Professional Documents
Culture Documents
Clinical Evaluation As Per CE Marking
Uploaded by
SaraOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Clinical Evaluation As Per CE Marking
Uploaded by
SaraCopyright:
Available Formats
CLINICAL EVALUATION
Recent amendments made to the Medical Device Directive (MDD 93/42/EEC) state that every
medical device affixing CE Logo, regardless of its classification, must have a clinical evaluation
report in its Technical File / Design Dossier.
The Directives specific focus on implantable or class III medical devices gives the false
impression that clinical investigation does not apply to other Medical devices. This has led to
the current situation where clinical trials are rarely done in the pre-market steps of medical
devices development.
With the implementation of Directive 2007/47/EC, clinical evaluation has become a central
element of regulatory compliance under the Medical Device Directive and Active Implantable
Medical Device Directive.
Manufacturers of innovative technologies face the greatest impact, as they must present
complete and sufficient clinical evaluation files to ensure timely assessment and favourable
review of their products.
BENEFITS OF CONDUCTING CLINICAL EVALUATION
Regulatory conformance: Conforming to the regulatory framework is necessary and should
be done in order to get Notified Body approval for Technical file followed by CE Certification.
Choosing to perform a clinical investigation is the best answer to conform to the new
requirements of the Medical Device Directive MDD 93/42/EEC.
Early detection and resolution of safety issues: Over 25% of safety alerts are due to
manufacturing defects. This high proportion demonstrates the importance of an early
detection of potential safety issues. Early assessment gives the manufacturer time to take
corrective action and to provide potential damages to the companys reputation.
Manufacturer Image: An early clinical investigation most often results in a positive image and
greater usage of the product throughout the healthcare system. A well-designed clinical
investigation based on a robust methodology gives a chance to publish a scientific article in a
peer-reviewed journal.
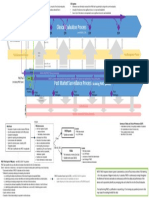
HOW TO FOLLOW STANDARD REQUIRMENT
ANALYSIS OF CLINICIAL DATA by
(a)
(b)
(c)
Clinical investigation(s) of the device concerned
Studies reported in the scientific literature of a
similar device
Published and/or unpublished reports on other
clinical experience of either the device in question
or a similar device
CLINICAL
EVALUATION
POST MARKET
a)
Surveillance (PMS)
b)
c)
Clinical Follow-up (PMCF)
Residual Risk
METHODOLOGY WHEN PERFORMING A CLINICAL EVALUATION
The most important part lies in the methodology used. Any evaluation must be conducted
following a strict adherence to regulatory requirements is the success of the clinical study. A
proper methodology saves lot of time and success.
APPOINT I 3 CONSULTING FOR BETTER NAVIGATION AND RESULTS
Working early with I 3 Consulting for smooth and error free submission to Notified Bodies.
Our project managers work with clients to ensure clinical evaluation was adequate and in
line with intended use, labelling, equivalent devices and in conformance with MEDDev
Guidelines.
KNOW OUR FEES
Complete our Quote Request Form for a detailed proposal. For more urgent requests, feel
free to use our Website Skype link or direct dialling numbers to USA & India.
Class of Medical Device
Activity
1
1s/m
11a
11b
111
Guidance on MDD Requirements
$ 50
$ 100
$ 150
$ 200
$ 250
Development of Protocol / Procedure
$ 50
$ 50
$ 50
$ 50
$ 50
Clinical Evaluation & Documentation
$ 300
$ 400
$ 500
$ 600
$ 700
Post Market Surveillance Procedure & Plan
$ 100
$ 150
$ 200
$ 250
$ 300
Post Market clinical follow-up Procedure & Plan
$ 100
$ 150
$ 200
$ 250
$ 300
GAP Analysis of Risk Management File
$ 100
$ 150
$ 200
$ 250
$ 300
Vigilance Control & Related Forms
$ 100
$ 100
$ 100
$ 100
$ 100
Taxes Extra
GET A CUSTOMIZED PROPOSAL
Complete our Quote Request Form for a detailed proposal. For more urgent requests, feel
free to use our Website Skype link or direct dialling numbers to USA & India.
