Professional Documents
Culture Documents
Bansal Pattern Thermodynamics 2 Solution by SKsinha See Chemistry Animations at Sinhalabcom PDF
Uploaded by
Anant JoshiOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Bansal Pattern Thermodynamics 2 Solution by SKsinha See Chemistry Animations at Sinhalabcom PDF
Uploaded by
Anant JoshiCopyright:
Available Formats
SINHAs I. I.T.
CHEMISTRY
-1-
THERMODYNAMICS
Problems
1. 1.25 m3 of air at 1800C at 8 bar is undergoing a constant pressure until the volume is doubled. Determine the
change in the entropy and enthalpy of air. , Given CP = 1.005 kJ/kg 0K, Cv = 0.718 kJ/kg 0K and R = 0.287 kJ/kg
0
K
1 Solution: Assuming air behaves like a perfect gas we have, CP = 1.005 kJ/kg 0K,
Cv = 0.718 kJ/kg 0K and R = 0.287 kJ/kg 0K
Given: V1 = 1.25 m3, T1 = 180 + 273 = 4530 K, P1 = P2 = 8 x 105N/m2 and V2 = 2V1
For a constant pressure process, change in entropy is
T
S 2 S1 mC P ln 2
T1
We have P1V1 = mRT1
8 x10 5 x1.25
7.69kg
287 x 453
PV
PV
Also 1 1 2 2
T1
T2
2 x1.25 x 453
T2
906 0 K
1.25
m
Therefore change in entropy = 7.69 x1.005 ln
906
453
= 5.596 kJ/0K
Change in enthalpy = Q1-2 = mCP (T2 T1) = 3500.99 kJ
1kg of air initially at 270C is heated reversibly at constant pressure until the volume is doubled, and then is heated
at constant volume until the pressure is doubled. For the total path find i) Work transfer, ii) Heat transfer, iii)
Change in entropy
2. Solution: Given: m = 1 kg, T1 = 3000 K V2 = 2V1,
P3 = 2P2 = 2P1
2.
P
2
1
V
Process 1-2: Constant pressure process
i) Work done, W1-2 = P (V2 V1)
= PV2 PV1
= mR (T2 T1)
P1V1 P2V2
T1
T2
V V
But P1 = P2 1 2
T1 T2
Also
But V2 2V1
Therefore T2 = 2T1 = 6000K
Therefore work done W1-2 = 1 x 0.287 x (600 300)
= 86.1 kJ
1-D-10 Talwandi & B-7. JAWAHAR NAGAR, KOTA.(RAJ) Ph-0744-2422383 .Mo-93149-05055
SINHAs I. I.T. CHEMISTRY
-2-
ii) From first law of TD, Heat Transfer, Q1-2 = W1-2 + (U2 U1)
= W1-2 + mCv (T2 T1)
= 86.1 + 1 x 0.718 x (600 300)
= 301.5 kJ
iii) Change in entropy,
S 2 S1 mC P ln
T2
T1
1(1.005) ln
600
300
= 0.6966 kJ/0K
Process 2-3: Constant Volume Process
Given, P3 = 2P2, T2 = 600 K
P2V2 P3V3
T2
T3
T P T x 2 P2
T3 2 3 2
P2
P2
We have
But V2 V3
= 2 x T2 = 12000K
i) Work done, W2-3 = 0
ii) Heat transfer, Q2-3 = W2-3 + (U3 U2)
= mCv (T3 T2)
= 430.8 kJ
iii) Change in entropy
S 3 S 2 mC P ln
T3
T2
= 0.4977 kJ/0K
Therefore work transfer in total path, W1-3 = W1-2 + W2-3
= 86.1 + 0
= 86.1 kJ
Heat transfer in total path, Q1-3 = Q1-2 + Q2-3
= 301.5 + 430.8
= 732.3 kJ
Change in entropy for the total path = (S3 S1) = (S3 S2) + (S2 S1)
= 0.4977 + 0.6966
= 1.1943 kJ/0K
A mass of air is initially at 2600C and 700 kPa, and occupies 0.028m3. The air is expanded at constant pressure to
0.084m3. A polytropic process with n = 1.5 is then carried out, followed by a constant temperature process which
completes a cycle. All the processes are reversible. (i) sketch the cycle in the P-v and T-s diagrams. (ii) find the
heat received and heat rejected in the cycle. (iii) find the efficiency of the cycle.
3 Solution: P1 = 700 kPa, T1 = 533K = T3, V1 = 0.028m3, V2 = 0.084m3
3.
We have P1V1 = mRT1
1-D-10 Talwandi & B-7. JAWAHAR NAGAR, KOTA.(RAJ) Ph-0744-2422383 .Mo-93149-05055
SINHAs I. I.T. CHEMISTRY
-3-
700 x 0.028
0.128kg
0.287 x533
T
PV
0.084
3
Now 2 2 2
T1
P1V1 0.027
Therefore T2 = 3 x 533 = 1559 K
n
Again
P2 T2 n 1 1599 0.5
3
(3) 27
P3 T3
533
1.5
Heat transfer in process 1-2, Q1-2 = mcp (T2 T1)
= 0.128 x 1.005 (1599 533)
= 137.13kJ
Heat transfer in process 2 -3,
Q2 3 U pdv
mc v T3 T2
mc v
On substituting Q2-3 = - 19.59 kJ
mR T2 T3
n 1
n
T3 T2
n 1
For process 3-1
Q3-1 = dU + W3-1
Q31 W31 pdv mRT1 ln
1
But dU = 0, i.e.,
P
v1
mRT1 ln 3
v3
P1
On substituting, Q3-1 = - 64.53 kJ
Heat received in the cycle 137.13 kJ
Heat rejected in the cycle Q2 = 19.59 + 64.53 = 84.12 kJ
The efficiency of the cycle
cycle 1
Q2
84.12
1
0.39 or 39%
Q1
137.13
1 kg of air at a pressure of 7 bar and a temperature of 900C undergoes a reversible polytropic process which may be
represented by PV1.1 = C, final pressure is 1.4 bar. Evaluate i) The final specific volume, temperature and increase
in entropy, ii) Work done and heat transfer.
