Professional Documents
Culture Documents
NEBOSH International Diploma: Sample Material
Uploaded by
try123testOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
NEBOSH International Diploma: Sample Material
Uploaded by
try123testCopyright:
Available Formats
NEBOSH International Diploma
Unit IA - Managing Health and Safety
Unit IB - Hazardous Substances and Agents
Unit IC - Workplace and Work Equipment Safety
SAMPLE MATERIAL
RMS Publishing Ltd
Suite 3, Victoria House,
Lower High Street, Stourbridge, West Midlands DY8 1TA
Tel: +44 (0) 1384 447927 Email: sales@rmspublishing.co.uk
ELEMENT IA1 - PRINCIPLES OF HEALTH AND SAFETY MANAGEMENT
Figure IA1-6: Estimates of breakdown of costs to society of workplace injury and ill-health 2013/14. Source: UK, HSE Cost to Britain model.
The UK HSE also provided data in 2013/14 on the estimated costs of different types of case.
Non-financial human
cost
Financial cost
Total cost per case
Fatal injuries
1,153,000
421,600
1,575,000
Non-fatal injuries
4,600
2,900
7,500
Ill-health
9,900
8,700
18,700
Figure IA1-7: Estimates of costs to society of different cases of workplace injury and ill-health 2013/14.
Source: UK, HSE Cost to Britain model.
INSURED AND UNINSURED COSTS
Many case studies over recent years have illustrated the difference between insured costs and uninsured costs.
It has been shown in the case studies that uninsured costs were often between 8 and 36 times greater than the
insured costs.
INSURED COSTS
Employee/third party compensation
Damage to buildings and plant
Damage to vehicles, equipment and tools
Medical costs
Legal costs of compensation and prosecution
UNINSURED COSTS
8-36
Figure IA1-8: Main insured and uninsured costs, typical ratio of costs.
Product and materials damage
Emergency supplies and first-aid
Clean up costs
Delays and weakened morale
Loss of experience and expertise
Overtime and temporary labour
Investigation time
Supervisors and managers time diverted
Increase in insurance premium
Enforcement agency and court fines
Effects on goodwill and reputation
Source: RMS/UK, HSE HSG96 (no longer available).
Costs relating to injury and ill-health may also be classified on the basis of whether they are direct costs or
indirect costs.
Direct costs
Direct costs include:
Lost time of injured worker and any continued payments to worker or family.
Damage to equipment, tools, property, plant or materials.
Medical or first-aid costs.
Time and materials to clean up after the accident.
Insurance, indemnity or compensation payments.
Court costs.
Fines.
8
RMS
ELEMENT IA9 - THE ROLE OF THE HEALTH AND SAFETY PRACTITIONER
Figure IA9-3: The distinction between leadership and management.
Source: RMS.
The need to adopt to different management styles dependent on any given
situation
Many managers assume that leadership style is a function of their personality rather than a strategic decision
they can make. The manager should be able to choose the style that has most effect in the situation presented.
There are many different models of management styles. However, Daniel Goleman measured the impact of 6
different styles of management and leadership.
The six different styles of leadership following Goleman (2000) are:
Coercive: relatively aggressive style that demands compliance, the style works best in crisis situations i.e.
immediately following an accident, where decisive management is required.
Authoritative: it is considered to be the most effective motivating style of leadership that drives people to
improve performance. Works best when a new vision is required or a new safe system of work developed.
Affiliative: a style that focuses on people, it values employees and their emotions, rather than their
objectives. Works best when the manager is aware the team is operating in stressful situations, such as
the aftermath of a fire.
Democratic: a democratic leader builds trust and respect by listening to every members opinions. The
style works best when the manager wants everyone in the team to contribute i.e. risk profiling of an
organisation.
Pacesetting: a leader sets high standards and wants the goal to be reached in an excellent and fast way.
Works best with highly motivated, competent teams such as engineers when reviewing a safe system of
work in the oil processing industry.
Coaching: a coaching leader helps employees to identify their strengths and weaknesses. Works best
when the manager is improving the performance of the team i.e. compliance with a standard or convention.
