Professional Documents
Culture Documents
Why Doesn T The Electron Fall Into The Nucleus
Uploaded by
VanessaLassoOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Why Doesn T The Electron Fall Into The Nucleus
Uploaded by
VanessaLassoCopyright:
Available Formats
Why Doesn't the Electron Fall into the Nucleus?
Franklin P. Mason and Robert W. Richardson
San Jose State University, San Jose, CA 95192
A question that must come to the mind of every beginning
chemistry student is: "If the electron and the nucleus of an
atom are oppositely charged particles, why doesn't the elec terms.
tron fall into the nucleus?" In order to ward off a question of
such complexity, an instructor (although disclaiming the Qualitative Presentation
answer while eivine it) might say: "Because the electron is The factor that tends to bring the electron and nucleus to-
moving aroundthe ikcleus as a moves around the sun." gether is, of course, electrostat& attraction.
Of course, it is known that this answer is hopelessly naive. For The factor that tends to make the electron and nucleus
one thing, while in a 1s orbital, the electron has no orbital move apart must now be discussed. Consider an otherwise
angular momentum ( I = O), and therefore, cannot he "moving empty box containing one electron. All particles, including
around" anything in the usual sense of the word. And for an- electrons, display wave as well as particle properties. When
other, a charged particle such as an electron, when it is ac- an electron is contained in any way, for example in a box, its
celerated, as it would be if it were in orbit around a nucleus, . .
wave ~ronerties become verv.imnortant.
. When contained. the
radiates energy. This would cause the electron to spiral into electron becomes a "particle in a box," that is, it resembles a
the nucleus. not stav awav from it. This naoer nresents a three-dimensional standing wave of matter and electrical
simple, yet essentialry corr& model of theitom that can be charge (which has been calied an electron c l o ~ d ) . ~
-
used to answer the above auestion even for beeinninr - students Associated with anv particle in a box is a certain enerev (or
of chemistry. set of energy levels) which can he called the "energy orcon-
Even thouah - a ulanetarv
. model of the atom can no longer "
tainment" of the particle. All particles have associated with
he used, a clue can he taken from it for a new answer to the them a certain wavelength. When a particle is confined in a
question. As is well known, for stahle planetary motion there limited volume, its maximum wavelength is dictated hv the
are two forces at play which must balance each other. The size of the container. As the containerbecomes smaller, the
tendency of a planet to fall into the sun (because of gravita- wavelength must also become shorter. As the wavelength
tional attraction) must be balanced by the opposite tendency becomes shorter, the energy of the particle increases. This, in
for the planet to leave the sun (because of its forward motion). brief, is the source of the energy of containment.
For stable planetary motion the "force in" equals the "force
out," and for gravitational or electric attraction the potential
energy is twice the magnitude of the kinetic energy and op- ' This relationship, which is true for all inverse square force laws
posite in sign (2KE = -PE) ( I 1.' in both quantum mechanics and classical mechanics, is an example
of the virial theorem which is also true for many particle systems.
The model of the atom uresented here is formulated bv In calculating the energy of a multi-electron atom, the point-charge
analogy to stable planetary motion. In this model two factors model and not the electron-cloud model must be used.
are identified, one that tends to bring the electron closer to In the Schroedinger formulationof quantum mechanics, the "energy
the nucleus and one that tends to make the electron escape of containment" appears as the kinetic energy, which is proportional
from the nucleus. This model will first be discussed in quali- to the curvature of the wave function.
40 Journal of Chemical Education
where: F = force of attraction, Ze = charge on the nucleus in
esu, e = charge on the electron in esu, and r = distance sepa-
rating the particles. From this force a potential energy term
can be derived. For "r" a suitable average must be used (see
below)
where: E , = electrostatic potential energy, r,,, = average
value of r.