4 Solution: Given, m = 1 kg,
P1 = 7 bar,
T1 = 3630K,
PV1.1 = C,
P2 = 1.4 bar
Air is perfect gas i.e., P1V1 = mRT1
4.
v1
1( 287)(363)
0.14883m 3 / kg
7 x10 5
Also we have, P1V11.1 = P2V21.1
Therefore V2 = 0.6429 m3
Also P2V2 = mRT2
Therefore T2 = 313.610K
Change in entropy for a polytropic process is,
1-D-10 Talwandi & B-7. JAWAHAR NAGAR, KOTA.(RAJ) Ph-0744-2422383 .Mo-93149-05055
SINHAs I. I.T. CHEMISTRY
-4-
V
n
R. ln 2
S 2 S1
1
V1
Substituting the values, noting = 1.4 and R = 0.287 kJ/kg0K, we get
S2 S1 = 0.31495kJ/kg0K
Work done, W1 2
P1V1 P2V2
n 1
Substituting we get, W1-2 = 141.75 kJ/kg
Heat transfer Q1-2 = W1-2 + (U2 U1)
= W1-2 + mCv (T2 T1)
= 141.75 35.462
= 106.29 kJ/kg
5. Show that for a reversible adiabatic process the equation is Pv = Constant.
5 Solution: The general property relations for an ideal gas may be written as
Tds = du + pdv
= cvdT + pdv
And, also Tds = dh vdp
= cpdT vdp
For a reversible adiabatic change, ds = 0
Therefore cv dT = - pdv
And
cp dT = vdp
By division,
cp
cv
Or
vdp
pdv
dp
dv
0
p
v
Or
d (ln p) + d (ln v) = d (ln c)
Therefore ln p + ln v = ln c
i.e., pv = constant
where c is the constant
A mass of 0.25 kg of an ideal gas has a pressure of 300 kPa, a temperature of 800C, and a volume of 0.07 m3. The
gas undergoes an irreversible adiabatic process to a final pressure of 300 kPa and final volume of 0.1 m3 during
which the work done on the gas is 25 kJ. Evaluate the cp and cv of the gas and increase in entropy of the gas.
6 Solution: We have, P1 V1 = mRT1
6.
300 x 0.07
0.238kJ / kgK
0.25 x353
PV
300 x 0.1
Final temperature T2 2 2
505 K
mR
0.25 x 0.238
We have from first law of TD, Q W = U2 U1
But Q = 0
Therefore W = U1 U2
W = mcv (T1 T2)
On substituting, -25 = 0.25 x cv x (353 505)
Therefore cv = 0.658 kJ/kg-K
Now R = cp cv
Therefore cp = 0.896 kJ/kg-K
Entropy change
S 2 S1 mcv ln
P2
V
mc p ln 2
P1
V1
1-D-10 Talwandi & B-7. JAWAHAR NAGAR, KOTA.(RAJ) Ph-0744-2422383 .Mo-93149-05055
SINHAs I. I.T. CHEMISTRY
mc p ln
-5-
V2
0.10
0.25 x0.896 ln
V1
0.07
= 0.08 kJ/kg-K
7.
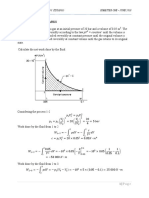
An ideal gas cycle consisting of three processes uses Argon (M = 40) as a working substance. Process 1-2 is a
reversible adiabatic expansion from 0.014m3, 700 kPa, 2800C to 0.056m3. Process 2-3 is a reversible isothermal
process and process 3-1 is a constant pressure process. Sketch the cycle on P-v and T-s diagrams and determine (i)
work transfer in each of the three processes (ii) heat transfer in each of the three processes and (iii) net work output
from the cycle. Assume for Argon = 1.67.
Solution: M = 40, V1 = 0.014m3, P1 = 700 kPa, T1 = 553 K
V2 = 0.056m3, = 1.67
Figure P-v and T-s diagrams
Process 1-2 Reversible adiabatic process
R 8.3143
0.208kJ / kg K
M
40
R
0.208
cv
0.311kJ / kg K
1 1.67 1
cp = 0.208 + 0.311 = 0.519 kJ/kg-K
Since the process is reversible adiabatic, the working substance is treated as an ideal gas
V
i.e., P V P2V or P2 P1 1
V2
or P2 69.13kPa
1 1
W12
0.014
700 x
0.056
1.67
V
0.014
Also T2 T1 1 553 x
218.45 K
0.056
V2
P V P2V1 700 x10 3 x0.014 69.13x10 3 x0.056
1 1
J
1
1.67 1
0.67
= 8.85 x 103J = 8.85 kJ
Q1-2 = 0 as the process is reversible adiabatic.