Development, implementation, maintenance and evaluation of health and
safety management systems
ROLE (AND FUNCTION) OF HEALTH AND SAFETY PRACTITIONERS
The role of the health and safety practitioner is to support line management in meeting their responsibilities by:
Providing information and advice.
Supporting line management with the coordination of health and safety effort.
Monitoring the effectiveness of actions to meet responsibilities.
The functions of the health and safety practitioner include activities to:
Identify problems (including hazards).
Assess the need for action.
Assist with the assessment of risks.
Design and develop strategies and plans.
Advise on relevant current best practice for prevention.
Present themselves and their advice in an independent manner.
Promote and communicate health, safety and welfare advances and practices.
Assist with the implementation of these strategies and plans.
Evaluate their effectiveness.
Maintain records relating to health and safety performance.
312
RMS
THE CONTROL OF HAZARDOUS SUBSTANCES - ELEMENT IB3
For efficient operation, exhaust ventilation should be located as close as possible to the source of dust, by the
use of captor hoods, booths or enclosures. Local exhaust systems should be designed to collect and remove
all dust-laden air. Openings in the enclosures should be as small as possible, while still allowing access to the
necessary work operation.
In the case of captor hoods and booths, the ventilation equipment should be so constructed that air turbulence
and eddies, created by the work process or by the workers, do not prevent the effective removal of dust.
PERSONAL PROTECTION
Respiratory protective equipment (RPE)
The provision and use of respiratory protective equipment (RPE) should be regarded only as a temporary or
emergency measure and not as an alternative to technical control.
A sufficient and suitable supply of equipment should be available in the workplace.
In the UK, for example, it is a requirement of the law under the Control of Asbestos Regulations 2012, that
suitable RPE is provided by the employer which reduces asbestos exposure so far as is reasonably
practicable. Supporting this regulation is an Approved Code of Practice (ACOP) which recommend that
employers undertake respiratory face-piece fit testing for their workers.
Such equipment should be provided for all workers employed in any situation where levels of airborne
asbestos fibre exceed, or are liable to exceed, the exposure limits.
Workers should be informed when concentrations of airborne asbestos fibre reach such levels.
When workers have been so informed, they should use the equipment provided.
Workers required to wear protective equipment should be fully instructed in its use.
Employers should provide supervision, to ensure that the equipment is properly used.
All respiratory equipment should be provided and maintained by the employer without cost to the worker.
Face piece fit testing is a method for ensuring that respiratory protective equipment (RPE) supplied by the
employer provides an adequate seal that prevents exposure and takes into account facial features.
The fit test will ensure that only correctly fitted RPE will be used to prevent exposure.
Figure IB3-6: Some of the frequent problems with fitting and possible solutions for filtering facepiece respirators.
Source: UK, HSE.
A fit test is not a substitute for correct and careful day-to-day fitting of the face piece. The latter should always
include a pre-use fit check.
A repeat fit test should be conducted in the following circumstances:
1) Where the wearer:
a) Loses or gains weight.
b) Undergoes any substantial dental work.
c) Develops any facial changes (scars, moles, etc.) around the face seal area.
2) If the employers health and safety policy requires it.
RMS
57
RADIATION - ELEMENT IB7
IB7.1 - The nature and different types of ionising and non-ionising
radiation
The distinction between ionising and non-ionising radiation
The part of the electromagnetic spectrum with greatest energy is where the gamma rays and the X-rays are.
They are so powerful that they are capable of removing the electron from an atom of material they encounter, a
process that is called ionisation.
These powerful rays are known as ionising radiation. The radiation from the lower energy part of the
electromagnetic spectrum, ultraviolet (UV), visible light, infrared (IR), microwaves and radio waves, do not
contain enough energy to ionise and are therefore known as non-ionising radiation. Radiation can therefore be
classified in terms of its energy into two types: ionising and non-ionising.
There is also a type of ionising radiation that is not
electromagnetic waves, but is particulate. The most
common particles of ionising radiation are known as
alpha (), beta () and neutron radiation.
Note: Other types of radiation exist, such as positrons
(+), thermal neutrons, fast neutrons and protons, but
are beyond the scope of this Element.
