
(1.04) 42.00 6.16
*
(14.28) 37.50 3.87
***
(34.78) 42.25 2.22
***
(28.99) 29.50 3.41
***
(54.26) 14.50 1.29
***
(55.38)
4 5.25 1.26
***
(65.00) 2.25 0.50
***
(90.62) 1.75 0.96
***
(96.43) 1.25 0.50
***
(97.83) 1.50 0.58
***
(97.48) 1.50 0.58
***
(97.67) 1.25 0.50
***
(96.15)
Immature fruits 1 13.25 2.22
ns
(11.66) 16.75 3.30
**
(30.21) 29.00 2.16
***
(40.82) 31.75 3.86
***
(44.78) 34.50 1.29
***
(43.70) 21.75 3.95
***
(66.02) 16.75 2.22
***
(48.46)
4 1.50 0.58
***
(90.00) 1.75 0.96
***
(92.71) 2.75 0.50
***
(94.39) 1.75 0.95
***
(97.65) 2.75 0.96
***
(95.38) 0.75 0.96
***
(98.84) 0.50 0.58
***
(98.46)
Ripe fruits 1 14.00 1.41
ns
(6.66) 21.75 1.50
*
(9.37) 42.75 2.63
**
(12.75) 43.75 3.09
***
(23.91) 50.25 1.70
***
(15.55) 43.00 3.83
***
(33.33) 17.75 2.05
*
(45.38)
4 3.25 0.96
***
(78.33) 2.50 0.57
***
(89.58) 3.5 0.57
***
(92.86) 4.25 2.63
***
(92.61) 4.75 0.50
***
(92.02) 1.75 0.50
***
(97.29) 0.75 0.50
***
(97.69)
Control 2 15.00 1.76 34.50 4.33 57.00 6.22 61.50 6.42 67.00 7.37 71.00 6.39 40.50 5.84
Stems 1 14.00 1.41
ns
(6.66) 31.75 1.25
*
(7.97) 44.25 2.75
***
(22.37) 46.75 3.09
***
(23.98) 53.75 2.99
***
(19.77) 47.00 1.82
***
(33.80) 26.75 1.5
***
(33.95)
4 13.75 2.21
ns
(8.33) 30.50 1.91
*
(11.59) 33.25 2.98
***
(41.66) 27.00 3.37
***
(56.10) 36.00 2.16
***
(46.27) 32.00 2.16
***
(54.93) 13.00 1.15
***
(67.90)
Roots 1 15.00 2.45
ns
(0) 34.25 1.70
ns
(0.72) 55.00 3.74
ns
(3.51) 51.50 1.29
***
(16.26) 59.50 1.29
***
(11.19) 49.50 1.91
***
(25.00) 28.00 0.82
***
(30.86)
4 13.75 2.22
ns
(8.33) 31.50 2.38
ns
(8.69) 46.50 2.08
***
(18.42) 47.00 3.16
***
(23.58) 53.00 1.82
***
(20.89) 53.25 3.20
***
(30.28) 18.25 1.71
***
(54.94)
ASL 300 7.25 0.96
***
(51.66) 14.25 1.71
***
(58.69) 17.50 2.38
***
(69.30) 15.75 2.22
***
(74.40) 19.50 2.08
***
(70.89) 19.50 1.29
***
(72.53) 23.75 1.71
***
(41.36)
Values are expressed as meanS.E.M. (N=8); ns: not signicant from the control; ASL: acetyl salicylate of lysine.
*
p0.05 signicant from the control.
**
p0.01 signicant from the control.
***
p0.001 signicant from the control.
B. Marzouk et al. / Journal of Ethnopharmacology 128 (2010) 1519 19
injected locally into the rat paw, produced a severe inammatory
reaction, which was discernible within 30min (John and Nodine,
1999). The development of edema induced by carrageenan is a
biphasic event: the early phase (90180min) of the inammation
is due to the release of histamine, serotonin and similar substances.