Process 2-3 Reversible isothermal process
T2 = T3 = 218.45 K
But
P
V
P
W2 3 mRT2 ln 3 mRT2 ln 2 mRT2 ln 2
V2
P1
P3
3
PV
700 x10 x0.014
m 1 1
0.2157 kg
RT1 0.208 x10 3 x 218.45
1-D-10 Talwandi & B-7. JAWAHAR NAGAR, KOTA.(RAJ) Ph-0744-2422383 .Mo-93149-05055
SINHAs I. I.T. CHEMISTRY
-6-
69.13
W2 3 0.2157 x 0.208 x10 3 x 218.45 x ln
700
= - 22.69 x 103J = - 22.69 kJ
By first law of TD Q2-3 = (U3 U2) + W2-3
But U3 U2 = mcv (T3 T2) = 0
Therefore Q2-3 = W2-3 = - 22.69 kJ
Process 3-1 Constant Pressure Process
W3-1 = (P1V1 P3V3) = mR (T1 T3)
= 0.2157 x 0.208 x 103 (553 218.45) = 15 kJ
Therefore Net work output from the cycle = Wc = W1-2 + W2-3 + W3-1
= 8.85 22.69 + 5.93
= - 7.91 kJ
For the cyclic process we have
Q W
or Q1 2 Q2 3 Q31 W
0 - 22.69 + Q3-1 = - 7.91
Therefore Q3-1 = 14.78 kJ
8.
A mass of an ideal gas exists initially at 200 kPa, 300K and 0.5 m3/ kg. The value of is 1.4. (i)Determine the
specific heats of the gas. (ii)What is the change in entropy when the gas is expanded to a pressure of 100 kPa
according to the law pv1.3 = constant (iii) What will be the change in entropy if the process is according to the law
pv1.5 = constant (iv) What inference you can draw from this example.
Solution: P1 = 200 kPa, T1 = 300 K, v1 = 0.5 m3/kg, =1.4, P2 = 100 kPa
(i) For an ideal gas P1v1 = RT1 or
P1v1
T1
333.33 J / kg K
cv
R
833.33 J / kg K
1
c p c v 1166.66 J / kg K
(ii) For the given process
P1v1n P2 v 2n
1
n
P
or v 2 1 v1 0.852m 3 / kg
P2
P v
Also T2 2 2 255.6 K
R
Change in entropy per unit mass is given by
T
S 2 S1 c v ln 2
T1
v
R ln 2
v1
= 44.18J/K
(iii) For the process for which the index n = 1.5 we have
1
200 1.5
3
v2
x0.5 0.764m / kg
100
1-D-10 Talwandi & B-7. JAWAHAR NAGAR, KOTA.(RAJ) Ph-0744-2422383 .Mo-93149-05055
SINHAs I. I.T. CHEMISTRY
-7-
100 x10 3 x0.794
T2
238.2 K
333.33
238.2
0.794
S 2 S1 833.33 x ln
333.33 x ln
300
0 .5
= - 38.07 J/K
(iv) It can be seen from the results of (ii) and (iii) above that entropy increases when n < and it decreases when n
> .
A closed rigid cylinder is divided by a diaphragm into two equal compartments, each of volume 0.1 m3. Each
compartment contains air at a temperature of 200C. The pressure in one compartment is 2.5 MPa and in the other
compartment it is 1 MPa. The diaphragm is ruptured so that the air in the compartments mix to bring the pressure to
a uniform value through the insulated cylinder. Find the net change in entropy due to mixing process.
9 Solution: Let suffix 1 denote the condition of air in the left half of the cylinder, suffix 2 the conditions in the right
half of the cylinder and suffix 3 the condition after mixing
It if given that,
V1 = V2 = 0.1 m3, T1 = T2 = 20 + 273 = 293K, P1 = 2.5 MPA, P2 = 1.0 MPa
After mixing the temperature is given by
9.
T3
m1 c p1T1 m2 c p 2T2
m1c p1 m 2 c p 2
Since T1 = T2 and cp1 = cp2, it follows that T3 = T1 = T2 = 293 K
(S)universe = (S)1 + (S)2 + (S)surroundings
(S)surroundings = 0 as the cylinder is insulated and no heat transfer can take place during the mixing process.
Therefore (S)universe = (S)1 + (S)2
T
v
T
v
m1 cv ln 3 R ln 3 m2 cv ln 3 R ln 3
T1
v1
T2
v2
6
Pv
2.5 x10 x0.1
m1 1 1
2.973kg
RT1
287 x 293
1.0 x10 6 x0.1
Similarly m2
1.189kg
287 x 293
Therefore on substituting, (S)universe = 0.828kJ/K
10. Air is compressed from 1 bar, 270C to 4.5 bar, 1770C. Determine the change in entropy per kg of air (i) taking
variation of specific heats into account and (ii) assuming specific heat at constant pressure to be constant and is
equal to 1.01 kJ/kg-K over this range of temperature.
10 Solution: T1 = 300 K, T2 = 450 K, P1 = 1 bar, P2 = 4.5 bar
p
s 2 s1 s 20 s10 R ln 2
p1
0
From table C-21 at T1 = 300 K, s1 2.5153kJ / kg K and
(i) We have change in entropy per kg of air,
at T2 = 450 K, s 2 2.9245kJ / kg
Therefore on substituting, s2 s1 = - 0.02508 kJ/kg-K
0
(ii) The change in entropy per kg of air
s 2 s1 c p ln
T2
P
R ln 2
T1
P1
1-D-10 Talwandi & B-7. JAWAHAR NAGAR, KOTA.(RAJ) Ph-0744-2422383 .Mo-93149-05055
SINHAs I. I.T. CHEMISTRY
1.01 ln
-8-
450
4 .5
0.289 ln
0.0252kJ / kg K
300
1
11 a) A gaseous mixture contains 21% by volume of N2, 50% by volume of H2 and 29% by volume of CO2. Calculate
the gas constant of the mixture and the ratio of specific heats. If the mixture pressure is at 1 bar and the mixture
temperature is 100C, calculate the partial pressures and mass fractions of the constituents.
b) A cylinder contains 0.085m3 of this mixture at 1 bar and 100C. The gas undergoes a polytropic process according
to the law Pv1.2 = constant to a final volume which is one fifth of the initial volume. Determine i) the magnitude and
direction of work transfer, ii) magnitude and direction of heat transfer and iii) the change in entropy.