Ionising radiation can be defined as radiation, typically
alpha and beta particles, and gamma and X-rays,
which has sufficient energy to produce ions by
interacting with matter. Non-ionising radiation can be
defined as radiation that does not possess sufficient
energy to cause the ionisation of matter.
Figure IB7-1: Ionisation.
Source: RMS.
Ionising radiation has sufficient energy to dislodge electrons from atoms thus changing the chemical properties
of the substance it interacts with. Ionising radiation includes alpha particles, beta particles, gamma rays, X-rays
and cosmic rays.
Figure IB7-2: Definition of ionising radiation.
Source: London Health Observatory.
Non-ionising radiation (NIR) is the term given to the part of the electromagnetic spectrum where there is
insufficient quantum energy to cause ionisations in living matter. It includes static and power frequency fields,
radiofrequencies, microwaves, infrared, visible and ultraviolet radiation.
Figure IB7-3: Definition of non-ionising radiation.
Source: Health Protection Agency.
The electromagnetic spectrum
BASIC CONCEPTS - WAVELENGTH, ENERGY AND FREQUENCY
Radiation is a form of energy that is emitted by a wide variety of sources. The types of radiation are grouped
and labelled according to the nature of the radiation and the amount of energy they have. One of these types of
energy is transmitted by waves called electromagnetic waves.
An electromagnetic wave travels, or propagates, in a
direction that is oriented at right angles to the
oscillations of both the electric (E) and magnetic (B)
fields, transporting energy away from the radiation
source.
The two oscillating energy fields are mutually
perpendicular and act in phase, as a sine wave.
Electric and magnetic field vectors are not only
perpendicular to each other, but are also
perpendicular to the direction of wave propagation.
By convention, and to simplify illustrations, the vectors
representing the electric oscillating fields of
electromagnetic waves are often omitted.
The
resulting diagrams show a single sinusoidal
electromagnetic wave.
Figure IB7-4: Electromagnetic wave.
Source: Olympus.
The distance between electromagnetic wave peaks is the wavelength. The number of wave peaks passing a
given point in one second is the frequency. The distance between electromagnetic wave peaks is the
wavelength. The number of wave peaks passing a given point in one second is the frequency.
RMS
181
ELEMENT IC2 - FIRE AND EXPLOSION
RELATIVE DENSITY
Relative density is the ratio of the specific density of a substance to the specific density of a standard
substance at specified conditions. For liquids and solids, the standard is usually water at 4C or some other
specified temperature. For vapours and gases, the standard is often air at the same temperature and pressure
as the substance.
For fire safety and fire protection reasons we are particularly concerned with the comparison of a flammable
material and its vapours in relation to air. The vapour density of air at standard temperature and pressure is
taken as 1 with a molecular weight of 28.97 grams per mole. All other gases are compared to air by dividing
their molecular weights. We can therefore see that the relative density for methane when compared to air is:
16.04
28.97
0.55 (a relative density less than 1, which makes it lighter than air).
Since both units are in g/mol, relative density (the ratio of the two units) has no units; it is a pure number. The
unit is used as a single comparator to determine the initial behaviour of a gas when released into the air at the
same temperature, i.e. will it rise or fall.
As can be seen, the difference between the UK vapour density and relative density is significant. It is important
to ensure that the relative density of a vapour is used when considering its hazards and the necessary safety
measures.
Density effects - gases and vapours
Few materials have a molecular weight less than air (molecular weight of approximately 29), so under normal
conditions most gases and vapours are heavier than air. Hydrogen (H2) (molecular weight of approximately 2),
methane (CH4) (molecular weight of approximately 16) and acetylene gas (C2H2) (molecular weight of
approximately 26) are exceptions. The higher the molecular weight of a gas/vapour, the greater its (specific)
density at constant temperature.
In order to establish a reference point, air is given a specific density of 1. This enables a relative density to be
established for other substances and to predict the effect of them being placed together. Flammable or
explosive gas/vapours heavier than air (relative density greater than 1), such as propane (which has a relative
density of 1.52 when compared to air), can spread and accumulate at a low level, for example, in pits, sumps or
drains. Also, vapours less dense than air at ambient temperature may spread at low level when they are cold,
because the specific density of the substance when cold could be greater than the surrounding warmer air.
