Professional Documents
Culture Documents
CTT
Uploaded by
carolinaCopyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
CTT
Uploaded by
carolinaCopyright:
Available Formats
Articles
The eects of lowering LDL cholesterol with statin therapy
in people at low risk of vascular disease: meta-analysis of
individual data from 27 randomised trials
Cholesterol Treatment Trialists (CTT) Collaborators*
Summary
Background Statins reduce LDL cholesterol and prevent vascular events, but their net eects in people at low risk of Lancet 2012; 380: 58190
vascular events remain uncertain. Published Online
May 17, 2012
http://dx.doi.org/10.1016/
Methods This meta-analysis included individual participant data from 22 trials of statin versus control (n=134 537;
S0140-6736(12)60367-5
mean LDL cholesterol dierence 108 mmol/L; median follow-up 48 years) and ve trials of more versus less statin
See Comment page 541 and 545
(n=39 612; dierence 051 mmol/L; 51 years). Major vascular events were major coronary events (ie, non-fatal
*Collaborators are listed at the
myocardial infarction or coronary death), strokes, or coronary revascularisations. Participants were separated into end of the report
ve categories of baseline 5-year major vascular event risk on control therapy (no statin or low-intensity statin) (<5%, Correspondence to:
5% to <10%, 10% to <20%, 20% to <30%, 30%); in each, the rate ratio (RR) per 10 mmol/L LDL cholesterol CTT Secretariat, Clinical Trial
reduction was estimated. Service Unit and Epidemiological
Studies Unit (CTSU), Richard Doll
Building, Old Road Campus,
Findings Reduction of LDL cholesterol with a statin reduced the risk of major vascular events (RR 079, Oxford OX3 7LF, UK
95% CI 077081, per 10 mmol/L reduction), largely irrespective of age, sex, baseline LDL cholesterol or previous ctt@ctsu.ox.ac.uk
vascular disease, and of vascular and all-cause mortality. The proportional reduction in major vascular events was at or
least as big in the two lowest risk categories as in the higher risk categories (RR per 10 mmol/L reduction from National Health and Medical
lowest to highest risk: 062 [99% CI 047081], 069 [99% CI 060079], 079 [99% CI 074085], 081 [99% CI Research Council (NHMRC)
077086], and 079 [99% CI 074084]; trend p=004), which reected signicant reductions in these two lowest Clinical Trial Centre, Mallett
Street Campus M02, University
risk categories in major coronary events (RR 057, 99% CI 036089, p=00012, and 061, 99% CI 050074,
of Sydney, NSW 2006, Australia
p<00001) and in coronary revascularisations (RR 052, 99% CI 035075, and 063, 99% CI 051079; both ctt@ctc.usyd.edu.au
p<00001). For stroke, the reduction in risk in participants with 5-year risk of major vascular events lower than 10%
(RR per 10 mmol/L LDL cholesterol reduction 076, 99% CI 061095, p=00012) was also similar to that seen in
higher risk categories (trend p=03). In participants without a history of vascular disease, statins reduced the risks of
vascular (RR per 10 mmol/L LDL cholesterol reduction 085, 95% CI 077095) and all-cause mortality (RR 091,
95% CI 085097), and the proportional reductions were similar by baseline risk. There was no evidence that
reduction of LDL cholesterol with a statin increased cancer incidence (RR per 10 mmol/L LDL cholesterol reduction
100, 95% CI 096104), cancer mortality (RR 099, 95% CI 093106), or other non-vascular mortality.
Interpretation In individuals with 5-year risk of major vascular events lower than 10%, each 1 mmol/L reduction in
LDL cholesterol produced an absolute reduction in major vascular events of about 11 per 1000 over 5 years. This
benet greatly exceeds any known hazards of statin therapy. Under present guidelines, such individuals would not
typically be regarded as suitable for LDL-lowering statin therapy. The present report suggests, therefore, that these
guidelines might need to be reconsidered.
Funding British Heart Foundation; UK Medical Research Council; Cancer Research UK; European Community Biomed
Programme; Australian National Health and Medical Research Council; National Heart Foundation, Australia.
Introduction yielded further reductions in risk. There was no evidence
The Cholesterol Treatment Trialists (CTT) Collaboration that lowering of LDL cholesterol increased the risk of
previously reported meta-analyses of individual data non-vascular death or of cancer, even in participants with
from 170 000 individuals in 21 trials of standard statin baseline LDL cholesterol less than 2 mmol/L (in whom
regimens versus control and ve trials of more intensive LDL cholesterol was reduced from about 17 mmol/L
versus less intensive regimens.1 That report showed that [65 mg/dL] to 13 mmol/L [50 mg/dL]).2
lowering of LDL cholesterol by 1 mmol/L with a standard In those analyses, reduction of LDL cholesterol with a
statin regimen reduced the incidence of major vascular statin in participants with no previous history of
events (dened as non-fatal myocardial infarction or vascular disease reduced the risk of major vascular
coronary death, any stroke, or coronary revascularisation events by about a fth,1 but there remains uncertainty
procedure) by around a fth, and that further reductions about whether statin therapy is of overall net benet
in LDL cholesterol with more intensive statin regimens in primary prevention.35 This question is important
www.thelancet.com Vol 380 August 11, 2012 581
Descargado para Anonymous User (n/a) en ClinicalKey Espanol Columbia, Ecuador & Peru Flood Relief de ClinicalKey.es por Elsevier en abril 19, 2017.
Para uso personal exclusivamente. No se permiten otros usos sin autorizacin. Copyright 2017. Elsevier Inc. Todos los derechos reservados.
Articles
because, although individuals without previous vascular from both primary prevention trials and low-risk
disease are at lower absolute risk, at least half of all participants in other trials) allows a more complete
vascular events occur among them.6 The availability of assessment of the eects of lowering of LDL cholesterol
individual participant data from each trial within the in low-risk people than was possible in previous meta-
CTT database (allowing the inclusion of information analyses of published data.3,4
Median predicted Estimated 5-year MVE risk Total
5-year MVE risk
<5% 5% to <10% 10% to <20%* 20% to <30% 30%
Statin vs control
MEGA 27% 7247 (147) 925 (91) 42 (4) 0 (0) 0 (0) 8214 (242)
JUPITER 44% 11 212 (118) 6117 (162) 472 (19) 1 (0) 0 (0) 17 802 (299)
AFCAPS/ TexCAPS 52% 2944 (72) 3329 (225) 331 (47) 1 (0) 0 (0) 6605 (344)
ASCOT-LLA 81% 1505 (25) 5383 (229) 3168 (245) 234 (22) 15 (3) 10 305 (524)
WOSCOPS 92% 34 (0) 3848 (219) 2576 (293) 134 (36) 3 (2) 6595 (550)
GISSI-HF 96% 875 (34) 1523 (78) 1789 (171) 357 (52) 30 (11) 4574 (346)
ALERT 102% 286 (8) 740 (54) 705 (105) 237 (59) 134 (49) 2102 (275)
CARDS 109% 156 (0) 1043 (43) 1524 (144) 109 (16) 6 (1) 2838 (204)
ASPEN 136% 108 (0) 648 (13) 980 (99) 517 (90) 157 (48) 2410 (250)
ALLHAT-LLT 140% 92 (2) 2331 (144) 5468 (803) 1871 (438) 593 (183) 10 355 (1570)
Post-CABG 170% 0 (0) 24 (2) 1022 (128) 279 (44) 26 (5) 1351 (179)
GISSI-P 183% 0 (0) 14 (1) 2816 (272) 1268 (140) 173 (26) 4271 (439)
HPS 186% 320 (14) 2041 (134) 9424 (1267) 6722 (1488) 2029 (651) 20 536 (3554)
LIPID 221% 0 (0) 27 (3) 2946 (484) 5144 (1278) 897 (324) 9014 (2089)
PROSPER 224% 0 (0) 108 (13) 2208 (224) 1858 (293) 1630 (396) 5804 (926)
CORONA 232% 11 (1) 194 (16) 1463 (144) 2151 (292) 1192 (177) 5011 (630)
CARE 261% 0 (0) 0 (0) 362 (58) 2776 (615) 1021 (313) 4159 (986)
ALLIANCE 263% 0 (0) 0 (0) 288 (46) 1419 (298) 735 (203) 2442 (547)
LIPS 295% 0 (0) 0 (0) 118 (28) 770 (158) 789 (173) 1677 (359)
AURORA 317% 0 (0) 61 (8) 546 (85) 676 (136) 1490 (501) 2773 (730)
SSSS 331% 0 (0) 0 (0) 139 (13) 1159 (275) 3146 (1063) 4444 (1351)
4D 382% 0 (0) 6 (1) 117 (20) 273 (52) 859 (233) 1255 (306)
Subtotal, 22 trials 137% 24 790 (421) 28 362 (1436) 38 504 (4699) 27 956 (5782) 14 925 (4362) 134 537 (16 700)
More vs less statin
SEARCH 162% 9665 (1969) 2157 (680) 242 (104) 12 064 (2753)
A to Z 173% 3097 (307) 1191 (180) 209 (52) 4497 (539)
TNT 218% 3507 (520) 5078 (1075) 1416 (458) 10 001 (2053)
IDEAL 241% 1754 (326) 5257 (1125) 1877 (593) 8888 (2044)
PROVE-IT 338% 27 (3) 1125 (185) 3010 (676) 4162 (864)
Subtotal, ve trials 208% 18 050 (3125) 14 808 (3245) 6754 (1883) 39 612 (8253)
Data are median risk or number of participants (number of rst major vascular events). Studies are shown in order of increasing median predicted 5-year MVE risk. The
predicted risk for the trials of more versus less statin is that under the less intensive statin regimen. We imputed missing data for age, sex, treatment for hypertension, lipids,
and blood pressure at baseline for the purpose of predicting 5-year MVE risk and for risk stratication. MEGA=Management of Elevated Cholesterol in the Primary Prevention
Group of Adult Japanese. JUPITER=Justication for the Use of Statins in Prevention: an Intervention Trial Evaluating Rosuvastatin. AFCAPS/TexCAPS=Air Force/Texas Coronary
Atherosclerosis Prevention Study. ASCOT-LLA=Anglo-Scandinavian Cardiac Outcomes Trial-Lipid Lowering Arm. WOSCOPS=West of Scotland Coronary Prevention Study.