11 Solution: From tables C-6, MN2 = 28.02, MH2 = 2.016 and MCO2 = 44
From table C-11, CPN2 = 1.039 kJ/kg-K, CPH2 = 14.15 kJ/kg-K and CPCO2 = 0.818 kJ/kg-K
a) Given VN2 / V = VfN2 = 0.21, VH2 / V = VfH2 = 0.5, VCO2 / V = VfCO2 = 0.29
P = 1 bar, T = 283 K
We have R N 2
R
8.3143
0.297kJ / kg K
M N 2 28.013
RH 2
R
8.3143
4.124kJ / kg K
MH2
2.016
RCO 2
R
8.3143
0.189kJ / kg K
M CO 2
44
CvN2 = 1.039 0.297 = 0.742 kJ/kg-K
CvH2 = 14.15 4.124 = 10.026 kJ/kg-K
CvCO2 = 0.818 0.189 = 0.629 kJ/kg-K
We know that volume fraction = mole fraction
i.e., yN2 = VfN2 = 0.21
yH2 = VfH2 = 0.5
yCO2 = VfCO2 = 0.29
Molecular weight of the mixture Mm
= yN2 MN2 + yH2 MH2 + yCO2 MCO2
= 0.21 x 28.013 + 0.5 x 2.016 + 0.29 x 44
= 19.654 kg/kg-mole
Gas constant for the mixture = Rm = 8.3143 / 19.654
= 0.423 kJ/kg-K
Therefore
Mass fraction of N2
m fN 2
mN 2 nN 2 M N 2
M
yN 2 N 2
mm
nm M m
Mm
28.013
0.21x
0.2993
19.654
Similarly m fH 2 0.5 x
m fCO 2
2.016
0.0513
19.654
44
0.29 x
0.6494
19.654
For the mixture Cvm = MfN2 CvN2 + MfH2 CvH2 + MfCO2 CvCO2
= 0.2993 x 0.742 + 0.0513 x 10.026 + 0.6494 x 0.629
= 1.145 kJ/kg-K
CPm = Cv + Rm
= 1.145 + 0.423
= 1.568 kJ/kg-K
Therefore the ratio of specific heat for the mixture is m = CPm / Cvm
= 1.568 / 1.145
= 1.369
1-D-10 Talwandi & B-7. JAWAHAR NAGAR, KOTA.(RAJ) Ph-0744-2422383 .Mo-93149-05055
SINHAs I. I.T. CHEMISTRY
-9-
p V 1x10 5 x0.085
m 1 1
0.071kg
423 x 283
RT1
1.2
1. 2
For the given process, p1V1 p 2V2
b) Mass of the mixture
V
p 2 1
V2
p1 5 x1 6.9 bar
1. 2
1.2
p1V1 p 2V2
n 1
5
5 1
1x10 x 0.085 6.9 x10 x 5 x 0.085
1.2 1
16150 J
W1 2 Wd 1 2
Negative sign indicates that work is done by the surroundings on the gas mixture.
Temperature of the mixture at the end of the process is,
1
6.9 x10 5 x x0.085
pV
5
T2 2 2
mR
0.071x 423
390.6 K
p
T
Or 2 2
T1 p1
n 1
n
0.2
6.9 1.2
i.e., T2 283
390.5 K
1
Change in internal energy, U2 U1 = mCv (T2 T1)
= 0.071 x 1.145 x (390.6 283)
= 8.747 kJ
By first law of TD, Q1-2
= W1-2 + (U2 U1)
= - 16.15 + 8.747
= - 7.403 kJ
Negative sign indicates that heat transfer takes place from the mixture to the surroundings
Change in entropy of the mixture S 2 S 1 m C v ln 2 R ln 2
T1
V1
390.6
1
0.0711.145 x ln
0.423 x ln
283
5
0.02214kJ / K
12. Calculate the constant volume and constant pressure specific heats of a gas mixture consisting of 1 kg of
oxygen and 2 kg of nitrogen at a pressure of 1.5 bar and temperature 200C. Also determine the change in internal
energy, enthalpy and entropy of the mixture when it is heated under constant volume to a temperature of 1000C.