Gas or vapour
Molecular weight
Relative density (air)
Air
28.97
Acetylene, C2H2
26.04
0.90
Cyclohexane, C6H12
84.16
2.91
Ethane, C2H6
30.07
1.04
Hydrogen, H
1.01
0.03
Methane, CH4
16.04
0.55
Propane, C3H8
44.10
1.52
Figure IC2-1: Densities of flammable gases/vapours.
Source: RMS/Multiple.
Accumulations of gas/vapour less dense than air (relative density of less than 1), for example, hydrogen and
methane, can occur at high points in poorly ventilated buildings. Consideration needs to be given to work
activities that represent a source of ignition when they take place where gas accumulation might occur in this
way, for example, maintenance work at a height.
The relative density of a flammable vapour or gas with air is important when considering local exhaust
ventilation (LEV) requirements provided to prevent a build-up of the vapour or gas; it will indicate whether LEV
is required at high or low level, or both.
Case Study
Computer utilities require batteries as a source of power, A hydrogen explosion occurred in an Uninterruptible
Power Source (UPS) battery room of a data processing centre. The explosion blew a 400 ft2 hole in the roof,
collapsed numerous walls and significantly damaged a large portion of the 50,000 ft2 building. Fortunately, the
computer/data centre was vacant at the time and there were no injuries. The explosion resulted from the lack
of hydrogen gas (produced when lead acid batteries are charged) detection and high level ventilation
equipment.
36
RMS
ELEMENT IC10 - WORKPLACE TRANSPORT AND MANAGING WORK-RELATED ROAD RISK
Figure IC10-13: Barriers and markings.
Source: RMS.
Figure IC10-14: Visual warning on a dumper truck.
Source: RMS.
Warnings of vehicle approach and reversing
Warnings may be audible or visual or a combination of each. They are used to warn that the vehicle is
operating in the area, such as a flashing light on the top of a fork-lift truck or to warn of a specific movement,
such as an audible warning that a large vehicle is reversing. These are designed to alert people in the area in
order that they can place themselves in a position of safety. They do not provide the driver with authority to
reverse the vehicle or to proceed in a work area without caution.
Control measures for reversing vehicles within the workplace include:
Avoiding the need for vehicles to reverse (by the use of one-way and drive-through systems or turning
circles).
Separation of vehicles and pedestrians as much as is practicable.
Signs that warn of the hazard.
Visual and audible warning systems fitted to vehicles.
Adequate mirrors/vehicle cameras that are kept clean.
Space to allow good visibility.
Adequate lighting minimising sudden changes in luminance.
Procedural measures, such as the use of trained signaller/banksman/reversing assistant.
Refuges to protect signaller/banksman/reversing assistant.
Appropriate site rules adequately enforced.
Site rules
It is important to establish clear and well understood site rules regarding vehicle operations. These may have
to be communicated to drivers by security staff at the time they visit the site. They would often stipulate where
the driver should be whilst vehicles are being loaded, where the keys to the vehicle should be, not reversing
without permission and what access they have to areas of the site for such things as refreshment. Pedestrians
should also know what the site rules are in order to keep themselves safe.
Site rules for pedestrians might include such things as using pedestrian exits/entrances or crossing points, not
entering hazardous areas or the need to wear personal protective equipment in hazardous areas, and not
walking behind a reversing vehicle. Where sites are made up of or join a highway, such as carriageway repairs
on a motorway, it is important to identify who has priority - the vehicle or the pedestrian. Site rules may clarify
that once the site boundary is crossed, the pedestrian has priority - this has to be clear. The site rules may be
re-enforced by the provision of additional signs to clarify a speed limit and the nature of the priority of workers.
Activity
Workplace transport activities that present a risk include:
Reversing up to structures or edges.
Banksmen (signallers).
Parking in car parks, hard standings or on a slope.
Coupling and uncoupling.
Access to fifth wheel area.
Loading and unloading.
Loading bays including dock levellers and preventing vehicles moving.
Dealing with shifted loads.
Work at height on vehicles platforms and gantries.
Trimming, sheeting and netting.
Many deaths and serious injuries involving vehicles at work happen during reversing, with poor visibility being
the main cause. There are several measures that can help to reduce the risk of reversing accidents, but
removing the need for reversing is the most effective.
