Professional Documents
Culture Documents
Exam Exercises
Uploaded by
Everton CollingCopyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Exam Exercises
Uploaded by
Everton CollingCopyright:
Available Formats
D
Process Dynamics and
Operations Group
Dynamic Simulation
Task 1) Flowsheet decomposition
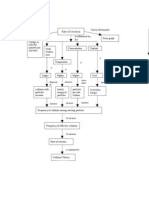
The flow sheet above shows the process to be modeled.
The educts A and B are fed into the tanks B1 and B2 via the two feed streams. Feed
stream 1 contains only raw material A, feed stream 2 raw material B. In the semi-batchreactor C1 they react to the products C and D. There is an internal heat exchanger
(radiator coil) in the reactor to control the reaction and an external heat exchanger W1
to cool down the products before starting the purification. In the following process step
the mixture is separated by a chromatography column K1. Tank B3 contains the
solvent. The purified components are collected in the product tanks B5 to B7.
Make a system analysis to determine the system border and divide the process into
subsystems for a following process simulation. Consider only the liquid phase and liquid
streams in the process!
Process Dynamics and
Operations Group
Task 2) A mechanical system.
A mechanical system is shown in the figure below. It consists of a pendulum having
mass m hung to a moving mass body M which is further attached to a spring and a
dashpot. An input force u is applied to mass M. is the angular displacement of the
pendulum and x1 is the displacement of the mass M when some input force is applied.
k1 and b1 are the spring and dashpot constants respectively. The pendulum string has a
length l. Let g be the gravitational acceleration acting on them. The contact forces
(equal and opposite) between the pendulum and the mass are also shown in the figure.
Following are the assumptions:
(i) The mass of the spring and the dashpot can be neglected.
(ii) There is no friction between the mass M and the surface.
(iii) There is no elongation of the pendulum string.
The simplified balance equations are given in state representation by
0
0
0
0
0
0
0
1
0
0
0
0
Parameter/Variable
g
k1
b1
l
M
m
u
Process Dynamics and
Operations Group
Value
9.81 m/s
0.4 N/m
0.2 N/(sm)
0.1 m
3 kg
1 kg
5 N
Implement the system in gPROMS and simulate the system for 10 seconds where the
reporting interval is set to 0.1 seconds and all states start at zero. What is final value of
state 2?
Task 3) Polymerization of styrol
In this task the polymerization of styrol to polystyrol is considered. The reaction is taking
place in a batch reactor which is equipped with a jacket (cf. figure 3.1 below). Further on
there exist a heat exchange with the environment.
Figure 3.1 : Batch-polymerization
Figure 3.2 : Exchange of the temperature
Here, only the reactions of polystyrol and the initiator are considered. The balance
equations are given by:
Process Dynamics and
Operations Group
Concentration initiator:
exp
Concentration styrol:
exp
Heat balance reactor temperature:
Heat balance jacket:
a) Make three models with the corresponding interfaces, where the models are for
the
Jacket
Reactor
Environment
Consider that the model for the environment consists only of the outlet of the
temperature.
b) Add a connection type where the only size to be exchanged is the temperature
(cf. figure 3.2).
c) Add a further model and connect the three models.
d) Write the process with the settings given in the table below and simulate the
process for two hours. Copy the curves for the concentrations here.
e) Write a schedule for the following problem.
The process start at 20C and the reactor contains only small traces of the
initiator and an initial concentration of styrol (cS(t=0) = 0.004 mol/m). At first the
reactor is heated up to 85C before the mass flow of the cooling medium is
Process Dynamics and
Operations Group
N
Y
D
switched on (
80 kg/s) and the initiator is given to the medium (cI(t=ti) = 1
mol/m). Then, the polymerization takes place for 2 hours.