12 Solution: From tables C-6, MO2 = 32, MN2 = 28
From table C-11, CPN2 = 1.038 kJ/kg-K, CPO2 = 0.917 kJ/kg-K
CvN2 = CpN2 RN2 = 1.038 8.3143 / 28 = 0.741 kJ/kg-K
CvO2 = CpO2 RO2 = 0.917 8.3143 / 32 = 0.653 kJ/kg-K
cv m m fi cv i
i
1-D-10 Talwandi & B-7. JAWAHAR NAGAR, KOTA.(RAJ) Ph-0744-2422383 .Mo-93149-05055
SINHAs I. I.T. CHEMISTRY
- 10 -
1
2
x 0.653 x 0.741 0.712kJ / kg K
3
3
m fi c p i
p m
1
2
x 0.917 x1.038 0.998kJ / kg K
3
3
U mm cv m T2 T1
Change in internal energy,
= 3 x 0.712 (100 20)
= 170.88 kJ
Change in enthalpy,
H mm c p m T2 T1
= 3 x 0.998 (100 20)
= 239.52 kJ
Change in entropy of the mixture,
S m
T
mm c v m ln 2
T1
V
Rm ln 2
V1
Since the volume is constant, the change in entropy,
T
mm c v m ln 2
T1
373
3x 0.712 ln
293
S m
= 0.5156 kJ/K
13. A gaseous mixture consisting of 1 kg of helium and 2.5 kg of nitrogen is compressed isentropically in a closed
system from a pressure of 1 bar, 270C to a pressure of 7 bar. Assuming specific heats of helium and nitrogen to
be constant, determine the specific heats of the mixture, the change in entropy of individual gases, and the
change in internal energy of the mixture. Also find the work required for the compression process. Assume
CvHe = 3.14 kJ/kg-K, CvN2 = 0.741 kJ/kg-K, CpHe = 5.233 kJ/kg-K and CpN2 = 1.038 kJ/kg-K
13 Solution:
cv m m fi cv i
i
p m
1
2 .5
x3.140
x 0.741 1.426kJ / kg K
3 .5
3 .5
m fi c p i
i
1
2 .5
x5.233
x1.038 2.236kJ / kg K
3 .5
3 .5
c p m 2.236
1.568
cv m 1.426
p
T2 T1 2
p1
m 1
m
0.568
7 1.568
300
607 K
1
1-D-10 Talwandi & B-7. JAWAHAR NAGAR, KOTA.(RAJ) Ph-0744-2422383 .Mo-93149-05055
SINHAs I. I.T. CHEMISTRY
- 11 -
Hence the change in entropy of helium
p He , 2
T
m He c p He ln 2 R He ln
T1
p He,1
p N ,2
p He , 2
p
7
2 2
We know that ,
p He ,1
p N 2 ,1
p1 1
S He
607
7
S He 15.233 ln
2.093 ln
300
1
= - 0.38 kJ/K
Similarly S N 2 2.51.038 ln
0.297 ln
300
1
607
= + 0.38 kJ/K
Internal energy of the mixture
U m mm cv m T2 T1
= 3.5 x 1.426 (607 300)
= 1532.24 kJ
For a closed system we have, q w = du.
Since the process adiabatic, q = 0
Therefore w = -dU.
Therefore the work required for the compression process,
W1-2 = - (dU)m = - 1532.24 kJ
14. Two thermally insulated vessels, each of 0.85 m3 in volume are isolated from each other by a partition wall. One of
the vessels contains nitrogen and the other oxygen, each at 5 bar and 1000C. As soon as the partition between the
vessels is removed, the two gases mix adiabatically. Determine the increase in entropy of the gas mixture.
14. Solution: Since the pressure and temperature of each gas are equal, the pressure and temperature of the mixture
will not change, i.e., the mixture will be at 5 bar and 373 K. But the volume of the mixture will be 2x0.85= 1.7m3 and
the pressure of N2 and O2 is
p N2
pm
v N2
vm
0.85 1
1.7
2
Therefore pN2=0.5x5=2.5 bar and pO2=2.5 bar
Using perfect gas equation, mass of the N2,
2.5 x10 5 x1.7
3.84kg
8.3143 x10 3
x373
28
2.5 x10 5 x1.7
and mO2
4.39kg
8.3143 x10 3
x373
32
mN2
S m
Therefore the increase in entropy of the mixture is,
m N 2 R N 2 ln y N 2 mO2 RO2 ln y O2
8.3143 2.5
8.3143 2.5
3.84 x
ln
4.39 x
ln
28
5
32
5
=1.579kJ/K
15. Two kg-mole of CO2 at a pressure of 1.8 bar, 800C is mixed in a thermally insulated vessel with 3 kg-mole of N2 at
2.2 bar, 600C. When the mixture is at equilibrium, determine the final temperature and pressure and the change in
entropy of the mixture. Assume CvCO2 = 0.653 kJ/kg-K, CvN2 = 0.741 kJ/kg-K
1-D-10 Talwandi & B-7. JAWAHAR NAGAR, KOTA.(RAJ) Ph-0744-2422383 .Mo-93149-05055
SINHAs I. I.T. CHEMISTRY
15 Solution: we have, Tm
cv CO
- 12 -
nCO2 cv CO2 TCO2 n N 2 cv N 2 TN 2
nm cv m
M CO2 xc v CO2
Now the molal specific heats at constant volume of the constituent gases are
2
= 44x0.653
=28.73 kJ/kg-mole K
cv N
And
M N 2 xcv N 2
=28x0.741
=20.75 kJ/kg-mole K
Tm
2 x 28.73 x353 3 x 20.75 x333
2 x 28.73 3 x 20.75
=342.6 K
The final pressure of the mixture is
pm
nm R Tm
Vm
The volume of the individual gases is,
nCO2 R TCO2
VCO2
VN2
pCO2
n N 2 R TN 2
p N2
2 x8.3143x353
32.61m 3
100 x1.8
3 x8.3143x333
37.75m 3
100 x 2.2
Vm VCO2 V N 2 32.61 37.75 70.36m 3
Therefore the final pressure of the gas mixture is,
pm
5 x8.3143 x342.6
2.02 bar
100 x 70.36
The partial pressure of the individual gases,
pCO2
nCO2
nm
xp m
2
x 2.02 0.808 bar
5
p N2
nN2
S CO2 S N 2
But
nm
xp m
3
x 2.02 1.212 bar
5
Therefore the entropy change of the mixture,
m
pCO2 , 2