334
RMS
You might also like
- Oil and Gas Safety Officer InterviewDocument91 pagesOil and Gas Safety Officer InterviewAhmed IsahNo ratings yet
- FES-Electrical Safety Module-2Document73 pagesFES-Electrical Safety Module-2bachrul ulumNo ratings yet
- Vital chain block specifications and lifting equipment guideDocument2 pagesVital chain block specifications and lifting equipment guideAthox QhurienNo ratings yet
- Safe Use of Cranes, Hoists & SlingsDocument79 pagesSafe Use of Cranes, Hoists & SlingsAhmed GaballaNo ratings yet
- Hand and Power Tools SafetyDocument3 pagesHand and Power Tools SafetyFairus Muhamad DarusNo ratings yet
- Challenger Chain Blocks - Instruction Manual PDFDocument22 pagesChallenger Chain Blocks - Instruction Manual PDFAbdul Rehman SheikhNo ratings yet
- Power and Machine Tools SectionDocument100 pagesPower and Machine Tools SectionSantiago UrgilesNo ratings yet
- Hand and Portable Power Tool Safety TipsDocument20 pagesHand and Portable Power Tool Safety TipsBrijgopal YadavNo ratings yet
- Ergonomics and SafetyDocument49 pagesErgonomics and SafetyJulianMcOlenNo ratings yet
- ErgonomicsDocument35 pagesErgonomicsPratik100% (1)
- Asssessing Ergonomic HazardDocument67 pagesAsssessing Ergonomic HazardYenni SuryansyahNo ratings yet
- Click Here To Download Nebosh Igc Course Details (Nebosh) PDFDocument2 pagesClick Here To Download Nebosh Igc Course Details (Nebosh) PDFWahyu Sunday75% (4)
- British Glass - Incident and Near Miss Reporting Guidance (Jan 2015)Document14 pagesBritish Glass - Incident and Near Miss Reporting Guidance (Jan 2015)knabpshoNo ratings yet
- Biomechanics of Work: Yansen Theopilus, S.T., M.TDocument68 pagesBiomechanics of Work: Yansen Theopilus, S.T., M.TuytyuytryuNo ratings yet
- Standard For Old Age PDFDocument65 pagesStandard For Old Age PDFRendani VeleNo ratings yet
- Portable Equp MNT GuideDocument0 pagesPortable Equp MNT GuideashawishNo ratings yet
- CHAIN FAILDocument2 pagesCHAIN FAILRusihan RehanNo ratings yet
- Prepared by A.Prasad RoshanDocument102 pagesPrepared by A.Prasad Roshansaswati roy barmanNo ratings yet
- 101 SafetyTalk Topics ZetekyDocument21 pages101 SafetyTalk Topics ZetekyMu'az ObadakiNo ratings yet
- Behavior Safety (Perilaku Keselamatan) : By: Rosdja PurnamaDocument14 pagesBehavior Safety (Perilaku Keselamatan) : By: Rosdja PurnamaMerah PastelNo ratings yet
- Gapura Company Profile - 17mar17Document43 pagesGapura Company Profile - 17mar17als izmiNo ratings yet
- Accident Prevention & Investigation Course OverviewDocument33 pagesAccident Prevention & Investigation Course OverviewarunNo ratings yet
- Examiners Reports NGC2 Only September 2011 NGC2Document11 pagesExaminers Reports NGC2 Only September 2011 NGC2dwayjayNo ratings yet
- Health and Safety Training: What You Need To KnowDocument6 pagesHealth and Safety Training: What You Need To KnowFrank MacauNo ratings yet
- Behavior Theory ABC Model by Thomas KrausDocument28 pagesBehavior Theory ABC Model by Thomas KrausChusnadiNo ratings yet
- Basic Principles in Occupational Hygiene: Evah MainaDocument35 pagesBasic Principles in Occupational Hygiene: Evah MainaMokua FedinardNo ratings yet
- Chain Block Inspection Checklist for SDHO ProjectDocument1 pageChain Block Inspection Checklist for SDHO ProjectIhsan yaqoobNo ratings yet
- 10 5 2013-16-23 Grad HSE Job DescriptionDocument3 pages10 5 2013-16-23 Grad HSE Job Descriptionemeka2012No ratings yet