The later phase (270360min) is associated with the activation of
kinin-like substances and the release of prostaglandins, proteases
and lysosome (Olajide et al., 1999). All organ extracts inhibited
hind pawedema and showed a dose-depending anti-inammatory
activity but the results were different for each organ depend-
ing on the early/later phases. Extracts from reproductive organs
(seeds and fruits: immature and ripe ones) inhibited the both
phases of the carrageenan-induced edema by reducing the release
of histamine and serotonin and also the kinin-like substances and
prostaglandins; while the extracts from vegetative organs (roots
and stems) inhibited particularly the later phases (424h) by
restraining the kinin-like substances and prostaglandins produc-
tions. This pharmacological property may be attributed to the plant
organcompositionandtoapossiblemolecular mechanismbyeffec-
tivelydecreasingthe productionof the pro-inammatorycytokines
of IL-6 and IL-1 and the expression of COX-2 and simultane-
ously elevating the level of anti-inammatory cytokine IL-4 in the
carrageenan-injected rat pawtissues (Moulin and Coquerel, 2002).
At any rate, these results indicate that the analgesic and anti-
inammatory activities could not be imputed to one family of
phytochemicals only (or its absence). Like for the antibacterial
and the anti-candidal activities, analgesic and anti-inammatory
ones may be attributed, possibly in combination, to various phy-
tochemicals detected (alkaloids, iridoids, avonoids, steroids, etc.)
(Marzouk et al., 2009). These compounds might be present in vari-
ous concentrations according to the maturation stage of seeds and
fruits (Marzouk et al., 2009). Alkaloids are commonly found to have
analgesic and anti-inammatory properties (Moulin and Coquerel,
2002); therefore their absence in roots could account for the lack of
activityof this organ. Iridoids, witchhave anti-inammatoryeffects
(Mesia-Vela et al., 2004), are detected in the aqueous root extract
but this extract showed no overall activity. However, alkaloids and
iridoids cannot be solely responsible for the activity. Flavonoids
(detected in all the seed extracts) are known to have analgesic and
anti-inammatory properties (Borgi et al., 2008); the same could
be said about steroids witch contribute to the better performance
as an anti-inammatory agent (Bames and Adcock, 2009).
With these analgesic and anti-inammatory properties, Citrul-
lus colocynthis Schrad. can be considered an effective agent to
treat inammation diseases. This plant, namely its seed and fruit
extracts, demonstrated a high activity at very low aqueous extract
doses (1 and 4mg/kg). The study corroborated the analgesic effects
of this specie, justied and supported scientically its ethnophar-
macological use as an anti-inammatory agent to treat pain and
rheumatoid arthritis. Therefore it could account for some of the
variations observedinthe Ethnopharmaceutical preparationmeth-
ods. From now the use of this plant is validated by the results
obtainedinthis work. Additional studies areongoingtoconrmthis
Citrullus colocynthis Schrad. properties with some ecological vari-
abilities. Further attempts to isolate and dene the active analgesic
and anti-inammatory fractions and its components.
Acknowledgements
Thanks to Dr. Zohra MARZOUK for the specimen identica-
tion and to Dr. Wahida BORGI and Dr. Rachel DECOR for their
help.
References
Adams, M., Berset, C., Kessler, M., Hamburger, M., 2009. Medicinal herbs for the
treatment of rheumatic disordersa survey of European herbals from the 16th
and 17th century. Journal of Ethnopharmacology 121, 343359.
Al-Rawi, A., Chaakravarty, H.L., 1964. Medicinal Plants of Iraq. Ministryof Agriculture
and Technology, Bulletin no.146. Baghdad, Iraq.
Bames, P.J., Adcock, I.M., 2009. Glucocorticoid resistance in inammatory diseases.
The Lancet 373, 19051917.
Batanouny, K.H., 1999. The mediterranean coast dunes in Egypt: an endangered
lamdscape. Estuarine, Coastal and Shelf Science 49, 39.
Bellakhdhar, J., 1999. La Pharmacope Marocaine Traditionnelle. IBIS Press, pp:
365387.
Bertrand, M., 1976. Les modles animaux en pharmacologie et en toxicologie. Sci-
ence et Techniques des Animaux de Laboratoire 1, 199214.