T
nCO2 cv CO2 ln m R ln
TCO2
p CO2 ,1
p N ,2
T
n N 2 c v N 2 ln m R ln 2
TN 2
p N 2 ,1
p CO
2
R c v CO2 8.3143 28.73
=37.04 kJ/kg mole K
1-D-10 Talwandi & B-7. JAWAHAR NAGAR, KOTA.(RAJ) Ph-0744-2422383 .Mo-93149-05055
and
SINHAs I. I.T. CHEMISTRY
p N
2
R c v N 2 8.3143 20.75
- 13 -
=29.064 kJ/kg mole K
342.6
1.212
2 37.04 ln
8.3143 ln
353
1.8
342.6
1.212
529.064 ln
8.3143 ln
333
2.2
=2[-1.108+6.659]+3[0.826+4.957]
=28.45 kJ/ K
1-D-10 Talwandi & B-7. JAWAHAR NAGAR, KOTA.(RAJ) Ph-0744-2422383 .Mo-93149-05055
You might also like
- Thermo ProblemsDocument8 pagesThermo ProblemsChrister John UyNo ratings yet
- AE 231 Thermodynamics Recitation 7 Recitation 7: Instructor: Assoc. Prof. Dr. Sinan EyiDocument10 pagesAE 231 Thermodynamics Recitation 7 Recitation 7: Instructor: Assoc. Prof. Dr. Sinan EyiOnur ÖZÇELİKNo ratings yet
- Thermodynamics Worked ExamplesDocument13 pagesThermodynamics Worked ExamplesSalah Salman100% (1)
- HWSolutions PDFDocument42 pagesHWSolutions PDFJames AhnNo ratings yet
- Engineering Mechanics: Second PartDocument9 pagesEngineering Mechanics: Second Partاحمد سلمان عزيز , مسائيCNo ratings yet
- ENGR 2213 Thermodynamics: F. C. Lai School of Aerospace and Mechanical Engineering University of OklahomaDocument20 pagesENGR 2213 Thermodynamics: F. C. Lai School of Aerospace and Mechanical Engineering University of OklahomaSebastián SantarrosaNo ratings yet
- Me6301 QBDocument46 pagesMe6301 QBNaveen Dhanuraj100% (1)
- Ideal Gas Processes ExampleDocument14 pagesIdeal Gas Processes ExampleMary Joy AlmerenoNo ratings yet
- Solution Week 9Document6 pagesSolution Week 9Ariadne ChuaNo ratings yet
- Thermodynamics Problem SetDocument2 pagesThermodynamics Problem SetElise BurchNo ratings yet
- Unit Four Homework Solutions, September 23. 2010: Mechanical Engineering 370 ThermodynamicsDocument3 pagesUnit Four Homework Solutions, September 23. 2010: Mechanical Engineering 370 ThermodynamicsRengganis Putri ParmudyaNo ratings yet
- Unit Nine Homework Solutions, November 9, 2010: V V R T T C S S Const C If V V R T DT C S SDocument5 pagesUnit Nine Homework Solutions, November 9, 2010: V V R T T C S S Const C If V V R T DT C S SAbubakar AdeniNo ratings yet
- CH 10Document34 pagesCH 10hirenpatel_universalNo ratings yet
- Tutorial 3 SolnDocument2 pagesTutorial 3 Solnkaeshav manivannanNo ratings yet
- Solved - Problems in ThermodynamicsDocument29 pagesSolved - Problems in ThermodynamicsAngelica Joyce Benito100% (6)
- HW 5 SolnDocument7 pagesHW 5 SolnNik Hafiy HafiziNo ratings yet
- Thermodynamics (Solved Prob)Document22 pagesThermodynamics (Solved Prob)Jubert Perez100% (1)
- Extra Sol8 PDFDocument8 pagesExtra Sol8 PDFJimmy JohnsNo ratings yet
- CLASS TUTORIAL 2 (Individual) 1.: v1 0.25779 m3/kg v2 0.5v1 0.12890 m3/kgDocument1 pageCLASS TUTORIAL 2 (Individual) 1.: v1 0.25779 m3/kg v2 0.5v1 0.12890 m3/kgmarkNo ratings yet
- Chapter 5 Processes of Ideal GasesDocument13 pagesChapter 5 Processes of Ideal Gasesyeuxvertsu100% (1)
- ChE234 F10 Exam2 Thermodynamics For Chemical EngineersDocument3 pagesChE234 F10 Exam2 Thermodynamics For Chemical EngineerskellykapperNo ratings yet
- Ps CsDocument15 pagesPs CsChristopher GalasNo ratings yet
- Thermodynamics Chapter 3 Solution Sta Maria PDFDocument7 pagesThermodynamics Chapter 3 Solution Sta Maria PDFZandie Garcia75% (4)
- Reviewlecture-I 20081001 48e3c2399f4d65 74115154Document37 pagesReviewlecture-I 20081001 48e3c2399f4d65 74115154Austin BarrilleauxNo ratings yet
- Workbook Workbook Workbook Workbook Workbook: Try Yourself QuestionsDocument14 pagesWorkbook Workbook Workbook Workbook Workbook: Try Yourself QuestionsAnu Sandeep AggrawalNo ratings yet
- Non Flow Processes Gases Closed SystemDocument9 pagesNon Flow Processes Gases Closed SystemNjabulo NgobeseNo ratings yet
- Homework 11Document8 pagesHomework 11Ha ViNo ratings yet
- Thermo 5th Chap17 P096Document19 pagesThermo 5th Chap17 P096UTA - Std - Elvin ChantreNo ratings yet
- Ciclo BraytonDocument17 pagesCiclo BraytonNubia Bergamini100% (2)
- Thermodynamics Chapter 3 Solution Sta MariaDocument7 pagesThermodynamics Chapter 3 Solution Sta MariaJean PD81% (21)
- Moving Boundary Work: 5 KG of Saturated Water Vapor in Cylinder Is Heated at Constant Pressure (300 Kpa)Document8 pagesMoving Boundary Work: 5 KG of Saturated Water Vapor in Cylinder Is Heated at Constant Pressure (300 Kpa)yeng botzNo ratings yet