- Accident Investigation SampleDocument32 pagesAccident Investigation SampleVela DanialNo ratings yet
- CNC Machine Job Safety AnalysisDocument1 pageCNC Machine Job Safety AnalysisMubarak RahmanNo ratings yet
- Kent Uni health and safety inductionDocument20 pagesKent Uni health and safety inductionMijanur RahmanNo ratings yet
- Expert Trainers Academy - Hse Interview Questions V1Document14 pagesExpert Trainers Academy - Hse Interview Questions V1greewin88No ratings yet
- TUV - COSH Hand & Power ToolsDocument94 pagesTUV - COSH Hand & Power ToolsjeromeNo ratings yet
- Introduction to Occupational Health and SafetyDocument13 pagesIntroduction to Occupational Health and Safetyangeh morilloNo ratings yet
- Behavior Based Safety: Gary Peacock Safety Consultant Ohio BWCDocument103 pagesBehavior Based Safety: Gary Peacock Safety Consultant Ohio BWCAkash Rs100% (1)
- Understanding the Hidden Costs of Workplace AccidentsDocument12 pagesUnderstanding the Hidden Costs of Workplace Accidentsdhavalesh1No ratings yet
- Hsg245 Investigating Accidents&IncidentsDocument89 pagesHsg245 Investigating Accidents&IncidentsVan SotNo ratings yet
- Investigasi Kecelakaan Kerja - R1Document25 pagesInvestigasi Kecelakaan Kerja - R1manggungNo ratings yet
- Hazard Identification, Risk Assessment and Risk ControlDocument4 pagesHazard Identification, Risk Assessment and Risk ControlNarayanasami Kannan0% (1)
- Hidden Costs of AccidentsDocument10 pagesHidden Costs of AccidentsNaga RajanNo ratings yet
- Introduction to human factors: understanding the job, individual and organizationDocument2 pagesIntroduction to human factors: understanding the job, individual and organizationAbu Aalif RayyanNo ratings yet
- hsg245 PDFDocument88 pageshsg245 PDFYousef OlabiNo ratings yet
- Form RULA - Office AssessmentDocument4 pagesForm RULA - Office AssessmentWisnuakbar NugrahaNo ratings yet
- Introductory Hazard Management (Bdph4103)Document4 pagesIntroductory Hazard Management (Bdph4103)anbu_331No ratings yet
- Occupational Safety and Health 1: Introduction To OSHADocument25 pagesOccupational Safety and Health 1: Introduction To OSHAnuris_85100% (2)
- IGC2 - QuestionsDocument27 pagesIGC2 - QuestionsAswin Magesh100% (3)
- Lab Safety Induction Manual 2014Document18 pagesLab Safety Induction Manual 2014Mustafa SaeedNo ratings yet
- NHK Spring Philippines Safety ManualDocument62 pagesNHK Spring Philippines Safety ManualJea RodegerioNo ratings yet
- Hazard Prevention Through Effective Safety and Health Training - Table - ContentsDocument1 pageHazard Prevention Through Effective Safety and Health Training - Table - ContentsAmeenudeen0% (1)
- SAFE DESIGN AND PPEDocument7 pagesSAFE DESIGN AND PPEAlfie Delos Reyes100% (1)
- Introduction To Health & Safety Management: Gitam UniversityDocument40 pagesIntroduction To Health & Safety Management: Gitam UniversitySavantNo ratings yet
- Formulation of Accident Prevention ProgrammeDocument24 pagesFormulation of Accident Prevention ProgrammeCHANDUNo ratings yet
- Elements of An Effective Safety and Health ProgramDocument20 pagesElements of An Effective Safety and Health ProgramJojo Betanio AkajoknoNo ratings yet
- Hand Safety PresentationDocument20 pagesHand Safety PresentationIsagani Vino100% (1)
- Dosecc Health ManualDocument62 pagesDosecc Health ManualRachmad HazanNo ratings yet