Parameter/
Variable
gPROMS
notation
c_PT
A_TU
A_TR
k_TU
k_TR
k_RU
c_PR
A_RU
E_I
E_s
k_I
k_s
R
DeltaH_R
V_R
m_T
T_in
dotM_J
m_R
T_U
T_J
0
T_R
Description
Value
Specific heat capacity
Area between jacket and
environment
Area between jacket and reactor
Heat transfer coefficient between
reactor and environment
Heat transfer coefficient between
jacket and reactor
Heat transfer coefficient between
reactor and environment
2500 J/(Kkg)
10.5 m
Area between reactor and
environment
Activation energy initiator
Activation energy styrole
Reaction constant initiator
Reaction constant styrol
Gas constant
Reaction enthalpy
Reactor volume
Mass Jacket
Inlet temperature jacket
Mass flow jacket
Mass reactor
Environment temperature
Jacket temperature
Initial jacket temperature
Reactor temperature
800 W/(Km)
50 W/(Km)
3550 J/(Kkg)
1.2 m
135103 J/mol
95560 J/mol
4.281011
21010
8-3145 J/(Kmol)
-67.7103
3 m
800 kg
85 C
80 kg/s
3000 kg
20 C
20 C
20 C
c_I
Initiator concentration
1 mol/m
c_S
10 m
50 W/(Km)
Styrol concentration
0.004 mol/m
Process Dynamics and
Operations Group
Task 4) Simulation of adsorption.
The exhaust air of a biological process is slightly polluted with an organic solvent. This
solvent is removed with unsteady bed adsorption by means of activated carbon.
a) Implement a model of a bed adsorber in gPROMS
The model of a bed adsorber consists of mass balance for the stationary phase (bed of
activated carbon) and the mobile phase (exhaust air) as well as an adsorption of the
Langmuir type.
Intrinsic velocity: u int
4V
d 2
Mass balance for mobile phase:
Y
Y
2Y
1 6
Y YP
u int
Dax 2 k eff
t
z
dP
z
Mass balance for stationary phase:
6
G
Y YP , G (1 P ) X P P YP
k eff
t
dP
Adsorption isotherme (Langmuir): X P
cYP
1 cYP
Notes:
As the adsorption isotherm parameter c is different for air and steam, it should be
declared as a variable.
Enhance your model by the inlet and outlet ports for the gas stream by using the
available connection type. Keep in mind that additional port variables require
additional model equations.
The system is bounded on one side: the mobile phase load for z = 0 must be set
by the inlet ports.
Comment your solution.
b) Create a process enabling you to simulate two adsorption-desorption-cycles of the
adsorber.
At the beginning of the simulation the load of the mobile phase Y and the surface load
of the stationary phase XP are zero. As the polluting solvent strongly interacts with the
activated carbon, it is nearly completely adsorbed in the top layer of a new bed. In the
course of time the adsorption capacity of the top layer is depleted and the solvent
Process Dynamics and
Operations Group
N
Y
D
penetrates farther into the activated carbon bed, thus causing a moving adsorption
zone. As soon as that zone reaches the adsorber bottom, the solvent load at the outlet
increases remarkably and the adsorber must be regenerated. For this purpose it is
disconnected from exhaust air and flooded by steam ( V 1.5m 3 s 1 , Y 0kg m 3 ) until
virtually complete adsorption can be stated by observing a very low solvent load of the
steam at the adsorber outlet. After drying the adsorber with air it can be reconnected to
exhaust air.
Design a schedule such that adsorption stops as soon as the outlet load exceeds Y =
0.0002 and desorption with steam continues until the outlet load falls below Y = 0.0001
again. The duration of a complete cycle (including drying) is exactly 800 seconds.
Notes:
No flowsheeting is required for this task!
Discretization method: Axial := [CDFM, 2, 150]
c) Implement a flowsheet.
Adsorption shall now be operated continuously. For that
purpose two serial adsorbers (L = 3 m, the rest of
dimensions is the same as in a) and b)) are employed
instead of one. As soon as the adsorption zone leaves the
first column and enters the second one, the first column is
regenerated and dried and then connected to the outlet of
the second column. As soon as the adsorption zone moves
out of the second adsorber into the (originally) first one, the
second adsorber is regenerated and so on.