The factor that tends to make the electron escape from the
nucleus is the energy of containment, Ec,, (2). The energy of
containment of any particle depends only upon the mass of
the particle and the shape and size of the box in which it is
. ~ an electron in a 1s orbital%f the hvdroeen atom
c ~ n f i n e dFor
(the simplest case) the confining "box" is spherically sym-
metrical. Thus, to find the energy of containment for an
electron of a hydrogen-like atom, s&tions must be found for
the energy of a particle of mass = m e in a spherical box. The
solutions to the Schroedinger equation for a particle in a
spherical box can be quite complex. Fortunately, however, the
solutions for radially symmetrical cases (s states) are easily
obtained and, in fact, turn out to be identical to those for a
particle in a one-dimensional box ( 3 ) .For radially symmetrical
states, the containment energy of a particle in a spherical box
is
Diameter ( A )
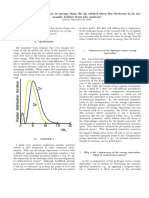
Figure 1. Wave functionsfor n = 1. 2,3, and 4. where: n = the number of waveleneths in the distance 2a, h
= Planck's constant, rn = mass o f i h e electron, and a = di-
ameter of the containing sphere. The wave functions for such
a system are well known(2) and are shown for n = 1,2,3,and
4 in Figure I
his-model suggests that the atom is stable because there
Merely confining an electron to a certain, restricted volume is an equilibrium established between the tendency of the
endows it with this energy of containment. If the electron were electron to be drawn to the nucleus due to electrostatic at-
not confined, it would have a lower energy. Therefore, the traction and the tendency of the electron to leave the nucleus
electron seeks to escape its confinement. Thus, the contain- due to its desire to escaue its confinement. For this model. the
ment energy is the factor that makes the electron of an atom total energy of the atom, E T is~ ~
tend to escaue from its nucleus.
When we add a nucleus to the box, which already contains
an electron, the electrostatic attraction lowers the potential
energy, and the total energy becomes a combination of this
wotential enerav -. and the containment enerev -- (kinetic en- nZh"e2
E.Tot=---
kg~). 8ma2 r,,,
In light of this model for the atom, the students' original
where "r,,," should be some average value between zero7 and
question can now be answered as follows:
An atom is made up of a positively charged nucleus and one or more We assume here that the barner to escape from the box is infinite,
negatively charged electrons around the nucleus. Since the nucleus but that within the box there are no forces llmiting the position of the
and the electron are of different charges, naturally they attract one particle.
another and tend to come together. However, an electron is not only The word "orbital," introduced by Mulliken, is used to describe
a particle; hut also it behaves as a wave. Because this is true, we can
visualize the electron not as a discrete particle, but rather as a diffuse individual wave function solutions to the Schroedinger equation.
For a onedimensional box, the equation is obtained as follows:
cloud af matter and electrical charge around the nucleus. As the
electron and nucleus come closer together that is, as the electron is
confinedmore and more (and the electran cloud becomes smaller and
smaller), the wavelength of the electron must become shorter and
shorter so that it can "fit" into the confining space. As its wavelength Position of nucleus
becomes shorter, the electron's,energy increases. Eventually, the
energy possessed by the electron due to its confinement becomes
sufficiently great that the electrostaticpotential energy can no longer Electron wave for n = 1
continue to draw the electron and nucleus closer together. At that
point an equilibrium is established in which the atom is stable and
in which the electron neither goes toward the nucleus (electron cloud
becoming smaller) nor does it escape from the nucleus (electron cloud
becoming larger).
Quantitative Presentation To give the maximum electron density at the nucleus, "a" must be equal
The factor that tends to bring the electron and nucleus to- to 112. Then p = h l k KE = p2/2m = V/2mh2 = V/8ma2
gether is electrostatic attraction It may be noticed that at r,,, = 0 it appears the potential energy
term becomes infinite. Because of the uncertainty principle, however,
r,, can never be equal to zero and therefore the potential energy term
cannot become infinite.
Volume 60 Number 1 January 1983 41
Energy of Containment (Eco.), Potential Energy (E.), and Total
Energy (ETO1)lor an electron in a spherical box of djameter a, in
which there is a central charge of + l .
of the containing sphere for the radially symmetrical case of
t h e n = 1state of a hydrogen-like atom (Z = 1). This data is
plotted in Figure 2.
The point of minimum energy in the ET", curve is obtained
by setting the first derivative of eqn. 6 equal to zero."
dETOi - 2 r ~ % ~
-=-
dr 8ma3
+-8Ze2
3a2
(7)
Solving for a, we obtain:
F o r n = l a n d Z = l , a =1.963A.