GISSI-HF=Gruppo Italiano per lo Studio della Sopravvivenza nellInsucienza cardiaca. ALERT=Assessment of Lescol in Renal Transplantation. CARDS=Collaborative
Atorvastatin Diabetes Study. ASPEN=Atorvastatin Study for Prevention of Coronary Heart Disease Endpoints in Non-Insulin-Dependent Diabetes Mellitus.
ALLHAT-LLT=Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial. Post-CABG=Post-Coronary Artery Bypass Graft. GISSI-P=Gruppo Italiano per lo
Studio della Sopravvivenza nellInfarto Miocardico. HPS=Heart Protection Study. LIPID=Long-term Intervention with Pravastatin in Ischaemic Disease. PROSPER=PROspective
Study of Pravastatin in the Elderly at Risk. CORONA=Controlled Rosuvastatin Multinational Trial in Heart Failure. CARE=Cholesterol And Recurrent Events.
ALLIANCE=Aggressive Lipid-Lowering Initiation Abates New Cardiac Events. LIPS=Lescol Intervention Prevention Study. AURORA=A Study to Evaluate the Use of Rosuvastatin
in Subjects on Regular Hemodialysis: an Assessment of Survival and Cardiovascular Events. SSSS=Scandinavian Simvastatin Survival Study. 4D=Die Deutsche Diabetes Dialyse
Studie. SEARCH=Study of the Eectiveness of Additional Reductions in Cholesterol and Homocysteine. A to Z=Aggrastat to Zocor. TNT=Treating to New Targets.
IDEAL=Incremental Decrease in End Points Through Aggressive Lipid Lowering Study Group. PROVE-IT=Pravastatin or Atorvastatin Evaluation and Infection Therapy.
*For trials of more versus less statin, this category includes 141 participants (48 [4 MVEs] from A to Z and 93 [11 MVEs] from SEARCH) with an estimated 5-year risk of MVE
between 5% and 10%. Includes 382 patients who were excluded from the original publication.
Table 1: Numbers of participants and number of rst major vascular events (MVEs) in each study contributing to vascular disease risk categories
582 www.thelancet.com Vol 380 August 11, 2012
Descargado para Anonymous User (n/a) en ClinicalKey Espanol Columbia, Ecuador & Peru Flood Relief de ClinicalKey.es por Elsevier en abril 19, 2017.
Para uso personal exclusivamente. No se permiten otros usos sin autorizacin. Copyright 2017. Elsevier Inc. Todos los derechos reservados.
Articles
Methods both treatment groups of each study were assigned to
Trials one of ve baseline categories of 5-year risk of a major
The methods of the CTT collaboration have been vascular event: <5%; 5% to <10%; 10% to <20%; 20%
described in detail elsewhere.1,7,8 In the present analyses, to <30%; or 30%. Further details of model development
a trial was eligible if it reported by the end of 2009 and are shown in the appendix pp 1516.
provided data before June, 2011, and met three criteria: Analyses included all participants who were randomly
(1) it included at least one intervention whose main assigned to treatment groups, irrespective of whether
eect was to lower LDL cholesterol concentration; (2) it they received their allocated treatment (ie, intention-to-
was unconfounded with respect to this intervention (ie, treat). Analyses of the eects on disease rates within each
no other dierences in risk factor modication between trial are derived from the logrank (oe) statistic and its
the treatment groups were intended); and (3) it variance (v) for rst events. Meta-analyses were weighted
recruited at least 1000 participants with scheduled by the absolute LDL cholesterol dierence in that trial at
treatment duration of at least 2 years. The main 1 year (d mmol/L), and are reported as eects per
outcomes of interest were major vascular events, major 10 mmol/L reduction in LDL cholesterol. In a meta-
coronary events (dened as non-fatal myocardial analysis of several trials, the log of the rate ratio (RR) per
infarction or coronary death), stroke (subdivided by 10 mmol/L was calculated as S/V with variance 1/V (and
type), coronary revascularisation procedures, cancers, hence, for example, with 95% CI of S/V196/V), where
and cause-specic mortality. S is the sum over all trials of d(oe) and V is the sum over
all trials of dv. For most subgroup analyses, the weight
Statistical analysis for a particular subgroup was the LDL cholesterol
Two dierent Cox proportional hazards models were dierence observed in the whole trial, but analyses by
developed to allow risk to be modelled separately in trials baseline LDL cholesterol concentration used LDL weights
of statin versus control (22 trials; model 1) and in trials specic to a particular subgroup of a particular trial.1 In
of more versus less intensive statin regimens (ve trials; trials comparing more versus less intensive statin therapy,
model 2). Both models incorporated terms derived from the relevant baseline lipid values were those achieved on
characteristics measured at the time of randomisation, the less intensive regimen. In three of these trials,911
terms that modelled average dierences in risk between however, any statin therapy was stopped before random-
trials (as well as within specic periods of time within isation, so we estimated their relevant baseline values by See Online for appendix
each trial), and interaction terms (appendix pp 12). On multiplying the values at the randomisation visit (ie, o
the basis of these risk prediction models, participants in statin treatment) by the mean proportional reduction
Number of Observed Observed Median Women (%) Mean age Baseline Diabetes (%) Baseline history of vascular disease (%)
participants annual MVE annual MCE follow-up (SD; years) LDL
rate in those rate in those in survivors cholesterol
allocated allocated (years)* (mmol/L)*
control or control or
less statin less statin
Previous CHD Other vascular None
Statin vs control
<5% 24 790 06% 02% 40 54% 59 (8) 343 7% 0% 4% 96%
5% to <10% 28 362 16% 08% 43 27% 61 (9) 368 18% 2% 11% 87%
10% to <20% 38 504 34% 15% 47 29% 64 (9) 361 24% 43% 22% 44%
20% to <30% 27 956 57% 26% 50 16% 65 (9) 372 19% 80% 28% 13%
30% 14 925 95% 51% 49 14% 66 (9) 392 44% 86% 39% 7%
Subtotal, 22 trials 134 537 36% 18% 48 29% 63 (9) 370 21% 39% 20% 52%
More vs less statin
10% to <20% 18 050 37% 16% 59 22% 60 (10) 237 4% 100% 1% 0%
20% to <30% 14 808 59% 24% 52 17% 62 (9) 259 17% 100% 12% 0%
30% 6754 107% 42% 24 18% 64 (10) 281 35% 100% 35% 0%
Subtotal , ve trials 39 612 53% 22% 51 19% 62 (10) 253 14% 100% 11% 0%
CHD=coronary heart disease. MCE=major coronary event. *Estimated using standard Kaplan-Meier methods with participants censored at their date of death; median follow-up and baseline LDL cholesterol for
trial subgroups weighted by trial subgroup-specic variances of observed logrank (oe) for major vascular events. History of intracerebral bleed, transient ischaemic attack, ischaemic stroke, unknown stroke,
peripheral artery disease or heart failure (if known). No known history of CHD or other vascular disease. The estimated 5-year major vascular event risk is with the less intensive statin regimen and observed
MVE and MCE rates are for participants allocated the less intensive statin regimen; in three more versus less statin trials (A to Z, PROVE-IT, and IDEAL) there was no active run-in period before randomisation and
so for the purpose of risk stratication and presentation of results the LDL cholesterol at baseline for the participants in these trials was adjusted for the observed LDL cholesterol reduction from baseline to year 1
in those allocated low intensity statin in the respective trial. Includes 141 participants (48 [4 MVEs] from A to Z and 93 [11 MVEs] from SEARCH) with an estimated 5-year risk of MVE between 5% and 10%.
Table 2: Baseline characteristics of participants, by predicted 5-year risk of a major vascular event (MVE)
www.thelancet.com Vol 380 August 11, 2012 583
Descargado para Anonymous User (n/a) en ClinicalKey Espanol Columbia, Ecuador & Peru Flood Relief de ClinicalKey.es por Elsevier en abril 19, 2017.
Para uso personal exclusivamente. No se permiten otros usos sin autorizacin. Copyright 2017. Elsevier Inc. Todos los derechos reservados.
Articles
5-year MVE risk Events (% per annum) RR (CI) per 10 mmol/L reduction Trend test
Results
at baseline in LDL cholesterol Individual participant data were available from 27 trials
Statin/more Control/less in 174 149 participants. 22 trials compared a standard
Major coronary event statin regimen versus control (134 537 participants; mean
<5% 50 (011) 88 (019) 057 (036089) baseline LDL cholesterol 370 [SD 07] mmol/L; mean
5% to <10% 276 (050) 435 (079) 061 (050074) dierence at 1 year 108 mmol/L; median follow-up
10% to <20% 1644 (129) 1973 (157) 077 (069085) 21=566 duration in survivors 48 years)1233 and ve trials assessed
20% to <30% 1789 (193) 2282 (249) 077 (071083) (p=002) a more intensive versus a less intensive statin regimen
30% 1471 (373) 1887 (486) 078 (072084) (39 612 participants; mean baseline LDL cholesterol
Overall 5230 (145) 6665 (187) 076 (073079) 253 [SD 06] mmol/L; mean dierence at 1 year
p<00001
051 mmol/L; median follow-up duration in survivors
Any stroke 51 years).911,34,35 Individual participant data were unavail-
<5% 71 (016) 90 (020) 074 (046119) able from only two eligible trials in 6331 higher-risk
5% to <10% 190 (034) 240 (043) 077 (060098) patients with pre-existing vascular disease (SPARCL36
10% to <20% 797 (062) 907 (071) 086 (075098) 21=103 and GREACE37).
20% to <30% 781 (084) 900 (097) 086 (075097) (p=03)
The baseline prognostic factors that were strong
30% 571 (145) 661 (168) 086 (075099)
predictors of major vascular event risk (ie, at the 1%
Overall 2410 (067) 2798 (078) 085 (080089)
p<00001 signicance level) were broadly similar in the trials of
statin versus control and the trials of more versus less
Coronary revascularisation intensive statin regimens (appendix pp 12). Predicted
<5% 73 (016) 135 (030) 052 (035075)
risk compared well with observed risk for each trial, as
5% to <10% 224 (040) 342 (062) 063 (051079)
well as within each 5-year risk group (appendix p 3).