Implement a flowsheet of this column setup. Write at first
the corresponding models for the splitter and the mixer.
Consider that you have either two or three ports (cf. figure
right). The equations for the splitter are given by,
Vout ,i
Vin
, i 1, , NoOutlet ,
NoOutlet
c out ,i cin , i 1, , NoOutlet .
Where NoOutlet is the number of outlet streams which
corresponds to the number of ports.
Process Dynamics and
Operations Group
The models for the mixer are given by,
Vout
NoInlett
V
i 1
coutVout
in ,i
NoInlet
c
i 1
in ,i
Vin ,i .
Where NoInlet is the number of inlet streams corresponding to the number of ports.
Simulate then the process for 2000 seconds.
Notes:
Use the same notation given in the figure.
Comment your solution.
Symbol
c
Meaning
Langmuir coefficient of the adsorption isotherm
d
dP
Dax
P
G(z,t)
keff
L
Diameter of the bed adsorber
Particle diameter
Axial dispersion coefficient
Void fraction of the bed
Porosity of the particles
Total load of the activated carbon
Effective mass transfer coefficient
Length of the bed adsorber
Ludolph number
Volume flow
Value
1000 (Adsorption)
100 (Desorption)
0.0015 m
0.002 m2/s
0.4
0.6
0.01 m/s
6m
3.1416
1 (Adsorption)
1.5 (Desorption)
You might also like
- Igcc UnitsDocument40 pagesIgcc UnitsEverton CollingNo ratings yet
- 12 - Sampled Data ControlDocument16 pages12 - Sampled Data ControlEverton CollingNo ratings yet
- EMSO: A New Environment For Modelling, Simulation and OptimisationDocument6 pagesEMSO: A New Environment For Modelling, Simulation and OptimisationEverton CollingNo ratings yet
- 13 - Digital Controller DesignDocument22 pages13 - Digital Controller DesignEverton CollingNo ratings yet
- Binary and Ternary Mixture of Polar CompoundsDocument6 pagesBinary and Ternary Mixture of Polar CompoundsEverton CollingNo ratings yet
- OPT MatlabDocument19 pagesOPT MatlabPraveen KrsNo ratings yet
- 1.1 Questions: D N Y DD NN YYDocument4 pages1.1 Questions: D N Y DD NN YYEverton CollingNo ratings yet
- Lecture 3 - Flowsheeting in GPROMSDocument18 pagesLecture 3 - Flowsheeting in GPROMSEverton CollingNo ratings yet
- Lecture 5 - PDE's in GROMSDocument25 pagesLecture 5 - PDE's in GROMSEverton CollingNo ratings yet
- 1.1. Questions: D N Y DD NN YYDocument2 pages1.1. Questions: D N Y DD NN YYEverton CollingNo ratings yet
- Lecture 4 - Scheduling KP 110512bDocument16 pagesLecture 4 - Scheduling KP 110512bEverton CollingNo ratings yet
- Tratamento GLPDocument30 pagesTratamento GLPEverton CollingNo ratings yet
- Questions: D N Y DD NN YYDocument3 pagesQuestions: D N Y DD NN YYEverton CollingNo ratings yet
- PO SS2012 Tutorial00 AdditionalExerciseDocument2 pagesPO SS2012 Tutorial00 AdditionalExerciseEverton CollingNo ratings yet
- Lecture1-PO SS2011 02 ScalarOptimization p8Document8 pagesLecture1-PO SS2011 02 ScalarOptimization p8Everton CollingNo ratings yet
- 4.3 Problems With Inequality Constraints: General FormDocument6 pages4.3 Problems With Inequality Constraints: General FormEverton CollingNo ratings yet
- PO SS2011 05.1 DynamicOptimization p11Document11 pagesPO SS2011 05.1 DynamicOptimization p11Everton CollingNo ratings yet
- Lecture4-PO SS2011 04.2 MultidimensionalOptimizationEqualityConstraint p5Document5 pagesLecture4-PO SS2011 04.2 MultidimensionalOptimizationEqualityConstraint p5Everton CollingNo ratings yet