Thus, indeed this very simple model yields a stable atom.'O
Figure 2. Plot for a hydrogen-like atom ( 2= 1).
There is a minimum in the E T curve, ~ ~ and a t the min-
imum:
aI2. This average value can be approximated as 318 a which
now gives Emt in terms of "a" only as: The model predicts that for a stable hydrogen-like atom in its
Is state, the electron cloud will be 100%contained in a sphere
of 1.963 A in diameter, and that the ionization potential for
this atom will he 9.8 eV. These values compare reasonably well
For the model to yield a stahle atom there must be a radius with those obtained in the exact solution of the hydrogen atom
of the confining sphere a t which E.rotis a t a minimum and a t (5) for which the 1s electron is 71.7%11 contained within a
which: sphere of 1.963 A diameter and has an ionization potential of
13.6 eV.
2 Ec." = -ED It should be noted that a "box" is not necessary to have
In the Table are listed Ec,,,, Ep, and ErOtfor various radii confinement energy. For example, if a wave function equal to
N[exp - rla] is chosen and the variation principle applied to
find "a," the exact enerev is obtained. This is so because a
function has been c h o s e z h a t is ofthe exact form as the true
The average distance of points (and thus of electron charge density ground-state function. In this case, the walls of the "box" fade
of the "smeared-out" particie-in-the-box) from the center of the sphere
is 314 R or 318 D. away gradually. The present model is based upon a spherical
box because it is more easily visualized than a "box" with no
sharp walls. Thus, some accuracy is sacrificed in order to have
a model that students will be more readily able to visualize.
Acknowledgment
Franklin Mason wishes to thank Dr. Larry Domash for his
This is an example of the famousvariation principle of quantum inspiration, Professor Gordon G. Goles for his encouragement,
mechanics which states that approximate wave functions always give and especially Professor John A. Neptune who made it pos-
a higher energy than the true ground-state wave function. This allows sible.
a determination of which appr&mate wave function is best and is the
basis of much theoretical chemistrv.
" S m e n = 1 s Jied. ln.5 . j inc lo\restenerg, state poss ole for
Ine s)slem. I Inerrlore canna lose an) 1-nher encrg) o, ma at on an0 .""-,,.". .,.
1n.S inc e cclron canno: sp f a nlo l o r n.cle-s 12) For anexample of ihiiconcept ofi'Energy u l c o n f a i n m e n t ~ S e eLey~Km.E.,andHu-
" Tn s La Le s oola ne3 oy ntegratmg me proom r) oensi? fmaion binstein,S., J. i h ~ n t Phy,
. .71,353 (19791.
of the electron in a given spherical volume element: (S) R a m D.,"Quantum iMechmics/ Holt. RinehsrL and Wmstun. Inc.. New Y w k , 1971.
pp. 22PrZB.
(41 Piuling, L.. and Wilson, E. B., "lntmduciion to quvntum Mechanics." McGrav~H~lI.
New York, 1915, p. 97.
(51 Pnuling. L., and Wiisun, E. B., "Intr,,d"ction to qaantum Mechanics." ChaptPi 5.
(fir See Padin:, L.. alid Wilson, F. R., "Introduction to Quantum Mochanlcs." pp. 13%
between zero and 1.96312 A (6). 40.