CONTACT INFORMATION
INDIA
S8, J J PARK 02, 4 CROSS, BHAVANI NAGAR,
CHRIST SCHOOL ROAD, S.G PALYA,
BANGALORE 560029
FAX + 91 804 160 8963
TURKEY
ANADOLU BULVARI GIMAT SITESI 3. BLOCK NO: 29
P06105 MUCUNKOY, ANKARA, TURKEY
FAX + 90 312 397 0058
USA
mail to us
2871 COASTAL DRIVE, AURORA
ILLINOIS 60503
FAX + 1 815 986 2632
www.i3cglobal.com
You might also like
- Medical Device Reporting Regulation and Electronic SubmissionDocument26 pagesMedical Device Reporting Regulation and Electronic Submissionvinaysetty100% (1)
- Life Strategy 101: How To Live Live by Vision, Work With Purpose, and Achieve More SuccessDocument31 pagesLife Strategy 101: How To Live Live by Vision, Work With Purpose, and Achieve More SuccessMichelle Casto100% (2)
- US Prep 510k Submission White Paper EMERGODocument10 pagesUS Prep 510k Submission White Paper EMERGORamboNo ratings yet
- Guide To Good Distribution Practice of Medicinal Products For Human Use v4Document36 pagesGuide To Good Distribution Practice of Medicinal Products For Human Use v4Roberto M. Vilches CerdaNo ratings yet
- 510 (K) Format Guidance, Including Standards Form, and Extensions Clinical Trial Form and 510 (K) PDFDocument27 pages510 (K) Format Guidance, Including Standards Form, and Extensions Clinical Trial Form and 510 (K) PDFMichael wangNo ratings yet
- NDA ProcessDocument3 pagesNDA Processdustymarie100% (2)
- MedDRA N SOPDocument32 pagesMedDRA N SOPsuri33350% (2)
- Post Market Surveillance: Global Guidance For Adverse Event Reporting For Medical DevicesDocument37 pagesPost Market Surveillance: Global Guidance For Adverse Event Reporting For Medical DevicesSachin KumarNo ratings yet
- 06 PSUR PBRER Thomas MunzDocument35 pages06 PSUR PBRER Thomas MunzMohabKamalNo ratings yet
- 8-Step Transition Process From The MDD To The MDR: White PaperDocument11 pages8-Step Transition Process From The MDD To The MDR: White Papervlsi_forever100% (1)
- Bsi Smart Support Post Market SurveillanceDocument10 pagesBsi Smart Support Post Market SurveillanceMauro CostaNo ratings yet
- 06 MDCG 2020-6 Guidance On Sufficient Clinical Evidence For Legacy DevicesDocument22 pages06 MDCG 2020-6 Guidance On Sufficient Clinical Evidence For Legacy DevicesSantiago IbañezNo ratings yet
- Court Document SummaryDocument15 pagesCourt Document SummaryAkAsh prAkhAr vErmA100% (1)
- The Duty for Sponor Oversight in Clinical Trials: Practical Guide: 2nd Edition Clinical Data ReviewFrom EverandThe Duty for Sponor Oversight in Clinical Trials: Practical Guide: 2nd Edition Clinical Data ReviewNo ratings yet
- Medical Device Technical Specification: TS-01: Good Distribution Practice For Medical Devices - RequirementsDocument33 pagesMedical Device Technical Specification: TS-01: Good Distribution Practice For Medical Devices - RequirementsSeanNo ratings yet
- Clinical Evaluation Report SampleDocument12 pagesClinical Evaluation Report Sampleibrahim kademogluNo ratings yet
- FDA Guidance Documents For Software Contained in Medical DevicesDocument23 pagesFDA Guidance Documents For Software Contained in Medical Deviceschance909100% (1)