- Personal protective equipment Complete Self-Assessment GuideFrom EverandPersonal protective equipment Complete Self-Assessment GuideRating: 5 out of 5 stars5/5 (1)
- Engineering Health and SafetyDocument24 pagesEngineering Health and SafetyKobby BrineNo ratings yet
- Lab risk assessment guideDocument8 pagesLab risk assessment guideReni Swara Mahardika50% (2)
- Mis 021Document40 pagesMis 021Shahab AlamNo ratings yet
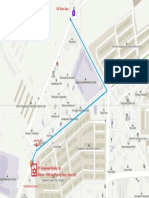
- Technip Parking Lot - MapDocument1 pageTechnip Parking Lot - Maptry123testNo ratings yet
- Upload: or Drag & Drop Supported File Types: PDF, TXT, Doc, PPT, XLS, Docx, and MoreDocument7 pagesUpload: or Drag & Drop Supported File Types: PDF, TXT, Doc, PPT, XLS, Docx, and Moretry123testNo ratings yet
- DD3 Schedule - Monday To FridayDocument1 pageDD3 Schedule - Monday To Fridaytry123testNo ratings yet
- Commuting Shuttle To Gyuldo-LD & Transporation in Yard - Saturday-SundayDocument2 pagesCommuting Shuttle To Gyuldo-LD & Transporation in Yard - Saturday-Sundaytry123testNo ratings yet
- Prelude Pisol CMO Gyuldo KE Quay Shuttle - Monday To Friday PDFDocument1 pagePrelude Pisol CMO Gyuldo KE Quay Shuttle - Monday To Friday PDFtry123testNo ratings yet
- Commuting Bus Stop Point Photo - Shuttle To Pisol - Gyuldo - LDDocument5 pagesCommuting Bus Stop Point Photo - Shuttle To Pisol - Gyuldo - LDtry123testNo ratings yet
- Rotators' Schedule - Shift - Bus New Stop Point - WVDocument1 pageRotators' Schedule - Shift - Bus New Stop Point - WVtry123testNo ratings yet
- Diploma Name InstructionsDocument7 pagesDiploma Name Instructionstry123testNo ratings yet
- Commuting Shuttle To Pisol Monday To FridayDocument2 pagesCommuting Shuttle To Pisol Monday To Fridaytry123testNo ratings yet
- Am Bank App FormDocument8 pagesAm Bank App Formtry123testNo ratings yet
- Commuting Shuttle Schedule To CMO - Pisol - Monday To FridayDocument2 pagesCommuting Shuttle Schedule To CMO - Pisol - Monday To Fridaytry123testNo ratings yet
- Rotators' Schedule PDFDocument1 pageRotators' Schedule PDFtry123testNo ratings yet
- NEBOSH International Diploma in Occupational Health and SafetyDocument3 pagesNEBOSH International Diploma in Occupational Health and Safetytry123testNo ratings yet
- Rotators' Schedule 20161024Document1 pageRotators' Schedule 20161024try123testNo ratings yet
- ReadmeaaaaaaaaaaaaDocument9 pagesReadmeaaaaaaaaaaaaChristian Marcos AvalosNo ratings yet
- Rotators' Schedule - Shift - Monday To SundayDocument1 pageRotators' Schedule - Shift - Monday To Sundaytry123testNo ratings yet
- NEBOSH International Diploma in Occupational Health and SafetyDocument3 pagesNEBOSH International Diploma in Occupational Health and Safetytry123testNo ratings yet
- Upload: or Drag & Drop Supported File Types: PDF, TXT, Doc, PPT, XLS, Docx, and MoreDocument7 pagesUpload: or Drag & Drop Supported File Types: PDF, TXT, Doc, PPT, XLS, Docx, and Moretry123testNo ratings yet
- Optimize Coverage and Performance with 12dB MTMA AmplifierDocument3 pagesOptimize Coverage and Performance with 12dB MTMA AmplifierpecqueurNo ratings yet
- Memento Mori: March/April 2020Document109 pagesMemento Mori: March/April 2020ICCFA StaffNo ratings yet
- How To Open and Convert An .SCM FileDocument5 pagesHow To Open and Convert An .SCM FilejackNo ratings yet