- Lecture5-PO SS2011 05 EvolutionaryAlgorithm p51Document50 pagesLecture5-PO SS2011 05 EvolutionaryAlgorithm p51Everton CollingNo ratings yet
- OptimizationExample Finkler FinalDocument32 pagesOptimizationExample Finkler FinalEverton CollingNo ratings yet
- Heat Exchanger Performance PDFDocument18 pagesHeat Exchanger Performance PDFcynaiduNo ratings yet
- Lecture1-PO SS2011 01 Introduction p26Document26 pagesLecture1-PO SS2011 01 Introduction p26Everton CollingNo ratings yet
- Refinery BasicsDocument43 pagesRefinery BasicsSalim ChohanNo ratings yet
- Lecture3-PO SS2011 04.1 MultidimensionalOptimizationUnconstrained p9Document9 pagesLecture3-PO SS2011 04.1 MultidimensionalOptimizationUnconstrained p9Everton CollingNo ratings yet
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeFrom EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeRating: 4 out of 5 stars4/5 (5794)
- Shoe Dog: A Memoir by the Creator of NikeFrom EverandShoe Dog: A Memoir by the Creator of NikeRating: 4.5 out of 5 stars4.5/5 (537)
- The Yellow House: A Memoir (2019 National Book Award Winner)From EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Rating: 4 out of 5 stars4/5 (98)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceFrom EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceRating: 4 out of 5 stars4/5 (895)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersFrom EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersRating: 4.5 out of 5 stars4.5/5 (344)
- The Little Book of Hygge: Danish Secrets to Happy LivingFrom EverandThe Little Book of Hygge: Danish Secrets to Happy LivingRating: 3.5 out of 5 stars3.5/5 (399)
- Grit: The Power of Passion and PerseveranceFrom EverandGrit: The Power of Passion and PerseveranceRating: 4 out of 5 stars4/5 (588)
- The Emperor of All Maladies: A Biography of CancerFrom EverandThe Emperor of All Maladies: A Biography of CancerRating: 4.5 out of 5 stars4.5/5 (271)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaFrom EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaRating: 4.5 out of 5 stars4.5/5 (266)
- Never Split the Difference: Negotiating As If Your Life Depended On ItFrom EverandNever Split the Difference: Negotiating As If Your Life Depended On ItRating: 4.5 out of 5 stars4.5/5 (838)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryFrom EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryRating: 3.5 out of 5 stars3.5/5 (231)
- On Fire: The (Burning) Case for a Green New DealFrom EverandOn Fire: The (Burning) Case for a Green New DealRating: 4 out of 5 stars4/5 (73)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureFrom EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureRating: 4.5 out of 5 stars4.5/5 (474)
- Team of Rivals: The Political Genius of Abraham LincolnFrom EverandTeam of Rivals: The Political Genius of Abraham LincolnRating: 4.5 out of 5 stars4.5/5 (234)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyFrom EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyRating: 3.5 out of 5 stars3.5/5 (2259)
- The Unwinding: An Inner History of the New AmericaFrom EverandThe Unwinding: An Inner History of the New AmericaRating: 4 out of 5 stars4/5 (45)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreFrom EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreRating: 4 out of 5 stars4/5 (1090)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)From EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Rating: 4.5 out of 5 stars4.5/5 (120)