Borgi, W., Recio, M.-C., Ros, J.L., Chouchane, N., 2008. Anti-inammatory and anal-
gesic activities of avonoid and saponin fractions from Zizyphus lotus (L.) Lam.
South African Journal of Botany 74, 320324.
Boukef, M.K., 1986. Mdecine traditionnelle et pharmacope. Les plantes dans la
mdecine traditionnelle tunisienne. Agence de Coopration Culturelle et Tech-
nique, Paris, p. 165.
Bruneton, J., 1999. Plantes Toxiques, Vgtaux Dangereux pour lHomme et les Ani-
maux. Technique & Documentation, Paris, p. 236.
Collier, H.O., Dinneen, L.C., Johnson, C.A., Schneider, C., 1968. The abdominal con-
struction response and its suppression by analgesic drugs in the mouse. British
Journal of Pharmacology 32, 295310.
Delazar, A., Gibbons, S., Kosari, A.R., Nazemyeh, H., Modarresi, M., Nahar, L., Sarker,
S.D., 2006. Flavone C-glycosides and cucurbitacin glycosides fromCitrullus colo-
cynthis. Journal of Faculty of Pharmacy, Tehran University of Medicinal Sciences
14, 109114.
John, H., Nodine, M.D., 1999. Chicago: Year Book Medical. Publishers Inc, p. 492.
Koster, R., Anderson, M., De Beer, E.J., 1959. Acetic acid for analgesic screening.
Federation Proceedings 18, 418420.
Le Flock, E., 1983. Contribution une tude Ethnobotanique de la ore tunisi-
enne. Imprimerie ofcielle de la rpublique tunisienne, Tunis, Tunisia, pp. 241
244.
Marzouk, B., Marzouk, Z., Dcor, R., Edziri, H., Haloui, E., Fenina, N., Aouni, M., 2009.
Antibacterial and anticandidal screening of TunisianCitrullus colocynthis Schrad.
from Medenine. Journal of Ethnopharmacology 125, 344349.
Mesia-Vela, S., Souccar, C., Lima-Landman, M.T.R., Lapa, A.J., 2004. Pharmacolog-
ical study of Stachytarpheta cayennensis Vahl in rodents. Phytomedicine 11,
616624.
Meyer, F., 1989. Toxicit des baies et des fruits bacciformes. Quize ans dexprience
du centre anti-poisons de Lyon. Thse de Doctorat en medecine. Univercit
Claude Bernart, Lyon, pp: 1530.
Miller, L.C., Tainter, M.L., 1944. Estimation of the ED
50
and its error by means of log-
probit graphic paper. Proceeding of the Society for Experimental Biology and
Medicine 57, 261264.
Moulin, M., Coquerel, A., 2002. Pharmacologie, connaissance et pratique, 2me di-
tion. Masson, Paris, 845 p.
Nadkami, A.K., 1954. Indian Materia Medica, 3rd ed. Popular Prakashan, Bombay,
India.
Olajide, O.A., Makinde, M.J., Awe, S.O., 1999. Effects of the aqueous extract of Bridelia
ferruginea stem bark on carrageenan-induced oedema and granuloma tissue
formation in rats and mice. Journal of Ethnopharmacology 66, 113117.
Pottier-Alapetite, G., 1981. Flore De La Tunisie, Angiospermes-Dicotyldones:
Gamoptales. Ed. Imprimerie ofcielle de la rpublique tunisienne, Tunis,
Tunisia, p. 930.
Rico, A.G., 1978. Modle animal et activit des mdicaments. Annals of Clinical
Biology 36, 149334.
Riddle, J.M., 1999. Eves Herbs: A History of Contraception and Abortion in the West.
Harvard University Press, Harvard, USA, p. 350.
Winter, C.A., Risley, E.A., Nuss, G.W., 1962. Carrageenan induced edema hind pawof
the rat as an essay for anti-inammatory drugs. Proceedings of the Society for
Experimental Biology and Medicine 3, 544547.