- Entrepreneurship and EconomicDocument2 pagesEntrepreneurship and EconomicSukruti BajajNo ratings yet
- City Gas Distribution ReportDocument22 pagesCity Gas Distribution Reportdimple1101100% (9)
- Frequency Meter by C Programming of AVR MicrocontrDocument3 pagesFrequency Meter by C Programming of AVR MicrocontrRajesh DhavaleNo ratings yet
- Etp ListDocument33 pagesEtp ListMohamed MostafaNo ratings yet
- Namal College Admissions FAQsDocument3 pagesNamal College Admissions FAQsSauban AhmedNo ratings yet
- Eship 1Document18 pagesEship 1Yash SoniNo ratings yet
- ASM Architecture ASM Disk Group AdministrationDocument135 pagesASM Architecture ASM Disk Group AdministrationVamsi ChowdaryNo ratings yet
- 13 Daftar PustakaDocument2 pages13 Daftar PustakaDjauhari NoorNo ratings yet
- Why Companies Choose Corporate Bonds Over Bank LoansDocument31 pagesWhy Companies Choose Corporate Bonds Over Bank Loansতোফায়েল আহমেদNo ratings yet
- Manual de Instalare Centrala de Incendiu Adresabila 1-4 Bucle Teletek IRIS PRO 250bucla 96 Zone 10000 EvenimenteDocument94 pagesManual de Instalare Centrala de Incendiu Adresabila 1-4 Bucle Teletek IRIS PRO 250bucla 96 Zone 10000 EvenimenteAlexandra DumitruNo ratings yet
- Siyaram S AR 18-19 With Notice CompressedDocument128 pagesSiyaram S AR 18-19 With Notice Compressedkhushboo rajputNo ratings yet
- Enhancing reliability of CRA piping welds with PAUTDocument10 pagesEnhancing reliability of CRA piping welds with PAUTMohsin IamNo ratings yet
- Kuliah Statistik Inferensial Ke4: Simple Linear RegressionDocument74 pagesKuliah Statistik Inferensial Ke4: Simple Linear Regressionvivian indrioktaNo ratings yet
- Coa - Ofx-8040a (H078K5G018)Document2 pagesCoa - Ofx-8040a (H078K5G018)Jaleel AhmedNo ratings yet
- Upgrade DB 10.2.0.4 12.1.0Document15 pagesUpgrade DB 10.2.0.4 12.1.0abhishekNo ratings yet
- Part 9. Wireless Communication Towers and Antennas 908.01 Purpose and IntentDocument12 pagesPart 9. Wireless Communication Towers and Antennas 908.01 Purpose and IntentjosethompsonNo ratings yet
- Pass Issuance Receipt: Now You Can Also Buy This Pass On Paytm AppDocument1 pagePass Issuance Receipt: Now You Can Also Buy This Pass On Paytm AppAnoop SharmaNo ratings yet
- Method StatementDocument11 pagesMethod StatementMohammad Fazal Khan100% (1)
- AB InBev Code of Business Conduct PDFDocument10 pagesAB InBev Code of Business Conduct PDFcristian quelmis vilca huarachiNo ratings yet
- HealthFlex Dave BauzonDocument10 pagesHealthFlex Dave BauzonNino Dave Bauzon100% (1)
- Research Design Elements for ScenariosDocument25 pagesResearch Design Elements for Scenariosrohizal ibrahimNo ratings yet
- HW3Document3 pagesHW3Noviyanti Tri Maretta Sagala0% (1)
- Letter of Recommendation For Nicolas Hallett From Big Ten Network's Casey Peterson.Document1 pageLetter of Recommendation For Nicolas Hallett From Big Ten Network's Casey Peterson.Nic HallettNo ratings yet
- Analysis of Trend Following SystemsDocument52 pagesAnalysis of Trend Following SystemsClement Li100% (1)
- 2014 March CaravanDocument48 pages2014 March CaravanbahiashrineNo ratings yet
- Vydyne® R533H NAT: Ascend Performance Materials Operations LLCDocument4 pagesVydyne® R533H NAT: Ascend Performance Materials Operations LLCJames FaunceNo ratings yet
- Fleck 3150 Downflow: Service ManualDocument40 pagesFleck 3150 Downflow: Service ManualLund2016No ratings yet