- Special and Abbreviated 510 (K)Document18 pagesSpecial and Abbreviated 510 (K)hemkenbpNo ratings yet
- 4.2 Clinical Evaluation Report (RevDocument58 pages4.2 Clinical Evaluation Report (RevMarina Sova100% (2)
- MEDDEV 2.7.1 Clinical Evaluation Rev 3Document46 pagesMEDDEV 2.7.1 Clinical Evaluation Rev 3Kevin Shane50% (2)
- BT200 - Clinical Evaluation ReportDocument5 pagesBT200 - Clinical Evaluation ReportfjvillamunozNo ratings yet
- CE Guidelines Classification Medical DeviceDocument17 pagesCE Guidelines Classification Medical DeviceMehdi100% (4)
- Simap - MDR-2017-MDDDocument44 pagesSimap - MDR-2017-MDDCRYSTAL2100100% (1)
- FDA Guidance - 510 K ChecklistDocument3 pagesFDA Guidance - 510 K ChecklistHila Cohen100% (3)
- Cost-Contained Regulatory Compliance: For the Pharmaceutical, Biologics, and Medical Device IndustriesFrom EverandCost-Contained Regulatory Compliance: For the Pharmaceutical, Biologics, and Medical Device IndustriesNo ratings yet
- 2 - 7-1 - 04-2003 Clinical EvaluationDocument20 pages2 - 7-1 - 04-2003 Clinical EvaluationGABYNo ratings yet
- Template For PSUR - MPVDocument6 pagesTemplate For PSUR - MPV-MPV Thảo - QAQC100% (2)
- PMS PMCF CER RelationshipDocument1 pagePMS PMCF CER RelationshipMohammed HammedNo ratings yet
- European Medical Device Usability RequirementsDocument3 pagesEuropean Medical Device Usability RequirementsHong-Nam KimNo ratings yet
- A Practice Teaching Narrative of Experience in Off Campus InternshipDocument84 pagesA Practice Teaching Narrative of Experience in Off Campus InternshipClarenz Jade Magdoboy MonserateNo ratings yet
- Clinical EvaluationDocument30 pagesClinical Evaluationnicop100% (1)
- 800m Training Plan For Mando EmployeDocument7 pages800m Training Plan For Mando EmployeSunder Veera Karthikeyan100% (1)
- The FDA Group - The Complete Guide To EU-MDR TransitionDocument26 pagesThe FDA Group - The Complete Guide To EU-MDR TransitionMauro Costa100% (1)
- Applicable Standards and Common SpecificationsDocument4 pagesApplicable Standards and Common Specificationsaymohamad2020No ratings yet
- GHTF Sg5 n4 Post Market Clinical Studies 100218Document10 pagesGHTF Sg5 n4 Post Market Clinical Studies 100218India RoseNo ratings yet
- Clinical Evluation GiudenceDocument20 pagesClinical Evluation GiudenceabcNo ratings yet
- Substantial Equivalence Review of Medical DevicesDocument24 pagesSubstantial Equivalence Review of Medical DevicesJames LindonNo ratings yet
- Bsi MD Ivdr Readiness Review en GBDocument13 pagesBsi MD Ivdr Readiness Review en GBlsmetiskoNo ratings yet
- MDCG 2020 - 5 Clinical Evaluation - Equivalence - April 2020Document20 pagesMDCG 2020 - 5 Clinical Evaluation - Equivalence - April 2020Kangjin JeonNo ratings yet
- Supplement 1 Medical Device Product QuestionnaireDocument14 pagesSupplement 1 Medical Device Product QuestionnaireJi YuNo ratings yet
- Guide To Med Device ClassificationDocument15 pagesGuide To Med Device ClassificationTracy TreacherNo ratings yet
- MDR New Update GeneralDocument6 pagesMDR New Update Generalpramod baghelNo ratings yet
- Standard Operating Procedure Trial Master File and 421 Main Evidence in TheDocument33 pagesStandard Operating Procedure Trial Master File and 421 Main Evidence in Theahmed.bouchenakNo ratings yet