10% to <20% 1706 (136) 2061 (167) 075 (067083) 21=493
(p=003)
When trials were ordered by their median 5-year
20% to <30% 2206 (246) 2717 (308) 079 (073086)
30% 1260 (328) 1655 (440) 076 (069083)
predicted risk of a major vascular event, the ve trials
Overall 5469 (155) 6910 (198) 076 (073079) with the lowest median predicted risks (all <10%) were
p<00001 primary prevention trials (table 1).13,15,22,29,31 By contrast,
almost all participants with predicted 5-year risk of 20%
Major vascular event
<5% 167 (038) 254 (056) 062 (047081)
or higher were recruited into trials in patients with a
5% to <10% 604 (110) 847 (157) 069 (060079)
denite history of vascular disease.12,14,1619,25,28 The
10% to <20% 3614 (296) 4195 (350) 079 (074085) 21=429 predicted 5-year risk of a major vascular event was also
20% to <30% 4108 (474) 4919 (580) 081 (077086) (p=004) 20% or higher in most dialysis patients.26,30 In two trials
30% 2787 (764) 3458 (982) 079 (074084) in patients with heart failure,32,33 there was a high risk of
Overall 11 280 (327) 13 673 (404) 079 (077081) sudden death, but such deaths were categorised
p<00001 dierently, with a much smaller proportion of such
99% limits 95% limits 050 075 1 125 150 deaths thought to be due to coronary occlusion in the
Statin/more better Control/less better GISSI-HF trial33 than in CORONA;32 this dierence is
Figure 1: Eects on major coronary events, strokes, coronary revascularisation procedures, and major the main reason why the predicted 5-year risk of major
vascular events per 10 mmol/L reduction in LDL cholesterol at dierent levels of risk vascular events was more than twice as high in CORONA
MVE=major vascular event. RR=rate ratio. CI=condence interval. (23%) as in GISSI-HF (10%).
Among the 22 trials of statin versus control, the
observed at 1 year in participants allocated the less observed annual major vascular event rate ranged from
intensive regimen.1 Proportional risk reductions in 06% in the lowest predicted risk category to 95% in the
dierent subgroups were compared by standard tests highest risk category, whereas in trials of more versus
for heterogeneity or, where appropriate, trend. To allow less intensive statin therapy (which were undertaken
for multiple testing by subdivisions, only overall summary solely in patients with previous coronary disease) the
rate ratios have 95% CIs; all other rate ratios have observed annual event rate varied between 37% and
99% CIs. Analyses used SAS version 9.1 (SAS Institute, 107% across the categories studied (table 2). In both sets
Cary, NC, USA), Stata version 11.2 (StataCorp, TX, USA), of trials, the achieved reduction in LDL cholesterol at
For more on R see and R version 2.11.1. 1 year with statin therapy or more intensive statin therapy
www.R-project.org was greater in people with higher predicted 5-year risk of
Role of the funding sources major vascular events (appendix p 4).
The funding sources had no involvement in the study Among all 27 trials, statins reduced the risk of major
design, data collection, analysis and interpretation, the vascular events by 21% per 10 mmol/L LDL cholesterol
writing of the report, or the decision to submit for reduction (RR 079, 95% CI 077081, p<00001), with
publication. The writing committee had full access to all separately signicant proportional reductions in each
data and accepts full responsibility for the content of risk group (gure 1). In particular, there were signicant
this report. reductions in major vascular event risk in each of the two
584 www.thelancet.com Vol 380 August 11, 2012
Descargado para Anonymous User (n/a) en ClinicalKey Espanol Columbia, Ecuador & Peru Flood Relief de ClinicalKey.es por Elsevier en abril 19, 2017.
Para uso personal exclusivamente. No se permiten otros usos sin autorizacin. Copyright 2017. Elsevier Inc. Todos los derechos reservados.
Articles
lowest risk categories (RR per 10 mmol/L LDL 5-year MVE Events (% per annum) RR (CI) per 10 mmol/L reduction Trend test
cholesterol reduction 062, 99% CI 047081, for 5-year risk at in LDL cholesterol
predicted risk <5%, and 069, 99% CI 060079, for baseline
5-year predicted risk 5% to <10%; both p<00001; Statin/more Control/less
gure 1). These results were qualitatively similar after Participants without vascular disease
exclusion of ve trials12,15,22,24,31 that ended early on the <5% 148 (035) 229 (053) 061 (045081)
advice of their data monitoring committees (data not 5% to <10% 487 (102) 716 (153) 066 (057077)
10% to <20% 854 (252) 1003 (298) 082 (072093) 21=910
shown). The proportional reductions in major vascular
20% to <30% 294 (440) 351 (528) 081 (065101) (p=0003)
events per 10 mmol/L LDL cholesterol reduction in the
30% 121 (729) 126 (816) 083 (058118)
two lowest risk categories seemed to be at least as large
Subtotal 1904 (144) 2425 (184) 075 (070080)
as for other participants (gure 1), even after further p<00001
stratication by age and sex (appendix p 6) or by baseline
Participants with vascular disease
LDL cholesterol (appendix p 7).
<5% 19 (087) 25 (118) 073 (033161)
The reductions in risk of major vascular events among 084 (062114)
5% to <10% 117 (156) 131 (180)
the two categories of participant at lowest risk reected 10% to <20% 2760 (313) 3192 (371) 078 (072085) 21=001
reductions in major coronary events (RR per 10 mmol/L 20% to <30% 3814 (477) 4568 (585) 081 (076086) (p=09)
LDL cholesterol reduction 057, 99% CI 036089, 30% 2666 (766) 3332 (990) 079 (074084)
p=00012, and 061, 99% CI 050074, p<00001), Subtotal 9376 (441) 11 248 (543) 080 (077082)
mainly non-fatal myocardial infarction, and in coronary p<00001
revascularisations (RR 052, 99% CI 035075, and All participants
063, 99% CI 051079; both p<00001), that were at <5% 167 (038) 254 (056) 062 (047081)
least as large as those seen in higher risk participants 5% to <10% 604 (110) 847 (157) 069 (060079)
(trend p=002 for major coronary events and p=003 for 10% to <20% 3614 (296) 4195 (350) 079 (074085) 21=429
coronary revascularisations; gure 1, appendix p 8). The 20% to <30% 4108 (474) 4919 (580) 081 (077086) (p=004)
reduction in stroke risk per 10 mmol/L LDL cholesterol 30% 2787 (764) 3458 (982) 079 (074084)
reduction (RR 085, 95% CI 080089) was similar at all Overall 11 280 (327) 13 673 (404) 079 (077081)
Heterogeneity between participants without p<00001
levels of baseline major vascular event risk (trend p=03;
and with vascular disease: 21=274 (p=010)
gure 1). In particular, the reduction in stroke risk in
99% limits 95% limits 050 075 1 125 150
those with predicted 5-year major vascular event risk
Statin/more better Control/less better
lower than 10% (ie, the two lowest risk groups combined;
RR 076, 99% CI 061095; p=00012) was similar to Figure 2: Eects on major vascular events per 10 mmol/L reduction in LDL cholesterol at dierent levels of
that seen in higher risk categories. The proportional risk, by history of vascular disease
MVE=major vascular event. RR=rate ratio. CI=condence interval.
reductions in ischaemic stroke (RR per 10 mmol/L LDL
cholesterol reduction 079, 95% CI 074085) and in
strokes of unknown cause (RR 087, 95% CI 079097) predicted risk 5% to <10%). The LDL-weighted
were similar irrespective of baseline major vascular event proportional reduction in major coronary events in par-
risk, and there was no evidence that the RR for ticipants with no history of vascular disease was also at
haemorrhagic stroke (115, 95% CI 097138) varied by least as large in the two lowest risk groups as in those at
baseline risk (appendix p 9). Separate analyses of major higher risk (appendix p 12).
vascular events and its components in the trials that In all participants, there was a proportional reduction
tested statin versus control and the trials that tested more in vascular mortality of 12% per 10 mmol/L LDL
versus less intensive statin regimens are shown in the cholesterol reduction (RR 088, 95% CI 084091;
appendix pp 1011. gure 3), which was chiey attributable to reductions in
For participants with previous vascular disease, the coronary deaths of 20% (RR 080, 95% CI 076085)
proportional reductions in major vascular event risk and in other cardiac deaths of 8% (RR 092, 95% CI
were broadly similar irrespective of predicted risk of 087098; appendix p 13). There were too few deaths
these events (gure 2). In participants with no history of among the lower risk participants to allow reliable direct
vascular disease, the proportional reduction in major assessment of the eects of statin therapy (appendix
vascular events was at least as large in the two lowest risk p 13). However, the LDL-weighted proportional reduc-
groups (RR per 10 mmol/L LDL reduction 061, 99% CI tions seemed similar in each risk category both for the
045081, and 066, 99% CI 057077) as in those at aggregate of all vascular deaths (trend p=07; gure 3)
higher risk. Further exclusion from these lowest risk and for each specic cause of vascular death (all trend
groups of participants with diabetes or chronic kidney p=NS; appendix p 13), and there was no signicant trend
disease had little eect on the proportional reductions in towards an increase in non-vascular mortality in those at
major vascular events (RR per 10 mmol/L LDL lower risk (trend p=09; gure 3). In participants with
cholesterol reduction 063, 99% CI 046085, for 5-year no history of vascular disease, reduction of LDL
predicted risk <5%; 064, 99% CI 054076, for 5-year cholesterol with statin therapy reduced the risk of
www.thelancet.com Vol 380 August 11, 2012 585
Descargado para Anonymous User (n/a) en ClinicalKey Espanol Columbia, Ecuador & Peru Flood Relief de ClinicalKey.es por Elsevier en abril 19, 2017.
Para uso personal exclusivamente. No se permiten otros usos sin autorizacin. Copyright 2017. Elsevier Inc. Todos los derechos reservados.