42 Journal of Chemical Education
You might also like
- Thomson Atom Model: Physics NotesDocument13 pagesThomson Atom Model: Physics NotesEs ENo ratings yet
- Unit 3 - MSCDocument22 pagesUnit 3 - MSCAndroid DotNo ratings yet
- Atomic Structure 4.1 The Nuclear AtomDocument12 pagesAtomic Structure 4.1 The Nuclear AtomMat Mattt100% (1)
- NCERT PUNCH Chemistry Class 11 Complete Book Flattened SignedDocument304 pagesNCERT PUNCH Chemistry Class 11 Complete Book Flattened Signedsd0806787No ratings yet
- Barandes On Magnetic Forces and WorkDocument7 pagesBarandes On Magnetic Forces and Workdavid zilbermanNo ratings yet
- ATOM2Document13 pagesATOM2linkgogo69No ratings yet
- Orbital EnergyDocument3 pagesOrbital EnergyaneNo ratings yet
- Plasma OscillationDocument5 pagesPlasma OscillationMuhammad AkramNo ratings yet
- 무기화학 1 ch2Document5 pages무기화학 1 ch2최마리아No ratings yet
- Chem3420 Topic3 5 New2Document28 pagesChem3420 Topic3 5 New2justinhadinata283No ratings yet
- Atom 33Document18 pagesAtom 33linkgogo69No ratings yet
- General Chemistry 1: Module 4, Lesson 1: Quantum Mechanical Model of An AtomDocument4 pagesGeneral Chemistry 1: Module 4, Lesson 1: Quantum Mechanical Model of An AtomKeano GelmoNo ratings yet
- Chapter 1 Electronic Structure of AtomsDocument6 pagesChapter 1 Electronic Structure of AtomsHong Hong WongNo ratings yet
- Quantum TheoryDocument12 pagesQuantum TheorySameer KumarNo ratings yet
- Electrical Conductivity Notes - Unit-4 (March-2022)Document19 pagesElectrical Conductivity Notes - Unit-4 (March-2022)mandar desurkarNo ratings yet
- Chemistry For EngineersDocument152 pagesChemistry For Engineersmacky 2No ratings yet
- SEM 4 Nuclear Physics Part 2Document5 pagesSEM 4 Nuclear Physics Part 2Anil HirparaNo ratings yet
- Dressing Asdas PDFDocument8 pagesDressing Asdas PDFGovardhan ThammisettiNo ratings yet
- Chapter 2 Structure of AtomDocument2 pagesChapter 2 Structure of Atomgibinshaji2No ratings yet
- A Me H E: 3.4. The Quantum Structure of Atoms 115Document3 pagesA Me H E: 3.4. The Quantum Structure of Atoms 115MatteoPisanoNo ratings yet
- Final Exam Study GuideDocument43 pagesFinal Exam Study GuideAndrés Ignacio Irribarra RamírezNo ratings yet
- AbhishekDocument11 pagesAbhishekNaresh kumar ChauhanNo ratings yet
- Energy Chem Summary - Libre TextsDocument4 pagesEnergy Chem Summary - Libre Textsmacky 2No ratings yet
- Is Classical Electrodynamics AN Internally Inconsistent Theory?Document19 pagesIs Classical Electrodynamics AN Internally Inconsistent Theory?fersoto07No ratings yet
- Semiconductor Cheat Sheet Ver 1Document18 pagesSemiconductor Cheat Sheet Ver 1testerJesterNo ratings yet
- Chapter 2: The Importance ofDocument8 pagesChapter 2: The Importance ofProsanta Kumar BiswasNo ratings yet
- Chapter 5 Nuclear ForcesDocument12 pagesChapter 5 Nuclear ForcesKy KetsukiNo ratings yet
- General Chemistry: Use Quantum Numbers To Describe An Electron in An AtomDocument16 pagesGeneral Chemistry: Use Quantum Numbers To Describe An Electron in An AtomDenver John Caloza LamarcaNo ratings yet
- Basics of Atoms, Molecules and Solids: Dr. Rajan Pandey Associate Professor, SENSEDocument19 pagesBasics of Atoms, Molecules and Solids: Dr. Rajan Pandey Associate Professor, SENSEDEEPIKA PAVUNDOSS 20BEC0285No ratings yet
- Como o Eltron Passa Atravs Dos NSDocument2 pagesComo o Eltron Passa Atravs Dos NSMeus filhos Evellyn e EnzoNo ratings yet
- Structure of AtomDocument8 pagesStructure of AtompreetikoravNo ratings yet