- Her Body and Other Parties: StoriesFrom EverandHer Body and Other Parties: StoriesRating: 4 out of 5 stars4/5 (821)
- Weld StrengthDocument6 pagesWeld StrengthAqil GhaffarNo ratings yet
- All-Dielectric Self Supporting (ADSS) Aerial Loose Tube Fiber Optic CableDocument3 pagesAll-Dielectric Self Supporting (ADSS) Aerial Loose Tube Fiber Optic CableAhmad TaheriNo ratings yet
- 12H, 140H and 160H Electrical System Motor Graders (NA Version)Document2 pages12H, 140H and 160H Electrical System Motor Graders (NA Version)jorge william ramirezNo ratings yet
- Duct Work PDFDocument2 pagesDuct Work PDFRiza BahrullohNo ratings yet
- LAB REport 7Document6 pagesLAB REport 7Misbah TehseenNo ratings yet
- Electromagnetic Plunger With Stopper DynamicsDocument28 pagesElectromagnetic Plunger With Stopper DynamicsCatanescu Alexandru-LaurentiuNo ratings yet
- Midterm Study GuideDocument7 pagesMidterm Study GuidejohntothepNo ratings yet
- XFOIL: An Analysis and Design System For Low Reynolds Number AirfoilsDocument2 pagesXFOIL: An Analysis and Design System For Low Reynolds Number Airfoilsmunawar mahtabNo ratings yet
- HDPEDocument14 pagesHDPESandy YansikuNo ratings yet
- Beep 2 CdevDocument1,241 pagesBeep 2 CdevIzio SilvaNo ratings yet
- Environmental Science 7 3 QuarterDocument5 pagesEnvironmental Science 7 3 QuarterMichael Angelo ConuiNo ratings yet
- Concept MapDocument1 pageConcept MapMonis Diana Abu BakarNo ratings yet
- DataLog Drilling Fluids ManualDocument57 pagesDataLog Drilling Fluids ManualJohnSmithNo ratings yet
- A Study On The Peculiar Problems of Cold Formed Steel DesignDocument3 pagesA Study On The Peculiar Problems of Cold Formed Steel DesignarjunNo ratings yet
- Trajectory Analysis and Staging Trades For Smaller Mars Ascent VehiclesDocument8 pagesTrajectory Analysis and Staging Trades For Smaller Mars Ascent VehiclesJuanJoNo ratings yet
- 02 Whole X XXXXXXXXXXXXXX 80Document109 pages02 Whole X XXXXXXXXXXXXXX 80Danem HalasNo ratings yet
- MONTHLY TEST Unit 2 Earth and SpaceDocument2 pagesMONTHLY TEST Unit 2 Earth and SpaceRowena Sta MariaNo ratings yet
- Defaults Wave SolderingDocument8 pagesDefaults Wave SolderingTANNo ratings yet
- Advanced Inspection MethodsDocument38 pagesAdvanced Inspection MethodsBenNo ratings yet
- Mine VentilationDocument37 pagesMine VentilationKemal Cengiz100% (3)
- Mate1000 Sln21aDocument3 pagesMate1000 Sln21aclearcastingNo ratings yet
- Grade 10 March 2024 Term 1 Physical Sciences TestDocument11 pagesGrade 10 March 2024 Term 1 Physical Sciences TestTererai Lalelani Masikati HoveNo ratings yet
- DiffusionDocument4 pagesDiffusionROBERTO SHERWIN JR TANDASNo ratings yet
- Internal Combustion EngineDocument40 pagesInternal Combustion EngineMingNo ratings yet
- KinematcsDocument6 pagesKinematcsAdit Kumar100% (1)
- 2015-10-23 - High PC - 2015 - Fahim - 03Document28 pages2015-10-23 - High PC - 2015 - Fahim - 03jonarmijosNo ratings yet
- Positron Emission Tomography (Pet)Document15 pagesPositron Emission Tomography (Pet)ManojNo ratings yet
- Technical Whitepaper: Sonic V - Acoustic Pulse Reflectometry (APR) Inspection SystemDocument11 pagesTechnical Whitepaper: Sonic V - Acoustic Pulse Reflectometry (APR) Inspection SystemassurendranNo ratings yet
- Flowmeter Model MP KytolaDocument2 pagesFlowmeter Model MP KytolaJimmy MirandaNo ratings yet