Articles
vascular mortality (RR per 10 mmol/L reduction 085,
5-year MVE Events (% per annum) RR (CI) per 10 mmol/L reduction Trend test 95% CI 077095, p=0004; gure 3) and, since there
risk at in LDL cholesterol
baseline was no increase in the risk of non-vascular causes of
Statin/more Control/less death (RR 097, 95% CI 088107; gure 3), reduced
Any vascular death the risk of all-cause mortality (RR 091, 95% CI
Participants without vascular disease 085097, p=0007; appendix p 14). These mortality
<5% 31 (007) 40 (009) 080 (043147) benets remained even after further exclusion of
5% to <10% 117 (024) 153 (032) 075 (055104) participants with diabetes or chronic kidney disease at
21=146
10% to <20% 307 (087) 342 (096) 084 (067105) baseline (vascular death, RR 080, 95% CI 067095;
(p=02)
20% to <30% 164 (232) 168 (234) 097 (072132) any death, 087, 95% CI 078095). There was no
30% 93 (521) 98 (584) 088 (059133) evidence of an increase in cancer incidence (RR per
Subtotal 712 (053) 801 (059) 085 (077095) 10 mmol/L LDL reduction 100, 95% CI 096104) or
p=0004
of cancer death (RR 099, 95% CI 093106) at any level
Participants with vascular disease of major vascular event risk (gure 4).
<5% 48 (216) 52 (240) 093 (053162)
5% to <10% 193 (252) 177 (235) 107 (081141)
21=149
Discussion
10% to <20% 1166 (124) 1249 (134) 089 (079100)
(p=02)
The most recent CTT meta-analysis of individual
20% to <30% 1432 (161) 1665 (189) 087 (080095) participant data from randomised trials showed that
30% 1247 (314) 1435 (360) 087 (079095) lowering of LDL cholesterol with standard statin
Subtotal 4086 (176) 4578 (198) 088 (084092)
p<00001
regimens safely reduced the 5-year incidence of major
coronary events, coronary revascularisations, and
All participants ischaemic strokes by about one fth per 10 mmol/L
<5% 79 (018) 92 (020) 087 (058131)
reduction in LDL cholesterol, and that additional
5% to <10% 310 (055) 330 (059) 092 (074113)
21=018 reductions in LDL cholesterol obtained with more
10% to <20% 1473 (114) 1591 (123) 088 (079097)
(p=07) intensive statin regimens further reduced the incidence
20% to <30% 1596 (167) 1833 (192) 088 (081096)
30% 1340 (323) 1533 (369) 087 (080095)
of these major vascular events.1 The present results show
Overall 4798 (130) 5379 (147) 088 (084091)
that reduction of LDL cholesterol with statin therapy
Heterogeneity between participants without p<00001 signicantly reduced the risk of major vascular events in
and with vascular disease: 21=028 (p=06) individuals with 5-year risk lower than 10% (in whom
Non-vascular death
the mean risks were 26% for major coronary events
Participants without vascular disease plus 3% for other major vascular events), even in those
<5% 98 (023) 87 (020) 113 (076169) with no previous history of vascular disease, diabetes, or
5% to <10% 205 (042) 238 (049) 087 (067111) chronic kidney disease (panel).
21=047
10% to <20% 352 (099) 377 (106) 094 (076115) The estimated absolute reduction in major vascular
(p=05)
20% to <30% 169 (239) 148 (207) 113 (081157) events in participants with 5-year risk of these events
30% 79 (443) 71 (423) 107 (068169) lower than 10% was around 11 per 1000 over 5 years for
Subtotal 903 (067) 921 (068) 097 (088107) each 10 mmol/L reduction in LDL cholesterol (41%
p=060
statin or more intensive statin regimen vs 52% control
Participants with vascular disease or less intensive regimen). Modern statin regimens,
<5% 18 (081) 14 (065) 138 (053363) however, can often reduce LDL cholesterol by more than
5% to <10% 65 (085) 71 (094) 092 (061141) 1 mmol/L,38,39 which would yield even larger absolute
21=004
10% to <20% 702 (074) 727 (078) 095 (081111)
(p=08)
reductions in major vascular events. The avoidance of
20% to <30% 794 (090) 793 (090) 098 (086112) life-threatening or potentially disabling events in
30% 602 (152) 634 (159) 095 (082109) apparently healthy low-risk people might be deemed
Subtotal 2181 (094) 2239 (097) 096 (090102)
p=018
worthwhile provided that they are not accompanied by
any denite hazard that is of comparable severity.
All participants Although there was no evidence of any increased risk of
<5% 116 (026) 101 (022) 116 (080168) death from non-vascular causes or of cancer in those at
5% to <10% 270 (048) 309 (055) 088 (071109)
21=002 low risk (which is consistent with previous detailed
10% to <20% 1054 (081) 1104 (086) 094 (083107)
(p=09) analyses of the eect of statins on cancer2), several known
20% to <30% 963 (101) 941 (099) 100 (089113)
30% 681 (164) 705 (170) 096 (083110)
Overall 3084 (084) 3160 (086) 096 (092101) Figure 3: Eects on vascular and non-vascular deaths per 10 mmol/L reduction
Heterogeneity between participants without p=016 in LDL cholesterol at dierent levels of risk, by history of vascular disease
and with vascular disease: 21=007 (p=08) MVE=major vascular event. RR=rate ratio. CI=condence interval. There were a
further 179 (statin/more statin) versus 210 (control/less statin) deaths of
99% limits 95% limits 050 075 1 125 150 unknown cause among participants without vascular disease and 309 (statin/
Statin/more better Control/less better more statin) versus 338 (control/less statin) deaths of unknown cause among
participants with vascular disease.
586 www.thelancet.com Vol 380 August 11, 2012
Descargado para Anonymous User (n/a) en ClinicalKey Espanol Columbia, Ecuador & Peru Flood Relief de ClinicalKey.es por Elsevier en abril 19, 2017.
Para uso personal exclusivamente. No se permiten otros usos sin autorizacin. Copyright 2017. Elsevier Inc. Todos los derechos reservados.
Articles
or potential hazards of statin therapy need to be 5-year MVE risk Events (% per annum) RR (CI) per 10 mmol/L reduction Trend test
considered when estimating the net eects of statin at baseline in LDL cholesterol
therapy in people at lowest risk. Statin/more Control/less
First, statin therapy is associated with a small increased Cancer incidence
risk of myopathy (excess incidence of about 05 per 1000 <5% 324 (073) 315 (070) 105 (085131)
over 5 years) and, more rarely, of rhabdomyolysis (excess 5% to <10% 605 (110) 663 (121) 091 (078105)
incidence of about 01 per 1000 over 5 years).38 The risks 10% to <20% 1804 (142) 1827 (145) 100 (091110) 21=083
of myopathy are dose-related but, with the exception of 20% to <30% 1667 (179) 1628 (176) 102 (093112) (p=04)
simvastatin 80 mg daily (or lower doses in Asian 30% 821 (204) 777 (192) 102 (090116)
populations), intensive statin regimens have not been Overall 5221 (145) 5210 (145) 100 (096104)
p=099
shown to result in substantial myopathy risks.38 Second, Cancer death
the most recent CTT report raised the possibility that <5% 64 (014) 57 (013) 114 (069189)
statin therapy might increase the risk of haemorrhagic 5% to <10% 171 (030) 186 (033) 093 (071123)
stroke.1 The present analyses suggest that the annual 10% to <20% 697 (054) 718 (056) 100 (086116) 21=001
excess risk of haemorrhagic strokes per 10 mmol/L LDL 20% to <30% 609 (064) 584 (061) 102 (088119) (p=09)
cholesterol reduction might be of the order of 05 per 30% 293 (071) 304 (073) 097 (080119)
1000 people treated over 5 years (appendix p 9), although Overall 1834 (050) 1849 (050) 099 (093106)
p=086
it might be higher in populations in which haemorrhagic
99% limits 95% limits 050 075 1 125 150
stroke accounts for a higher proportion of strokes (eg,
Statin/more better Control/less better
Asian populations40). But, since statin therapy produced a
clear reduction in overall stroke that was independent of Figure 4: Eects on cancer incidence and cancer mortality per 10 mmol/L reduction in LDL cholesterol at
predicted risk, such an increase in haemorrhagic stroke dierent levels of risk
MVE=major vascular event. RR=rate ratio. CI=condence interval.
risk would typically be outweighed by the reduction in
the risk of ischaemic stroke (as well as the reduction in
other occlusive vascular events and deaths) even in Panel: Research in context
individuals whose 5-year risk of major vascular events is
lower than 5%. Third, recent meta-analyses have Systematic review
suggested that statin therapy might be associated with a Lowering of LDL cholesterol with a statin reduces the risk of
proportional increase in the diagnosis of diabetes myocardial infarction, coronary death, ischaemic stroke, and
mellitus of about 10%41 and that more intensive statin coronary revascularisation by about one fth per 1 mmol/L
therapy produces a bigger increase.42 The observed LDL cholesterol reduction in a wide range of people.1
incidence of diabetes recorded in the primary prevention However, tabular meta-analyses of people at low risk of these
trials was about 5% over 5 years, so the absolute excess events, studied predominantly in primary prevention trials,
was about 01% per year.41 If new diagnoses of diabetes have concluded that statin therapy might not result in
were associated with an immediate doubling in worthwhile net benet in this group.3,4
cardiovascular risk43 in individuals with 5-year risk of Interpretation
major vascular events lower than 10%, then the expected Individual participant data in the Cholesterol Treatment
eect would be only about 02 fewer events avoided per Trialists Collaboration of 27 trials involving
1000 individuals treated over 5 years. Such an eect is 175 000 participants showed that statin therapy reduces the
more than 50-times smaller than the absolute benet risk of major vascular events (non-fatal myocardial
observed with statin therapy in such individuals (about infarction, coronary death, coronary revascularisation, or
11 fewer major vascular events per 1000 treated over stroke) in people with 5-year risk of such an event lower than
5 years per 10 mmol/L reduction in LDL cholesterol; see 10% (and, separately, in those at 5-year risk <5%), and in
gure 5 for absolute benets corresponding to particular these people each 10 mmol/L reduction in LDL cholesterol
reductions in LDL cholesterol in individuals at dierent produces 11 fewer major vascular events per 1000 treated
levels of major vascular event risk). Moreover, long-term over 5 years, a benet that greatly exceeds any known
follow-up of statin trials has shown that the absolute hazards of statin therapy.
reductions in major vascular events increase while the
statin treatment is continued1 and that these benets
persist for at least 5 years after the treatment has stopped, The observed event rates shown in the gures for
with no evidence of any adverse eects emerging with each risk category can be readily compared with risk
extended follow-up.4447 These ndings would suggest thresholds used in treatment guidelines. For example,
that any long-term eects of any small excesses in under present guidelines, including those of the Adult
haemorrhagic strokes and in diagnoses of diabetes are Treatment Panel III,48,49 the Fourth Joint Task Force of
not associated with long-term eects on major vascular the European Society of Cardiology,50 the Task Force for
events that are suciently large to outweigh the the management of dyslipidaemias of the European
persistent benets of statin therapy. Society of Cardiology and the European Atherosclerosis
www.thelancet.com Vol 380 August 11, 2012 587
Descargado para Anonymous User (n/a) en ClinicalKey Espanol Columbia, Ecuador & Peru Flood Relief de ClinicalKey.es por Elsevier en abril 19, 2017.