You might also like
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeFrom EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeRating: 4 out of 5 stars4/5 (5794)
- The Little Book of Hygge: Danish Secrets to Happy LivingFrom EverandThe Little Book of Hygge: Danish Secrets to Happy LivingRating: 3.5 out of 5 stars3.5/5 (399)
- Unit 1Document14 pagesUnit 1M.kishore KumarNo ratings yet
- Engg Drawing Mid I Exam Question Paper 2014Document2 pagesEngg Drawing Mid I Exam Question Paper 2014M.kishore KumarNo ratings yet
- Venturimeter & OrificemeterDocument8 pagesVenturimeter & OrificemeterRajib DasNo ratings yet
- Potential Flow TheoryDocument13 pagesPotential Flow TheoryM.kishore KumarNo ratings yet
- Leaders Beyond Media Images (Politcal Profiles of Indira Ga Narasimha Rao, N.T.R., YSR, Chandrababu Naidu) - K. BalagopalDocument126 pagesLeaders Beyond Media Images (Politcal Profiles of Indira Ga Narasimha Rao, N.T.R., YSR, Chandrababu Naidu) - K. BalagopalM.kishore KumarNo ratings yet
- KeysDocument6 pagesKeysgolden_bhagatNo ratings yet
- JNTU Hyderabad B.Tech ME Course StructureDocument89 pagesJNTU Hyderabad B.Tech ME Course StructureM.kishore KumarNo ratings yet
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryFrom EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryRating: 3.5 out of 5 stars3.5/5 (231)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceFrom EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceRating: 4 out of 5 stars4/5 (894)
- The Yellow House: A Memoir (2019 National Book Award Winner)From EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Rating: 4 out of 5 stars4/5 (98)
- Shoe Dog: A Memoir by the Creator of NikeFrom EverandShoe Dog: A Memoir by the Creator of NikeRating: 4.5 out of 5 stars4.5/5 (537)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureFrom EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureRating: 4.5 out of 5 stars4.5/5 (474)
- Never Split the Difference: Negotiating As If Your Life Depended On ItFrom EverandNever Split the Difference: Negotiating As If Your Life Depended On ItRating: 4.5 out of 5 stars4.5/5 (838)
- Grit: The Power of Passion and PerseveranceFrom EverandGrit: The Power of Passion and PerseveranceRating: 4 out of 5 stars4/5 (587)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaFrom EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaRating: 4.5 out of 5 stars4.5/5 (265)
- The Emperor of All Maladies: A Biography of CancerFrom EverandThe Emperor of All Maladies: A Biography of CancerRating: 4.5 out of 5 stars4.5/5 (271)
- On Fire: The (Burning) Case for a Green New DealFrom EverandOn Fire: The (Burning) Case for a Green New DealRating: 4 out of 5 stars4/5 (73)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersFrom EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersRating: 4.5 out of 5 stars4.5/5 (344)
- Team of Rivals: The Political Genius of Abraham LincolnFrom EverandTeam of Rivals: The Political Genius of Abraham LincolnRating: 4.5 out of 5 stars4.5/5 (234)
- The Unwinding: An Inner History of the New AmericaFrom EverandThe Unwinding: An Inner History of the New AmericaRating: 4 out of 5 stars4/5 (45)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyFrom EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyRating: 3.5 out of 5 stars3.5/5 (2219)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreFrom EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreRating: 4 out of 5 stars4/5 (1090)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)From EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Rating: 4.5 out of 5 stars4.5/5 (119)