- Introduction To Plasma Physics: Plasma Types and DefinitionsDocument15 pagesIntroduction To Plasma Physics: Plasma Types and DefinitionsNelly Milagros Esperilla LupoNo ratings yet
- Of Electron Interactions. Interactions : Collective Description MagneticDocument10 pagesOf Electron Interactions. Interactions : Collective Description MagneticSandeep BorgaonkarNo ratings yet
- Unit - 8 AtomsDocument9 pagesUnit - 8 AtomsSahil Chawla100% (1)
- Chemistry Questions OnlyDocument66 pagesChemistry Questions OnlyBHUVAN I (RA2111003011433)No ratings yet
- Krane NotesDocument14 pagesKrane NotesSubhasis PandaNo ratings yet
- The Atomic Construct A DiscussionDocument17 pagesThe Atomic Construct A DiscussionBranko R BabicNo ratings yet
- Bohr's Model, Energy Bands, Electrons and Holes: Dual Character of Material ParticlesDocument30 pagesBohr's Model, Energy Bands, Electrons and Holes: Dual Character of Material ParticlespurseyNo ratings yet
- Fine-Structure Constant - WikipediaDocument16 pagesFine-Structure Constant - WikipediaAnkit KambleNo ratings yet
- Atomic Orbital: 1 Electron PropertiesDocument15 pagesAtomic Orbital: 1 Electron PropertiesMMGNo ratings yet
- BSC VI Sem Physics Paper-I Unit-III Nuclear PhysicsDocument34 pagesBSC VI Sem Physics Paper-I Unit-III Nuclear PhysicsAnil KumarNo ratings yet
- PB - Device Physics 2022 Lecture 1Document27 pagesPB - Device Physics 2022 Lecture 1keke renNo ratings yet
- MIT6 007S11 Lec39Document25 pagesMIT6 007S11 Lec39THARMARAJNo ratings yet
- Theoretical Justification of Madelung's Rule: Pan WongDocument4 pagesTheoretical Justification of Madelung's Rule: Pan WongEduardo CostaNo ratings yet
- What is Charge? – The Redefinition of Atom - Energy to Matter ConversionFrom EverandWhat is Charge? – The Redefinition of Atom - Energy to Matter ConversionNo ratings yet
- ChemistryDocument188 pagesChemistryCricwiz 10No ratings yet
- Chapter 1 Basics of Semiconductor Physics b2 Ec 19Document48 pagesChapter 1 Basics of Semiconductor Physics b2 Ec 19Venkata Ashok Naidu SingamsettyNo ratings yet
- Solid State Physics: Module 1 Part-Ii Lecture 4 PH101 AUTUMN 2020Document35 pagesSolid State Physics: Module 1 Part-Ii Lecture 4 PH101 AUTUMN 2020Rohith RohanNo ratings yet
- Atomic Structure... (3) PPTDocument32 pagesAtomic Structure... (3) PPTVaibhav KargetiNo ratings yet
- Hsslive - Plus Two Chapter 12-2024Document7 pagesHsslive - Plus Two Chapter 12-202416739No ratings yet
- PHS 304 (Modern)Document179 pagesPHS 304 (Modern)Eniibukun Keji-ayodejiNo ratings yet
- Mt-201Electronic Conduction2023Document150 pagesMt-201Electronic Conduction2023RashmiNo ratings yet
- Quantum Mechanics NotesDocument42 pagesQuantum Mechanics Notessimonm_92No ratings yet
- Electrons Matter Interaction PDFDocument18 pagesElectrons Matter Interaction PDFAbhilash VermaNo ratings yet
- Chapter1 Basics of Semiconductor Physics PDFDocument48 pagesChapter1 Basics of Semiconductor Physics PDFJ Vikas100% (1)
- Atomic StructureDocument21 pagesAtomic StructureShahriar ShafiNo ratings yet
- EdcDocument135 pagesEdcamartya mukherjeeNo ratings yet
- Gamma Ray Spectroscopy VerDocument12 pagesGamma Ray Spectroscopy VerLiberata MigloriaNo ratings yet
- Relacion de Normas DVS PDFDocument8 pagesRelacion de Normas DVS PDFNeto Paucar ChancaNo ratings yet
- Standard Maxwell-Boltzmann Distribution - Definition and PropertiesDocument16 pagesStandard Maxwell-Boltzmann Distribution - Definition and PropertiesHugo HernandezNo ratings yet
- Bibliography: 1. NCERT Text Book 3. Cbse - Nic.inDocument19 pagesBibliography: 1. NCERT Text Book 3. Cbse - Nic.inYashwanth RameshNo ratings yet