Para uso personal exclusivamente. No se permiten otros usos sin autorizacin. Copyright 2017. Elsevier Inc. Todos los derechos reservados.
Articles
A Observed Observed Broad eligibility under
MCE vascular current guidelines
event rate death rate
(% per (% per
160 142 annum)* annum)*
ATP-III ESC task NICE
140 119 force
CTT risk category
Major vascular events avoided per 1000
120 93
100 <5% 02 01
100 84 5% to <10% 08 03
61
10% to <20% 16 10 3 3 3
66
80 20% to <30% 32 23 3 3 3
45 68
57 30% 56 58 3 3 3
60 45
31 CTT=Cholesterol Treatment Trialists. MCE=major coronary event. *Among
40 control-allocated participants without a history of vascular disease. The Adult
30% Treatment Panel III (ATP III) of the National Cholesterol Education p rogram in the
15 21 27 31 20% to <30% 5-year USA. The Fourth Joint Task Force of the European Society of Cardiology (ESC)
20 risk of and Other Societies on Cardiovascular Disease Prevention in Clinical Practice and
10% to <20% major the ESC/EAS Guidelines for the management of dyslipidaemias. The National
6 8 vascular
0 10 5% to <10% Institute for Health and Clinical Excellence (NICE) in the National Health Service in
1 12 event England and Wales.
15 <5%
2
25 Table 3: Eligibility of CTT participants without a history of vascular
disease for statin therapy under existing major guidelines, by estimated
B 5-year major vascular event risk
vascular events in patients at lower risk also depend on
50 46
the cost eectiveness of such a strategy, which in turn
45
depends on the local availability and cost of therapy.
38
Generic statin interventions, if eective, are likely to be
40
30
cost-eective in individuals at annual vascular disease
Vascular deaths avoided per 1000
35
risk down to at least about 1%.5355 The present report
shows that statins are indeed both eective and safe for
30 20
25 people with 5-year risk of major vascular events lower
25
21 than 10% and, therefore, suggests that these guidelines
16
might need to be reconsidered.
20 11
17
14 Contributors
11 The writing committee accepts full responsibility for the content of
15 8
this paper. All of the members contributed to collection and analysis of
30%
10 the data and to the preparation of the report. All collaborators had an
7 20% to <30% 5-year opportunity to contribute to the interpretation of the results and to
4 5 8
5 risk of
10% to <20% drafting of the report.
major
12 vascular
0 17 5% to <10% Current membership of the CTT Collaboration
22 event
1 27 Writing committee: B Mihaylova, J Emberson, L Blackwell, A Keech,
15 <5%
2 J Simes, E H Barnes, M Voysey, A Gray, R Collins, C Baigent.
25 Collaborating trialists: A to Z trial (phase Z) J de Lemos, E Braunwald,
LDL cholesterol reduction (mmol/L)
M Blazing, S Murphy; AFCAPS/TEXCAPS (AirForce/Texas Coronary
with statin treatment
Atherosclerosis Prevention Study) J R Downs, A Gotto, M Cleareld;
Figure 5: Predicted 5-year benets of LDL cholesterol reductions with statin treatment at dierent levels of risk ALERT (Assessment of Lescol in Transplantation) H Holdaas; ALLHAT
(A) Major vascular events and (B) vascular deaths. Lifetable estimates using major vascular event risk or vascular (Antihypertensive Lipid Lowering Heart Attack Trial) D Gordon, B Davis;
death risk in the respective risk categories and overall treatment eects per 10 mmol/L reduction in LDL ALLIANCE (Aggressive Lipid-Lowering Initiation Abates New Cardiac
cholesterol with statin. Events) M Koren; ASCOT (Anglo-Scandinavian Cardiac Outcomes Trial)
B Dahlof, N Poulter, P Sever; ASPEN (Atorvastatin Study for the
Prevention of Coronary Heart Disease Endpoints in Non-Insulin Dependent
Society,51 and the UK National Institute of Clinical Diabetes Mellitus) R H Knopp (deceased); AURORA (A study to evaluate
Excellence,52 people with 5-year risk of major vascular the Use of Rosuvastatin in subjects On Regular haemodialysis: an
Assessment of survival and cardiovascular events) B Fellstrm, H Holdaas,
events lower than 10% (ie, the lowest two categories of A Jardine, R Schmieder, F Zannad; BIP (Bezabrate Infarction
risk in these analyses) would typically not be judged Prevention Study) U Goldbourt, E Kaplinsky; CARDS (Collaborative
suitable for statin treatment (table 3, appendix p 5). Atorvastatin Diabetes Study) H M Colhoun, D J Betteridge,
P N Durrington, G A Hitman, J Fuller, A Neil; 4D (Die Deutsche
Judgments about the appropriateness of widespread
Diabetes Dialyse Studie) C Wanner, V Krane; CARE (Cholesterol And
prescription of statins for the primary prevention of
588 www.thelancet.com Vol 380 August 11, 2012
Descargado para Anonymous User (n/a) en ClinicalKey Espanol Columbia, Ecuador & Peru Flood Relief de ClinicalKey.es por Elsevier en abril 19, 2017.
Para uso personal exclusivamente. No se permiten otros usos sin autorizacin. Copyright 2017. Elsevier Inc. Todos los derechos reservados.
Articles
Recurrent Events Study) F Sacks, L Moy, M Pfeer; C M Hawkins, 2 Cholesterol Treatment Trialists (CTT) Collaboration. Lack of eect
E Braunwald; CORONA (Rosuvastatin in Older Patients with Systolic of lowering LDL cholesterol on cancer: meta-analysis of individual
Heart Failure) J Kjekshus, H Wedel, J Wikstrand; FIELD (Fenobrate data from 175,000 people in 27 randomised trials of statin therapy.
Intervention and Event Lowering in Diabetes) P Barter, A Keech; GISSI PLoS One 2012; 7: e29849.
(Gruppo Italiano per lo Studio della Sopravvivenza nellInfarto 3 Ray KK, Seshasai SR, Erqou S, et al. Statins and all-cause mortality
miocardico)Heart Failure L Tavazzi, A Maggioni; GISSIPrevention in high-risk primary prevention: a meta-analysis of 11 randomized
R Marchioli, G Tognoni, M G Franzosi, A Maggioni; HIT (Veteran controlled trials involving 65,229 participants. Arch Intern Med 2010;
170: 102431.
Administration Low HDL Intervention Trial) H Bloomeld, S Robins;
HPS (Heart Protection Study) R Collins, J Armitage, A Keech, S Parish, 4 Taylor F, Ward K, Moore TH, et al. Statins for the primary
prevention of cardiovascular disease. Cochrane Database Syst Rev
R Peto, P Sleight; IDEAL (Incremental Decrease in Endpoints through
2011; 1: CD004816.
Aggressive Lipid-lowering) T R Pedersen; JUPITER (Justication for the
5 Redberg RF, Katz M, Grady D. Editors Noteto make the case
Use of Statins in Prevention: an Intervention Trial Evaluating
evidence is required: comment on Making the case for selective
Rosuvastatin) P M Ridker; LDS (Lipids in Diabetes Study) R Holman; use of statins in the primary prevention setting. Arch Intern Med
LEADER (Lower Extremity Arterial Disease Event Reduction trial) 2011; 171: 1594.
T Meade; LIPID (Long-term Intervention with Pravastatin in Ischaemic 6 Kerr AJ, Broad J, Wells S, Riddell T, Jackson R. Should the rst
Disease) J Simes, A Keech, S MacMahon, I Marschner, A Tonkin, priority in cardiovascular risk management be those with prior
J Shaw; LIPS (Lescol Intervention Prevention Study) P W Serruys; MEGA cardiovascular disease? Heart 2009; 95: 12529.
(Management of Elevated cholesterol in the primary prevention Group of 7 Cholesterol Treatment Trialists (CTT) Collaboration. Protocol for a
Adult Japanese) H Nakamura; Post-CABG (Post- Coronary Artery Bypass prospective collaborative overview of all current and planned
Graft Study) G Knatterud; PPP (Pravastatin Pooling Project) C Furberg, randomized trials of cholesterol treatment regimens. Am J Cardiol
R Byington; PROSPER (Prospective Study of Pravastatin in the Elderly at 1995; 75: 113034.
Risk) P Macfarlane, S Cobbe, I Ford, M Murphy, G J Blauw, C Packard, 8 Cholesterol Treatment Trialists (CTT) Collaboration. Ecacy and
J Shepherd; 4S (Scandinavian Simvastatin Survival Study) J Kjekshus, safety of cholesterol-lowering treatment: prospective meta-analysis
T Pedersen, L Wilhelmsen; PROVE-IT (Pravastatin or Atorvastatin of data from 90 056 participants in 14 randomised trials of statins.
Evaluation and Infection Therapy) E Braunwald, C Cannon, S Murphy; Lancet 2005; 366: 126778.
SEARCH (Study of Eectiveness of Additional Reductions in Cholesterol 9 de Lemos JA, Blazing MA, Wiviott SD, et al. Early intensive vs a
and Homocysteine) R Collins, J Armitage, L Bowman, S Parish, R Peto, delayed conservative simvastatin strategy in patients with acute
P Sleight; SHARP (Study of Heart and Renal Protection) C Baigent, coronary syndromes: phase Z of the A to Z Trial. JAMA 2004;
M Landray, R Collins; TNT (Treating to New Targets) J La Rosa; WHI 292: 130716.