- Engg ThermodynamicsgfDocument3 pagesEngg Thermodynamicsgfphysics a2No ratings yet
- BSGS Sample Problems 2 - BB CollabDocument21 pagesBSGS Sample Problems 2 - BB CollabNeo GarceraNo ratings yet
- Chapter 5 - Section A - Mathcad Solutions: 5.2 Let The Symbols Q and Work Represent Rates in Kj/s. Then by Eq. (5.8)Document21 pagesChapter 5 - Section A - Mathcad Solutions: 5.2 Let The Symbols Q and Work Represent Rates in Kj/s. Then by Eq. (5.8)light2618No ratings yet
- Thermodynamics Advanced HomeWork 1Document11 pagesThermodynamics Advanced HomeWork 1فیضان قادرNo ratings yet
- Section e To Be Solved Set 1Document14 pagesSection e To Be Solved Set 1Neil SequioNo ratings yet
- Since Volume Is Constant, Use Charles' Law On Constant VolumeDocument7 pagesSince Volume Is Constant, Use Charles' Law On Constant VolumetrishaNo ratings yet
- Unit 3 TNSDocument12 pagesUnit 3 TNSravitejabvNo ratings yet
- Term Odin A MicaDocument10 pagesTerm Odin A MicaFelipe De Lima RomeroNo ratings yet
- Phys 181 CH 2122 Sample ProblemsDocument4 pagesPhys 181 CH 2122 Sample Problemsluis atenciaNo ratings yet
- AlphaDocument7 pagesAlphaJojenNo ratings yet
- 2-2 First Law of Thermodynamics - Session 2Document17 pages2-2 First Law of Thermodynamics - Session 2Baddam Jayasurya ReddyNo ratings yet
- Sol Assign 1Document2 pagesSol Assign 1Jerome MagnoNo ratings yet
- Thermodyancs Chapter 9 Solution ManuelDocument36 pagesThermodyancs Chapter 9 Solution ManuelFarhad MojaverNo ratings yet
- Sample Problem ThermoDocument25 pagesSample Problem ThermoJonnah Faye Mojares0% (1)
- Theory of Elasticity by Sadhu SinghDocument7 pagesTheory of Elasticity by Sadhu Singhotonbara14% (7)
- Applied Thermodynamics Exam 2018 Wirh SolutionsDocument9 pagesApplied Thermodynamics Exam 2018 Wirh SolutionsFarouk BassaNo ratings yet
- Isobaric ProcessDocument24 pagesIsobaric ProcessBrando_Balagon75% (4)
- A Modern Course in Statistical PhysicsFrom EverandA Modern Course in Statistical PhysicsRating: 3.5 out of 5 stars3.5/5 (2)
- Solution Manual for an Introduction to Equilibrium ThermodynamicsFrom EverandSolution Manual for an Introduction to Equilibrium ThermodynamicsNo ratings yet
- Chapter 1Document18 pagesChapter 1Anant JoshiNo ratings yet
- Atomic Structure Type 1Document20 pagesAtomic Structure Type 1Sudhakar ChollangiNo ratings yet
- ॥ श्रीदत्तसहस्रनामस्तोत्रम् ॥Document8 pages॥ श्रीदत्तसहस्रनामस्तोत्रम् ॥Anant JoshiNo ratings yet
- Properties of Natural ResourcesDocument1 pageProperties of Natural ResourcesAnant JoshiNo ratings yet
- Chapter2 WorkbookDocument20 pagesChapter2 WorkbookAnant Joshi33% (3)
- Adding With Some Regrouping (A)Document2 pagesAdding With Some Regrouping (A)Nur Maizatun AtmadiniNo ratings yet
- 1 Carlys Family Free SampleDocument3 pages1 Carlys Family Free SampleDiego UrbanoNo ratings yet
- Venn Diagram - Two CirclesDocument2 pagesVenn Diagram - Two CirclesdennieboyNo ratings yet
- Basic-Column1 2 PDFDocument2 pagesBasic-Column1 2 PDFAnant JoshiNo ratings yet
- Student Name: - ScoreDocument2 pagesStudent Name: - ScoreGelay Gerlie Cadiente PitpitNo ratings yet
- 4D R Subtraction WS 1Document1 page4D R Subtraction WS 1noddynoddyNo ratings yet
- 3 Addends Unlike Improper2Document2 pages3 Addends Unlike Improper2Anant JoshiNo ratings yet
- Name: - ScoreDocument2 pagesName: - ScoreAnant JoshiNo ratings yet
- 12 Chemistry Exemplar Chapter 4 AnswerDocument3 pages12 Chemistry Exemplar Chapter 4 AnswerAnant JoshiNo ratings yet
- 3dice TemplateDocument1 page3dice TemplateAnant JoshiNo ratings yet
- 3 Addends Unlike Proper Mixed1Document2 pages3 Addends Unlike Proper Mixed1Anant JoshiNo ratings yet
- 3 Addends Unlike Proper Mixed3Document2 pages3 Addends Unlike Proper Mixed3Anant JoshiNo ratings yet
- Dice Addition: Count The Dots On Each Die and Add ThemDocument2 pagesDice Addition: Count The Dots On Each Die and Add ThemAnant JoshiNo ratings yet
- Dice Addition: Count The Dots On Each Die and Add ThemDocument2 pagesDice Addition: Count The Dots On Each Die and Add ThemAnant JoshiNo ratings yet
- 3 Addends Unlike Proper3Document2 pages3 Addends Unlike Proper3Anant JoshiNo ratings yet
- 3 Addends Unlike Improper2 PDFDocument2 pages3 Addends Unlike Improper2 PDFAnant JoshiNo ratings yet
- 3 Addends Like Proper Mixed1Document2 pages3 Addends Like Proper Mixed1Anant JoshiNo ratings yet
- 3 Addends Like Proper3 PDFDocument2 pages3 Addends Like Proper3 PDFAnant JoshiNo ratings yet