- Infections Associated With Reprocessed Urological Endoscopes - Letter To Health Care Providers - FDADocument3 pagesInfections Associated With Reprocessed Urological Endoscopes - Letter To Health Care Providers - FDAt merchant100% (1)
- 34-Standards and Regulatory Considerations (693-714)Document22 pages34-Standards and Regulatory Considerations (693-714)racut_khansatraNo ratings yet
- NIDCR Clinical TrialDocument67 pagesNIDCR Clinical TrialEnrique TrvjilloNo ratings yet
- Post MarketSurveillancePlanTemplateDocument3 pagesPost MarketSurveillancePlanTemplateVomanh HealthcareandFitness100% (1)
- EU Tech File White Paper EmergoDocument8 pagesEU Tech File White Paper Emergogobu269104No ratings yet
- 2a. Class A Compression DeviceDocument31 pages2a. Class A Compression DeviceCedric Bonneau100% (1)
- Biocontamination Control for Pharmaceuticals and HealthcareFrom EverandBiocontamination Control for Pharmaceuticals and HealthcareRating: 5 out of 5 stars5/5 (1)
- Clinical Study Report Template - GHTDocument22 pagesClinical Study Report Template - GHThpradeep100% (1)
- WWORTH SOP10ProjectManagementV2.2 140507Document21 pagesWWORTH SOP10ProjectManagementV2.2 140507Fery Alapola100% (1)
- NEWS CENTER Maine (NCM) Sent A List of Questions To The FDA and These Were Their ResponsesDocument2 pagesNEWS CENTER Maine (NCM) Sent A List of Questions To The FDA and These Were Their ResponsesNEWS CENTER MaineNo ratings yet
- Good Clinical Practice GuideFrom EverandGood Clinical Practice GuideRating: 5 out of 5 stars5/5 (1)
- Content of PSUR Medical DeviceDocument1 pageContent of PSUR Medical DeviceKabomed QANo ratings yet
- Medical Devices and IVDs: Fit for the new EU-Regulations: Your complete seminar for projekt, study and jobFrom EverandMedical Devices and IVDs: Fit for the new EU-Regulations: Your complete seminar for projekt, study and jobNo ratings yet
- Spentys - Technical File (Face Shield Mask)Document20 pagesSpentys - Technical File (Face Shield Mask)hitham shehataNo ratings yet
- DePuy ASR Surgeon Recall PackDocument10 pagesDePuy ASR Surgeon Recall PackKirkBernardNo ratings yet
- SOP Template For Preparation of A Clinical Trial Authorisation V4 1Document5 pagesSOP Template For Preparation of A Clinical Trial Authorisation V4 1DrSyeda Rima100% (1)
- BT750 - Clinical Evaluation ReportDocument45 pagesBT750 - Clinical Evaluation Reportfjvillamunoz100% (1)
- Postmarketing Drug Safety and Inspection ReadinessDocument57 pagesPostmarketing Drug Safety and Inspection Readinesssthug25No ratings yet
- HSA Post-Market Device Vigilance RequirementsDocument8 pagesHSA Post-Market Device Vigilance RequirementsSubashiиy PяabakaяaиNo ratings yet
- 8 Key Changes To Understand in The New European MDR and IVDRDocument6 pages8 Key Changes To Understand in The New European MDR and IVDRKabomed QANo ratings yet
- Medical Devices Proposal For A Regulation - 2012 - 542 - enDocument194 pagesMedical Devices Proposal For A Regulation - 2012 - 542 - enRoxanaBurlaNo ratings yet