(Womens Health Initiative) J Rossouw, J Probsteld; WOSCOPS (West 10 Cannon CP, Braunwald E, McCabe CH, et al. Intensive versus
of Scotland Coronary Prevention Study) J Shepherd, S Cobbe, moderate lipid lowering with statins after acute coronary
syndromes. N Engl J Med 2004; 350: 1495504.
P Macfarlane, I Ford.
Other members: M Flather, J Kastelein, C Newman, C Shear, J Tobert, 11 Pedersen TR, Faergeman O, Kastelein JJP, et al. High-dose
atorvastatin vs usual-dose simvastatin for secondary prevention
J Varigos, H White, S Yusuf.
after myocardial infarction: the IDEAL study: a randomized
Observers: Bristol-Myers Squibb M Mellies, M McGovern, J Barclay, controlled trial. JAMA 2005; 294: 243745.
R Belder; Merck Y Mitchel, T Musliner; Laboratoires Fournier
12 Scandinavian Simvastatin Survival Study Group. Randomised trial
J-C Ansquer; Bayer M Llewellyn; Novartis Pharma M Bortolini; of cholesterol lowering in 4444 patients with coronary heart disease:
AstraZeneca G Brandrup-Wognsen, B Bryzinski, G Olsson, J Pears; the Scandinavian Simvastatin Survival Study (4S). Lancet 1994;
Pzer D DeMicco. 344: 138389.
CTT secretariat: C Baigent, E H Barnes, A Baxter, N Bhala, L Blackwell, 13 Shepherd J, Cobbe SM, Ford I, et al. Prevention of coronary heart
G Buck, R Collins, J Emberson, W G Herrington, L E Holland, disease with pravastatin in men with hypercholesterolemia. West of
P M Kearney, A Keech, A Kirby, D A Lewis, I Marschner, C Pollicino, Scotland Coronary Prevention Study Group. N Engl J Med 1995;
C Reith, J Simes, T Sourjina. 333: 130107.
Conicts of interest 14 The Post Coronary Artery Bypass Graft Trial Investigators. The
eect of aggressive lowering of low-density lipoprotein cholesterol
Most of the trials in this report were supported by research grants from
levels and low-dose anticoagulation on obstructive changes in
the pharmaceutical industry. Some members of the writing committee saphenous-vein coronary-artery bypass grafts. N Engl J Med 1997;
have received reimbursement of costs to participate in scientic 336: 15362.
meetings from the pharmaceutical industry. AK and JS have also 15 Downs JR, Cleareld M, Weis S, et al. Primary prevention of acute
received honoraria from Solvay for lectures related to these studies. coronary events with lovastatin in men and women with average
Acknowledgments cholesterol levels: results of AFCAPS/TexCAPS. Air Force/Texas
Coronary Atherosclerosis Prevention Study. JAMA 1998;
The Clinical Trial Service Unit & Epidemiological Studies Unit (CTSU)
279: 161522.
in the UK and the National Health and Medical Research Council
16 The Long-Term Intervention with Pravastatin in Ischaemic
Clinical Trials Centre (CTC) in Australia coordinate this collaboration
Disease Study Group. Prevention of cardiovascular events and
jointly. The present meta-analysis work is supported at the Health
death with pravastatin in patients with coronary heart disease and
Economics Research Centre, University of Oxford (writing committee a broad range of initial cholesterol levels. N Engl J Med 1998;
members B Mihaylova, A Gray) by a British Heart Foundation research 339: 134957.
grant (Grant No PG/08/063/25397), and at the CTSU (writing 17 GISSI Prevenzione Investigators. Results of the low-dose (20 mg)
committee members J Emberson, L Blackwell, R Collins, C Baigent) by pravastatin GISSI Prevenzione trial in 4271 patients with recent
the UK Medical Research Council, British Heart Foundation, Cancer myocardial infarction: do stopped trials contribute to overall
Research UK and, previously, the European Community Biomed knowledge? GISSI Prevenzione Investigators (Gruppo Italiano per
Programme. The CTC is supported by a programme grant from the lo Studio della Sopravvivenza nellInfarto Miocardico). Ital Heart J
Australian National Health and Medical Research Council, and a grant 2000; 1: 81020.
from the National Heart Foundation, Australia. JE acknowledges 18 Serruys PW, de Feyter P, Macaya C, et al. Fluvastatin for prevention
support from the BHF Centre of Research Excellence, Oxford, UK of cardiac events following successful rst percutaneous coronary
(RE/08/04). intervention: a randomized controlled trial. JAMA 2002;
287: 321522.
References 19 Heart Protection Study Collaborative Group. MRC/BHF Heart
1 Cholesterol Treatment Trialists (CTT) Collaboration. Ecacy and Protection Study of cholesterol lowering with simvastatin in
safety of more intensive lowering of LDL cholesterol: a 20 536 high-risk individuals: a randomised placebo-controlled trial.
meta-analysis of data from 170 000 participants in 26 randomised Lancet 2002; 360: 722.
trials. Lancet 2010; 376: 167081.
www.thelancet.com Vol 380 August 11, 2012 589
Descargado para Anonymous User (n/a) en ClinicalKey Espanol Columbia, Ecuador & Peru Flood Relief de ClinicalKey.es por Elsevier en abril 19, 2017.
Para uso personal exclusivamente. No se permiten otros usos sin autorizacin. Copyright 2017. Elsevier Inc. Todos los derechos reservados.
Articles
20 Shepherd J, Blauw GJ, Murphy MB, et al, on behalf of the 38 Armitage J. The safety of statins in clinical practice. Lancet 2007;
PROSPER study group. Pravastatin in elderly individuals at risk of 370: 178190.
vascular disease (PROSPER): a randomised controlled trial. Lancet 39 Davidson MH, Robinson JG. Safety of aggressive lipid management.
2002; 360: 162330. J Am Coll Cardiol 2007; 49: 175362.
21 The Allhat Ocers and Coordinators for the ALLHAT Collaborative 40 Cheung BMY, Lam KSL. Is intensive LDL-cholesterol lowering
Research Group. Major outcomes in moderately benecial and safe? Lancet 2010; 376: 162224.
hypercholesterolemic, hypertensive patients randomized to 41 Sattar N, Preiss D, Murray HM, et al. Statins and risk of incident
pravastatin vs usual care: the Antihypertensive and Lipid-Lowering diabetes: a collaborative meta-analysis of randomised statin trials.
Treatment to Prevent Heart Attack Trial (ALLHAT-LLT). JAMA 2002; Lancet 2010; 375: 73542.
288: 29983007.
42 Preiss D, Seshasai SR, Welsh P, et al. Risk of incident diabetes with
22 Sever PS, Dahlf B, Poulter NR, et al, for the ASCOT investigators. intensive-dose compared with moderate-dose statin therapy: a
Prevention of coronary and stroke events with atorvastatin in meta-analysis. JAMA 2011; 305: 255664.
hypertensive patients who have average or lower-than-average
43 The Emerging Risk Factors Collaboration. Diabetes mellitus,
cholesterol concentrations, in the Anglo-Scandinavian Cardiac
fasting blood glucose concentration, and risk of vascular disease: a
Outcomes TrialLipid Lowering Arm (ASCOT-LLA): a multicentre
collaborative meta-analysis of 102 prospective studies. Lancet 2010;
randomised controlled trial. Lancet 2003; 361: 114958.
375: 221522.
23 Holdaas H, Fellstrm B, Jardine AG, et al, on behalf of the
44 Strandberg TE, Pyorala K, Cook TJ, et al, for the 4S Group. Mortality
Assessment of LEscol in Renal Transplantation (ALERT) Study
and incidence of cancer during 10-year follow-up of the
Investigators. Eect of uvastatin on cardiac outcomes in renal
Scandinavian Simvastatin Survival Study (4S). Lancet 2004;
transplant recipients: a multicentre, randomised, placebo-controlled
364: 77177.
trial. Lancet 2003; 361: 202431.
45 LIPID Study Group. Long-term eectiveness and safety of
24 Colhoun HM, Betteridge DJ, Durrington PN, et al, on behalf of the
pravastatin in 9014 patients with coronary heart disease and average
CARDS investigators. Primary prevention of cardiovascular disease
cholesterol concentrations: the LIPID trial follow-up. Lancet 2002;
with atorvastatin in type 2 diabetes in the Collaborative Atorvastatin
359: 137987.
Diabetes Study (CARDS): multicentre randomised
placebo-controlled trial. Lancet 2004; 364: 68596. 46 Ford I, Murray H, Packard CJ, et al. Long-term follow-up of the
West of Scotland Coronary Prevention Study. N Engl J Med 2007;
25 Sacks FM, Pfeer MA, Moye LA, et al. The eect of pravastatin on
357: 147786.
coronary events after myocardial infarction in patients with average
cholesterol levels. Cholesterol and Recurrent Events Trial 47 Heart Protection Study Collaborative Group. Eects on 11 year
investigators. N Engl J Med 1996; 335: 100109. mortality and morbidity of lowering LDL cholesterol with
simvastatin for about 5 years in 20 536 high-risk individuals. Lancet
26 Wanner C, Krane V, Marz W, et al. Atorvastatin in patients with
2011; 378: 201320.
type 2 diabetes mellitus undergoing hemodialysis. N Engl J Med
2005; 353: 23848. 48 Grundy SM, Cleeman JI, Merz CNB, et al. Implications of Recent
Clinical Trials for the National Cholesterol Education Program
27 Knopp RH, dEmden M, Smilde JG, Pocock SJ. Ecacy and safety
Adult Treatment Panel III Guidelines. Circulation 2004; 110: 22739.
of atorvastatin in the prevention of cardiovascular end points in
subjects with type 2 diabetes: the Atorvastatin Study for Prevention 49 Expert Panel on Detection Evaluation and Treatment of High Blood
of Coronary Heart Disease Endpoints in non-insulin-dependent Cholesterol in Adults. Executive Summary of the Third Report of
diabetes mellitus (ASPEN). Diabetes Care 2006; 29: 147885. the National Cholesterol Education Program (NCEP) Expert Panel
on Detection, Evaluation, and Treatment of High Blood Cholesterol
28 Koren MJ, Hunninghake DB, on behalf of the AI. Clinical outcomes
in Adults (Adult Treatment Panel III). JAMA 2001; 285: 248697.
in managed-care patients with coronary heart disease treated
aggressively in lipid-lowering disease management clinics: the 50 Graham I, Atar D, Borch-Johnsen K, et al. European guidelines on
ALLIANCE study. J Am Coll Cardiol 2004; 44: 177279. cardiovascular disease prevention in clinical practice: full text.