- Her Body and Other Parties: StoriesFrom EverandHer Body and Other Parties: StoriesRating: 4 out of 5 stars4/5 (821)
- 8 Cell - The Unit of Life-NotesDocument6 pages8 Cell - The Unit of Life-NotesBhavanya RavichandrenNo ratings yet
- Biology: Pearson EdexcelDocument24 pagesBiology: Pearson EdexcelAhmad MohdNo ratings yet
- Molecular Cloning Technical GuideDocument40 pagesMolecular Cloning Technical GuideRNo ratings yet
- Eagles and Eaglets gr2Document2 pagesEagles and Eaglets gr2vovanmtcNo ratings yet
- Morphology of Flowering PlantsDocument13 pagesMorphology of Flowering Plantschiragbhaskar16No ratings yet
- Essay On Physical SelfDocument2 pagesEssay On Physical Selfcmestrada03No ratings yet
- Biomedical Physics, Vol 7 @medphyslibDocument360 pagesBiomedical Physics, Vol 7 @medphyslibStats -MANNo ratings yet
- Organizing A TextDocument3 pagesOrganizing A TextMercedes Jimenez RomanNo ratings yet
- Follicular Dynamics in Bovine and Ovine 1Document19 pagesFollicular Dynamics in Bovine and Ovine 1israr yousafNo ratings yet
- Pain PathwayDocument17 pagesPain PathwaySalsabila Al-BasheerNo ratings yet
- Brigitte King - InformativeDocument2 pagesBrigitte King - Informativeapi-262025468No ratings yet
- A Review of The Environmental Fate, Effects, and Exposures of Bisphenol A PDFDocument25 pagesA Review of The Environmental Fate, Effects, and Exposures of Bisphenol A PDFTiago TorresNo ratings yet
- Natamycin Story - What You Need to KnowDocument13 pagesNatamycin Story - What You Need to KnowCharles MardiniNo ratings yet
- Sample Lab ReportDocument7 pagesSample Lab ReportPutri Syalieyana0% (1)
- The Vulcanization of BrainDocument30 pagesThe Vulcanization of BrainJohnny FragosoNo ratings yet
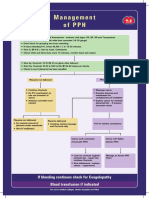
- Management of PPHDocument1 pageManagement of PPH098 U.KARTHIK SARAVANA KANTHNo ratings yet
- Plant Transport - IGCSE Biology Notes (2020)Document6 pagesPlant Transport - IGCSE Biology Notes (2020)Zhi En LeeNo ratings yet
- Effects of Different Fermentation Temperatures On Metabolites of KimchiDocument7 pagesEffects of Different Fermentation Temperatures On Metabolites of KimchiAngela ValdiviesoNo ratings yet
- MDU Open Elective 3rd Sem Date Sheet May 2019Document1 pageMDU Open Elective 3rd Sem Date Sheet May 2019Ratan DeshNo ratings yet
- Introspect For DealersDocument13 pagesIntrospect For DealersOBERON-INTROSPECT-BIOSPECTNo ratings yet
- Microbial Healing of Cracks in ConcreteDocument15 pagesMicrobial Healing of Cracks in ConcreteasdasdasdasNo ratings yet
- Metals: Methods To Evaluate Corrosion in Buried Steel Structures: A ReviewDocument21 pagesMetals: Methods To Evaluate Corrosion in Buried Steel Structures: A ReviewPeymanMajidiNo ratings yet
- Mengenali Konflik Dalam Negosiasi Oleh: Zumaeroh: PendahuluanDocument47 pagesMengenali Konflik Dalam Negosiasi Oleh: Zumaeroh: PendahuluanrahmatNo ratings yet
- Chapter 2 Medical Terminology Verified AnswersDocument5 pagesChapter 2 Medical Terminology Verified AnswersGregg ProducerNo ratings yet
- General Biology 2 Budget of WorkDocument3 pagesGeneral Biology 2 Budget of WorkMaricris BalboaNo ratings yet
- Development of The Planet EarthDocument14 pagesDevelopment of The Planet EarthHana CpnplnNo ratings yet
- MYCOVIRODocument11 pagesMYCOVIROPau SorianoNo ratings yet
- Diencephalon PresentationDocument19 pagesDiencephalon Presentation300rNo ratings yet
- Young Children's Biological Predisposition To Learn in Privileged DomainDocument6 pagesYoung Children's Biological Predisposition To Learn in Privileged DomainVeronica Dadal0% (1)
- TC QMM 56942Document120 pagesTC QMM 56942Fernando R EpilNo ratings yet