- Corruption PDFDocument11 pagesCorruption PDFkaleemullahNo ratings yet
- Assessing The NeckDocument3 pagesAssessing The NeckAnne Joyce Lara AlbiosNo ratings yet
- Sources of Coherent RadiationDocument7 pagesSources of Coherent RadiationSubhash DhungelNo ratings yet
- Novena in Honor of St. Elizabeth Ann SetonDocument3 pagesNovena in Honor of St. Elizabeth Ann SetonLadiesofCharityUSANo ratings yet
- SRDF S LabDocument9 pagesSRDF S LabUma SekharNo ratings yet
- SPELD SA A Trip To The Top End-DSDocument16 pagesSPELD SA A Trip To The Top End-DSThien Tho NguyenNo ratings yet
- What Is Link AdaptationDocument4 pagesWhat Is Link AdaptationAshutosh SinghNo ratings yet
- Greenbelt Place ApartmentsDocument8 pagesGreenbelt Place ApartmentsTricia EnnisNo ratings yet
- Magnolia Residences Tower D Promo - 20% Downpayment OptionDocument1 pageMagnolia Residences Tower D Promo - 20% Downpayment OptionLiv ValdezNo ratings yet
- ID Kajian Hukum Perjanjian Perkawinan Di Kalangan Wni Islam Studi Di Kota Medan PDFDocument17 pagesID Kajian Hukum Perjanjian Perkawinan Di Kalangan Wni Islam Studi Di Kota Medan PDFsabila azilaNo ratings yet
- Function Point and Cocomo ModelDocument31 pagesFunction Point and Cocomo ModelParinyas SinghNo ratings yet
- DocxDocument2 pagesDocxNashNo ratings yet
- SDSDDocument114 pagesSDSDbhatiasabNo ratings yet
- MCD2040 T2 2019 Exam Content and StructureDocument18 pagesMCD2040 T2 2019 Exam Content and StructureheyitsmemuahNo ratings yet
- Antenna & Wave Propagation - Course OutlineDocument2 pagesAntenna & Wave Propagation - Course OutlineSabuj AhmedNo ratings yet
- ĐỀ CHUẨN MINH HỌA SỐ 03Document17 pagesĐỀ CHUẨN MINH HỌA SỐ 03Lê Thị Ngọc ÁnhNo ratings yet
- Usa Field Hockey Teaching Games For Understanding WorkshopDocument5 pagesUsa Field Hockey Teaching Games For Understanding Workshopapi-341389340No ratings yet
- ML for Humans: A Journey from Ignorance to OxfordDocument27 pagesML for Humans: A Journey from Ignorance to OxfordDivyanshu Sachan50% (2)
- Phronesis Volume 7 Issue 1 1962 (Doi 10.2307/4181698) John Malcolm - The Line and The CaveDocument9 pagesPhronesis Volume 7 Issue 1 1962 (Doi 10.2307/4181698) John Malcolm - The Line and The CaveNițceValiNo ratings yet
- The Oikos in Athenian LawDocument13 pagesThe Oikos in Athenian LawTúlio CarvalhoNo ratings yet
- Draft Resolution 1.1 SPECPOL LUMUN 2013Document8 pagesDraft Resolution 1.1 SPECPOL LUMUN 2013Hamza Hashim100% (1)
- scn625 Summativeeval SarahltDocument6 pagesscn625 Summativeeval Sarahltapi-644817377No ratings yet
- Richard Payne v. Scottie Burns, Sheriff Escambia County, J.B. Redman, LT., and Cecil White, Officer, 707 F.2d 1302, 11th Cir. (1983)Document2 pagesRichard Payne v. Scottie Burns, Sheriff Escambia County, J.B. Redman, LT., and Cecil White, Officer, 707 F.2d 1302, 11th Cir. (1983)Scribd Government DocsNo ratings yet
- Holliday - 2006 - Native-Speakerism - ELT JournalDocument3 pagesHolliday - 2006 - Native-Speakerism - ELT JournalBob HowesNo ratings yet
- About Nysc Ict CD in Imo StateDocument9 pagesAbout Nysc Ict CD in Imo StateIsoft PexNo ratings yet
- Almeda v. CaDocument10 pagesAlmeda v. CaRuss TuazonNo ratings yet