Fourth Joint Task Force of the European Society of Cardiology and
29 Nakamura H, Arakawa K, Itakura H, et al. Primary prevention of
other societies on cardiovascular disease prevention in clinical
cardiovascular disease with pravastatin in Japan (MEGA Study): a
practice (constituted by representatives of nine societies and by
prospective randomised controlled trial. Lancet 2006; 368: 115563.
invited experts). Eur J Cardiovasc Prev Rehabil 2007;
30 Fellstrom BC, Jardine AG, Schmieder RE, et al. Rosuvastatin and 14 (suppl 2): 1113.
cardiovascular events in patients undergoing hemodialysis.
51 Reiner Z, Catapano AL, De Backer G, et al. ESC/EAS Guidelines for
N Engl J Med 2009; 360: 1395407.
the management of dyslipidaemias: the Task Force for the
31 Ridker PM, Danielson E, Fonseca FA, et al. Rosuvastatin to prevent management of dyslipidaemias of the European Society of
vascular events in men and women with elevated C-reactive protein. Cardiology (ESC) and the European Atherosclerosis Society (EAS).
N Engl J Med 2008; 359: 2195207. Eur Heart J 2011; 32: 1769818.
32 Kjekshus J, Apetrei E, Barrios V, et al. Rosuvastatin in older patients 52 Cooper A, Nherera L, Calvert N, et al. Clinical guidelines and
with systolic heart failure. N Engl J Med 2007; 357: 224861. evidence review for lipid modication: cardiovascular risk
33 Gissi-HF investigators. Eect of rosuvastatin in patients with assessment and the primary and secondary prevention of
chronic heart failure (the GISSI-HF trial): a randomised, double- cardiovascular disease. London: National Collaborating Centre for
blind, placebo-controlled trial. Lancet 2008; 372: 123139. Primary Care and Royal College of General Practitioners, 2008.
34 LaRosa JC, Grundy SM, Waters DD, et al. Intensive lipid lowering 53 Heart Protection Study Collaborative Group. Lifetime cost
with atorvastatin in patients with stable coronary disease. eectiveness of simvastatin in a range of risk groups and age
N Engl J Med 2005; 352: 142535. groups derived from a randomised trial of 20,536 people. BMJ
35 Study of the Eectiveness of Additional Reductions in Cholesterol 2006; 333: 114548.
and Homocysteine (SEARCH) Collaborative Group. Intensive 54 Lazar LD, Pletcher MJ, Coxson PG, Bibbins-Domingo K,
lowering of LDL cholesterol with 80 mg versus 20 mg simvastatin Goldman L. Cost-eectiveness of statin therapy for primary
daily in 12 064 survivors of myocardial infarction: a double-blind prevention in a low-cost statin era. Circulation 2011; 124: 14653.
randomised trial. Lancet 2010; 376: 165869. 55 Pletcher MJ, Lazar L, Bibbins-Domingo K, et al. Comparing impact
36 Amarenco P, Bogousslavsky J, Callahan A III, et al. High-dose and cost-eectiveness of primary prevention strategies for
atorvastatin after stroke or transient ischemic attack. N Engl J Med lipid-lowering. Ann Intern Med 2009; 150: 24354.
2006; 355: 54959.
37 Athyros VG, Papageorgiou AA, Mercouris BR, et al. Treatment with
atorvastatin to the National Cholesterol Educational Program goal
versus usual care in secondary coronary heart disease prevention.
The GREek Atorvastatin and Coronary-heart-disease Evaluation
(GREACE) study. Curr Med Res Opin 2002; 18: 22028.
590 www.thelancet.com Vol 380 August 11, 2012
Descargado para Anonymous User (n/a) en ClinicalKey Espanol Columbia, Ecuador & Peru Flood Relief de ClinicalKey.es por Elsevier en abril 19, 2017.
Para uso personal exclusivamente. No se permiten otros usos sin autorizacin. Copyright 2017. Elsevier Inc. Todos los derechos reservados.
You might also like
- More intensive LDL cholesterol lowering reduces vascular risksDocument12 pagesMore intensive LDL cholesterol lowering reduces vascular risksStefania CristinaNo ratings yet
- MRC-BHF Heart Protection Study PDFDocument16 pagesMRC-BHF Heart Protection Study PDFArif KurniadiNo ratings yet
- LDL cholesterol reduction effects by risk levelDocument18 pagesLDL cholesterol reduction effects by risk levelhenryweiNo ratings yet
- MRC/BHF Heart Protection Study of Cholesterol Lowering With Simvastatin in 20 536 High-Risk Individuals: A Randomised Placebocontrolled TrialDocument16 pagesMRC/BHF Heart Protection Study of Cholesterol Lowering With Simvastatin in 20 536 High-Risk Individuals: A Randomised Placebocontrolled Trialblopper123No ratings yet
- Cardiovascular Disease and Dyslipidemia: Beyond LDLDocument10 pagesCardiovascular Disease and Dyslipidemia: Beyond LDLGuillen CelisNo ratings yet
- Jurnal 4 WordDocument20 pagesJurnal 4 WordSri MaryatiNo ratings yet
- AURORA: Is There A Role For Statin Therapy in Dialysis Patients?Document4 pagesAURORA: Is There A Role For Statin Therapy in Dialysis Patients?Ravan WidiNo ratings yet
- CTTC Estatinas Lancet 2022Document14 pagesCTTC Estatinas Lancet 2022Silvia Montejo FareloNo ratings yet
- Intensive Lipid Lowering With Atorvastatin in Patients With Stable Coronary DiseaseDocument11 pagesIntensive Lipid Lowering With Atorvastatin in Patients With Stable Coronary Diseasesyaiful rinantoNo ratings yet
- High Dose Vs Low Dose Statin in MaceDocument9 pagesHigh Dose Vs Low Dose Statin in MaceErvan ZuhriNo ratings yet
- DyslipidemiaManagement Continuum 2011Document13 pagesDyslipidemiaManagement Continuum 2011Zuleika DöObsönNo ratings yet
- Optimal Low-Density Lipoprotein Is 50 To 70 MG - DL - Lower Is Better and Physiologically Normal - ScienceDirectDocument13 pagesOptimal Low-Density Lipoprotein Is 50 To 70 MG - DL - Lower Is Better and Physiologically Normal - ScienceDirectJNo ratings yet
- Benefit of Targeting A LDL (Low-Density Lipoprotein) Cholesterol 70 MG/DL During 5 Years After Ischemic StrokeDocument9 pagesBenefit of Targeting A LDL (Low-Density Lipoprotein) Cholesterol 70 MG/DL During 5 Years After Ischemic StrokeCorina DiaconescuNo ratings yet
- Association Between Baseline LDL-C Level and TotalDocument14 pagesAssociation Between Baseline LDL-C Level and TotalMr. LNo ratings yet
- Efficacy of Low-And Moderate-Intensity Statins For Achieving Low - Density Lipoprotein Cholesterol Targets in Thai Type 2 Diabetic PatientsDocument8 pagesEfficacy of Low-And Moderate-Intensity Statins For Achieving Low - Density Lipoprotein Cholesterol Targets in Thai Type 2 Diabetic Patientsnaufal12345No ratings yet
- Safety and Efficacy of Statin TherapyDocument13 pagesSafety and Efficacy of Statin TherapyAlexa ArceNo ratings yet
- Journal of Diabetes and Its ComplicationsDocument6 pagesJournal of Diabetes and Its ComplicationsNadia Rezky ElizaNo ratings yet
- Statin Therapy, Review of Safety and Potential Side EffectsDocument9 pagesStatin Therapy, Review of Safety and Potential Side EffectsDhita Dwi NandaNo ratings yet
- Levosimendan in Patients With Low Cardiac Output SyndromeDocument7 pagesLevosimendan in Patients With Low Cardiac Output SyndromeNehemias MD GuevaraNo ratings yet
- Efficacy and Safety of High-Density Lipoprotein Cholesterol-Increasing CompoundsDocument13 pagesEfficacy and Safety of High-Density Lipoprotein Cholesterol-Increasing CompoundsAdityasekar WangiNo ratings yet
- BMJ 2023 075837.fullDocument10 pagesBMJ 2023 075837.fullIgor KhedeNo ratings yet
- Effects of Niacin Combination Therapy With Statin or Bile-Acid Resin On Lipoproteins and Cardiovascular DiseaseDocument10 pagesEffects of Niacin Combination Therapy With Statin or Bile-Acid Resin On Lipoproteins and Cardiovascular Diseaselina budiartiNo ratings yet
- Measuring small, dense LDL cholesterolDocument2 pagesMeasuring small, dense LDL cholesterolNanda Nabilah UbayNo ratings yet
- Barter (2007) HDL Cholesterol, Very Low Levels of LDLDocument10 pagesBarter (2007) HDL Cholesterol, Very Low Levels of LDLBruno TeixeiraNo ratings yet
- Statins in Renal Disease - AndreaDocument18 pagesStatins in Renal Disease - AndreaNawel BenNo ratings yet
- European Journal of Preventive Cardiology-2016-Karlson-744-7 PDFDocument4 pagesEuropean Journal of Preventive Cardiology-2016-Karlson-744-7 PDFIrina Cabac-PogoreviciNo ratings yet
- New England Journal Medicine: The ofDocument13 pagesNew England Journal Medicine: The ofStefania CristinaNo ratings yet
- Pi Is 1933287417302908Document11 pagesPi Is 1933287417302908Anonymous yptjlwJNo ratings yet
- Intensive Statin Therapy - A Sea Change in Cardiovascular PreventionDocument3 pagesIntensive Statin Therapy - A Sea Change in Cardiovascular Preventionsyaiful rinantoNo ratings yet