- 3 Addends Unlike Proper Mixed2Document2 pages3 Addends Unlike Proper Mixed2Anant JoshiNo ratings yet
- Dice Addition: Count The Dots On Each Die and Add ThemDocument2 pagesDice Addition: Count The Dots On Each Die and Add ThemAnant JoshiNo ratings yet
- 3 Addends Unlike Improper Mixed1 PDFDocument2 pages3 Addends Unlike Improper Mixed1 PDFAnant JoshiNo ratings yet
- 3 Addends Unlike Improper Mixed2Document2 pages3 Addends Unlike Improper Mixed2Anant JoshiNo ratings yet
- 3 Addends Unlike Mixed3Document2 pages3 Addends Unlike Mixed3Anant JoshiNo ratings yet
- 3 Addends Like Proper1 PDFDocument2 pages3 Addends Like Proper1 PDFAnant JoshiNo ratings yet
- 3 Addends Like Proper1Document2 pages3 Addends Like Proper1Anant JoshiNo ratings yet
- Hukum Pertama TermodinamikaDocument123 pagesHukum Pertama TermodinamikaLia TrisnawatiNo ratings yet
- Thermodynamics - Shobhit Nirwan 2Document38 pagesThermodynamics - Shobhit Nirwan 2Utkarsh kumarNo ratings yet
- Measuring Plasma Arc Gas Enthalpy by Energy Balance: Standard Practice ForDocument5 pagesMeasuring Plasma Arc Gas Enthalpy by Energy Balance: Standard Practice Forjun floresNo ratings yet
- Liquefaction ProcessDocument4 pagesLiquefaction ProcessSwathiMKarunaNo ratings yet
- Conceptual Design I Project I: Prepared byDocument27 pagesConceptual Design I Project I: Prepared byAinulDiliansyah100% (2)
- TE - Mech - RAC - Chapter 5 - Thermodyn Propertis of Moist AirDocument48 pagesTE - Mech - RAC - Chapter 5 - Thermodyn Propertis of Moist AirAkshay ChandoleNo ratings yet
- Design of DryerDocument11 pagesDesign of DryerHARKULVINDER SINGHNo ratings yet
- Dịch Hóa Lý Nâng CaoDocument2 pagesDịch Hóa Lý Nâng CaoTrương Hữu LộcNo ratings yet
- ThermoDynamics Lecture NotesDocument45 pagesThermoDynamics Lecture NotesHarsha Gandikota100% (2)
- MA OPTISWIRL4200 en 200701 4003930808 R08Document124 pagesMA OPTISWIRL4200 en 200701 4003930808 R08abbas dehghanNo ratings yet
- Kloppers and KR Ger-2004 20 600 Dpi - 2004 20 1 24-29 PDFDocument6 pagesKloppers and KR Ger-2004 20 600 Dpi - 2004 20 1 24-29 PDFFernandoNo ratings yet
- Report Simulation PDDocument18 pagesReport Simulation PDAtika Mohd YatimNo ratings yet
- Pinch Technology AnalysisDocument20 pagesPinch Technology AnalysisPiyush JainNo ratings yet
- H2 Refuling Assement of Composite Storage Tank For Fuel Cell VehicleDocument9 pagesH2 Refuling Assement of Composite Storage Tank For Fuel Cell VehicleLuís BarbosaNo ratings yet
- 125 - Performance Guarantee - Maan - R4Document4 pages125 - Performance Guarantee - Maan - R4Ahmad ShekhNo ratings yet
- Gate ThermoDocument215 pagesGate Thermokapil kumarNo ratings yet
- AET Question Bank For AUC R2013 - SDocument5 pagesAET Question Bank For AUC R2013 - SGurunath AeroNo ratings yet
- Simulation of IGCC in PROII PDFDocument11 pagesSimulation of IGCC in PROII PDFkishna009No ratings yet
- Prs Pressure Reducing Control ValvesDocument4 pagesPrs Pressure Reducing Control ValvesAnkit PalNo ratings yet
- Pipe CNS 05Document42 pagesPipe CNS 05maria katherine pantojaNo ratings yet
- FluidEXL Graphics LibIF97 Docu Stud EngDocument50 pagesFluidEXL Graphics LibIF97 Docu Stud EngSahiFam SocioNo ratings yet
- Energy Balance Untuk Teknik KimiaDocument19 pagesEnergy Balance Untuk Teknik Kimiamelisa amaliaNo ratings yet
- StandardsDocument117 pagesStandardsaharish_iitkNo ratings yet
- A Definite Area or Space Where Some Thermodynamic Process Takes Place Is Known AsDocument13 pagesA Definite Area or Space Where Some Thermodynamic Process Takes Place Is Known Asrsankarganesh MECH-HICETNo ratings yet
- Consider The Reaction H2 (G) + 12 O2 (G) H2O (L) Delta H - 285.84 KJmol. How Many Grams of Hydrogen Gas Are Needed To ProduceDocument1 pageConsider The Reaction H2 (G) + 12 O2 (G) H2O (L) Delta H - 285.84 KJmol. How Many Grams of Hydrogen Gas Are Needed To ProduceAntonio Hernando MañeruNo ratings yet
- Energy BalanceDocument28 pagesEnergy BalanceMuhammad Zubair SharifNo ratings yet
- Asme PTC 2 - 2001Document40 pagesAsme PTC 2 - 2001bayuNo ratings yet
- Me 6301 Good QPDocument13 pagesMe 6301 Good QPMohanraj SubramaniNo ratings yet
- Thermodynamics Definition, Formulas, Laws and Equations - PhysicsDocument7 pagesThermodynamics Definition, Formulas, Laws and Equations - PhysicsRamaKrishnanGNo ratings yet
- Steam TablesDocument16 pagesSteam TablesVishal ShuklaNo ratings yet