- Classical Mechanics Arya 2nd EdDocument719 pagesClassical Mechanics Arya 2nd EdMaria HeloísaNo ratings yet
- © 2009 ANSYS, Inc. All Rights Reserved. ANSYS, Inc. ProprietaryDocument16 pages© 2009 ANSYS, Inc. All Rights Reserved. ANSYS, Inc. ProprietaryiitdvivNo ratings yet
- What Is The EM SpectrumDocument13 pagesWhat Is The EM SpectrumJones JonnyNo ratings yet
- Lab Report On Solid Liquid Equilibrium in A Binary SystemDocument7 pagesLab Report On Solid Liquid Equilibrium in A Binary SystemAyaz BhattiNo ratings yet
- Arab Oil Naturalgas - Com Crude Oil Storage TanksDocument3 pagesArab Oil Naturalgas - Com Crude Oil Storage TanksWael_Barakat_31790% (1)
- Long Range Forecasting of Low RainfallDocument8 pagesLong Range Forecasting of Low RainfallAssem AbozeedNo ratings yet
- Gs 31Document7 pagesGs 31memep1No ratings yet
- Simple Machines (p288) : Velocity Ratio VR S / SDocument8 pagesSimple Machines (p288) : Velocity Ratio VR S / SParamaet TamNo ratings yet
- Magnetism and ElectromagnetismDocument8 pagesMagnetism and ElectromagnetismArvin Palma Dela RocaNo ratings yet
- Aerodynamics of Wind Turbines: Part - 3Document37 pagesAerodynamics of Wind Turbines: Part - 3John TauloNo ratings yet
- Course Expert: Prof. Kiran Malipatil, KLECET, Belagavi Course Code: 18CV42 Course Name: Analysis of Determinate Structures Module: 03 and 04Document1 pageCourse Expert: Prof. Kiran Malipatil, KLECET, Belagavi Course Code: 18CV42 Course Name: Analysis of Determinate Structures Module: 03 and 04Chidananda GNo ratings yet
- RHC Fixed Beam Shear+moment+deflection CheckerDocument1 pageRHC Fixed Beam Shear+moment+deflection Checkerpobre3nga3bataNo ratings yet
- High Temperature CoatingsDocument2 pagesHigh Temperature CoatingsVinay TungeNo ratings yet
- Jurnal Nilai K Untuk Propilen OksidaDocument5 pagesJurnal Nilai K Untuk Propilen OksidaWidya Isti AriantiNo ratings yet
- Heat Exchangers (Shell & Tube)Document29 pagesHeat Exchangers (Shell & Tube)babak mirNo ratings yet
- (Paper) Correlation Between Low Strain Shear Modulus and Standard Penetration Test N' ValuesDocument11 pages(Paper) Correlation Between Low Strain Shear Modulus and Standard Penetration Test N' ValuesJOHANN STIVEN OCAMPO GARCIANo ratings yet
- Department of Education: I. II. Iii. IV. A. BDocument2 pagesDepartment of Education: I. II. Iii. IV. A. BLuz Marie CorveraNo ratings yet
- Tutorial The New PEMFC Model: SolutionDocument49 pagesTutorial The New PEMFC Model: Solutionmjvaze100% (6)
- Booklet 2Document7 pagesBooklet 2Jherico Jay SantosNo ratings yet
- Robins MagnusDocument9 pagesRobins MagnusAbdurrahman Abu AdibNo ratings yet
- 314 Tutorial Questions 9th EditionDocument12 pages314 Tutorial Questions 9th EditionAbdulrahim SegirNo ratings yet
- Duaso ElementsDocument202 pagesDuaso ElementsAnonymous hndZWISrhYNo ratings yet
- Physics ProjectDocument16 pagesPhysics ProjectSukalpo BanerjeeNo ratings yet
- Troubleshooting and Mitigating Gels in Polyethylene Film ProductsDocument24 pagesTroubleshooting and Mitigating Gels in Polyethylene Film ProductsTHEREZA TORRES BLANCASNo ratings yet
- Question Paper FIITJEE AITS MAINS - 2017Document22 pagesQuestion Paper FIITJEE AITS MAINS - 2017Vishesh Khandelwal75% (8)
- COMEDK Important Topics and Revised SyllabusDocument49 pagesCOMEDK Important Topics and Revised SyllabusVishal VermaNo ratings yet
- Nelson Mandela Secondary School Science Syllabus 5124 OutlineDocument4 pagesNelson Mandela Secondary School Science Syllabus 5124 OutlineMapalo Chirwa100% (1)