- Kontras StatinDocument10 pagesKontras Statinale_rhdNo ratings yet
- Rosuvastatia y PCR en ScaDocument3 pagesRosuvastatia y PCR en ScaWilliankmachoNo ratings yet
- LDL Cholesterol Reduction and Cardiovascular Outcomes with High-Intensity Statin TherapyDocument7 pagesLDL Cholesterol Reduction and Cardiovascular Outcomes with High-Intensity Statin TherapyWanda Novia P SNo ratings yet
- CEAAAADocument12 pagesCEAAAAfikebatuNo ratings yet
- Yu Pan, Li Li Guo, Ling Ling Cai, Xiao Juan Zhu, Jin Lian Shu, Xiao Li Liu and Hui Min JinDocument8 pagesYu Pan, Li Li Guo, Ling Ling Cai, Xiao Juan Zhu, Jin Lian Shu, Xiao Li Liu and Hui Min JinrosalinmarufNo ratings yet
- New England Journal Medicine: The ofDocument10 pagesNew England Journal Medicine: The ofAlina PirtacNo ratings yet
- Document PDFDocument12 pagesDocument PDFnikenauliaputriNo ratings yet
- How Statin Therapies Impact Cholesterol Levels in WomenDocument7 pagesHow Statin Therapies Impact Cholesterol Levels in WomenFa'iq Hanif MubarokNo ratings yet
- Jos 2021 01249jdfhjdsDocument13 pagesJos 2021 01249jdfhjdsAnonymous tG35SYROzENo ratings yet
- Abstracts - How To Critically Appraise From An AbstractDocument3 pagesAbstracts - How To Critically Appraise From An AbstractآكوجويNo ratings yet
- Blood Pressure and Cholesterol Lowering in Persons Without Cardiovascular DiseaseDocument46 pagesBlood Pressure and Cholesterol Lowering in Persons Without Cardiovascular DiseaseJirran CabatinganNo ratings yet
- Guidelines Made Simple Tool 2018 CholesterolDocument22 pagesGuidelines Made Simple Tool 2018 CholesterolSafira HalimatussadiahNo ratings yet
- A Clinical Evaluation of Statin Pleiotropy: Statins Selectively and Dose-Dependently Reduce Vascular InflammationDocument9 pagesA Clinical Evaluation of Statin Pleiotropy: Statins Selectively and Dose-Dependently Reduce Vascular InflammationzzakieNo ratings yet
- Optimal Low-Density Lipoprotein Is 50 To 70 Mg:Dl... 1Document1 pageOptimal Low-Density Lipoprotein Is 50 To 70 Mg:Dl... 1Odessa FileNo ratings yet
- Estatinas Riesgo y BeneficioDocument4 pagesEstatinas Riesgo y BeneficioHamandita MoraNo ratings yet
- Guidelines Made Simple Tool 2018 CholesterolDocument22 pagesGuidelines Made Simple Tool 2018 CholesterolHussain HameedNo ratings yet
- Effect of Statin Therapy On Muscle Symptoms Estudo CTTDocument14 pagesEffect of Statin Therapy On Muscle Symptoms Estudo CTTjoseNo ratings yet
- 2018 Cholesterol Guidelines Made Simple ToolDocument22 pages2018 Cholesterol Guidelines Made Simple ToolLeoberto Batista Pereira SobrinhoNo ratings yet
- Effects of Anacetrapib in Patients With Atherosclerotic Vascular DiseaseDocument11 pagesEffects of Anacetrapib in Patients With Atherosclerotic Vascular DiseaseCirca NewsNo ratings yet
- Jurnal CardioDocument7 pagesJurnal CardioJicko Street HooligansNo ratings yet
- Prevalence of Anemia and Dyslipidemia in Patients With Diabetes Mellitus Type 2Document5 pagesPrevalence of Anemia and Dyslipidemia in Patients With Diabetes Mellitus Type 2LokiCukiNo ratings yet
- Journal Pre-Proof: Clinical Lymphoma, Myeloma and LeukemiaDocument20 pagesJournal Pre-Proof: Clinical Lymphoma, Myeloma and LeukemiaYakobus Antonius SobuberNo ratings yet
- Statin Toxicity: ReviewDocument23 pagesStatin Toxicity: ReviewRico AditamaNo ratings yet
- Effectiveness of Statin Therapy in Ischemic Post Stroke PatientsDocument6 pagesEffectiveness of Statin Therapy in Ischemic Post Stroke Patientsvhania baniNo ratings yet
- PLATO糖尿病Document11 pagesPLATO糖尿病陈诗哲No ratings yet
- Baseline Low-Density LipoproteinDocument9 pagesBaseline Low-Density LipoproteinjoNo ratings yet
- New England Journal Medicine: The ofDocument11 pagesNew England Journal Medicine: The ofGonzalo LealNo ratings yet
- Gender Difference in Coronary Events in Relation To Risk Factors in Japanese Hypercholesterolemic Patients Treated With Low-Dose SimvastatinDocument5 pagesGender Difference in Coronary Events in Relation To Risk Factors in Japanese Hypercholesterolemic Patients Treated With Low-Dose SimvastatinasfwegereNo ratings yet
- Objective:: BackgroundDocument23 pagesObjective:: BackgroundJanine DimaangayNo ratings yet
- Complementary and Alternative Medical Lab Testing Part 3: CardiologyFrom EverandComplementary and Alternative Medical Lab Testing Part 3: CardiologyRating: 1 out of 5 stars1/5 (1)
- STE English Grade 10 Worksheet PracticeDocument2 pagesSTE English Grade 10 Worksheet PracticeAlbert ValezaNo ratings yet
- Daily Medical Report Laporan Harian MedikDocument18 pagesDaily Medical Report Laporan Harian MedikLehman ManNo ratings yet
- Article HealthDocument3 pagesArticle Healthkaii cutieNo ratings yet
- ICD 10 kEDOKTERAN GIGIDocument3 pagesICD 10 kEDOKTERAN GIGIninaainidewiNo ratings yet
- Non-invasive Ventilation Techniques for Respiratory FailureDocument58 pagesNon-invasive Ventilation Techniques for Respiratory FailuresachinNo ratings yet
- Antidiabetic Activity of Insulin Plant (Costus Igneus) Leaf Extract in Diabetic RatDocument5 pagesAntidiabetic Activity of Insulin Plant (Costus Igneus) Leaf Extract in Diabetic RatKhaleel BashaNo ratings yet
- BN1151 Lecture On Nursing Care Plan and Nursing Diagnosis Planning Implementation and Evaluation 0Document48 pagesBN1151 Lecture On Nursing Care Plan and Nursing Diagnosis Planning Implementation and Evaluation 0Steven LeungNo ratings yet
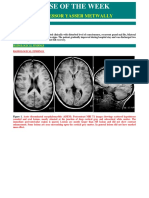
- Professor Yasser Metwally: Clinical PictureDocument14 pagesProfessor Yasser Metwally: Clinical PictureSabrin BadarneNo ratings yet
- Case Investigation Form for AEFIDocument4 pagesCase Investigation Form for AEFICHICKYNo ratings yet
- COVID Instant Order-62Document3 pagesCOVID Instant Order-62SanjayKumarReddyNo ratings yet
- NCM116 - Metabolic - Endocrine DisordersDocument24 pagesNCM116 - Metabolic - Endocrine DisordersDan Hizon100% (1)
- Monteggia FractureDocument11 pagesMonteggia FractureRonald Ivan WijayaNo ratings yet
- Welcome To Deped TV Mapeh 8 Class: I Am Aprilyn Grace Ganado ofDocument42 pagesWelcome To Deped TV Mapeh 8 Class: I Am Aprilyn Grace Ganado ofAprilyn Grace GanadoNo ratings yet
- Final OutlineDocument2 pagesFinal Outlineapi-302651894No ratings yet
- UrolithiasisDocument29 pagesUrolithiasis143bbNo ratings yet
- MAJ 19 w2Document6 pagesMAJ 19 w2Clinton D. ZamoraNo ratings yet
- Risk For Impaired Skin IntegrityDocument4 pagesRisk For Impaired Skin IntegrityArelys Rodriguez100% (2)
- Thyrotoxicosis: By: DR Mukesh Kumar Samota PG (M.D. Medicine) Medical College Jhalawar (RajasthanDocument40 pagesThyrotoxicosis: By: DR Mukesh Kumar Samota PG (M.D. Medicine) Medical College Jhalawar (RajasthanShravani ShagapuramNo ratings yet
- Self-Directed Learning (Nur 146 - Clinical Area)Document2 pagesSelf-Directed Learning (Nur 146 - Clinical Area)Dyan Bianca Suaso LastimosaNo ratings yet
- Management of Hyperkalaemia and HypokalaemiaDocument21 pagesManagement of Hyperkalaemia and Hypokalaemiaferoza97No ratings yet
- Parkinson's Disease and Peripheral Neuropathy Case StudiesDocument20 pagesParkinson's Disease and Peripheral Neuropathy Case StudiessharenNo ratings yet
- 2 COVID-19 Survivors' Psychological Health An Exploratory StudyDocument20 pages2 COVID-19 Survivors' Psychological Health An Exploratory StudyApplied Psychology Review (APR)No ratings yet
- NailSurgery With Name ChangesDocument55 pagesNailSurgery With Name ChangesIde Bagoes InsaniNo ratings yet
- Sample Questions For HAAD Prometric and DHA For Nurses PDFDocument23 pagesSample Questions For HAAD Prometric and DHA For Nurses PDFAasmeetaa Thapa100% (2)
- Syndromic Gastric PolypsDocument15 pagesSyndromic Gastric PolypsTheDrakairNo ratings yet
- CHN-2.5, KMU Past PaperDocument14 pagesCHN-2.5, KMU Past PaperGulshad AfridiNo ratings yet
- High Heels Shorten Women's Leg Muscles: English Workshop Let's Work 10ºDocument3 pagesHigh Heels Shorten Women's Leg Muscles: English Workshop Let's Work 10ºJose MiguelNo ratings yet
- Gelastic and Cursive Seizures - Neurology - 1973Document11 pagesGelastic and Cursive Seizures - Neurology - 1973---No ratings yet
- Pancreatitis - Powerpoint PresentationDocument43 pagesPancreatitis - Powerpoint PresentationHamid Colangcag AmborNo ratings yet
- 4 HIV AIDS Workplace Policy & Program-1Document4 pages4 HIV AIDS Workplace Policy & Program-1Osfmc LogisticsNo ratings yet