Professional Documents
Culture Documents
Unit 1 Courselink Notes
Uploaded by
Lauren Stam0 ratings0% found this document useful (0 votes)
15 views27 pagesOriginal Title
Unit 1 Courselink Notes.docx
Copyright
© © All Rights Reserved
Available Formats
DOCX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
15 views27 pagesUnit 1 Courselink Notes
Uploaded by
Lauren StamCopyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
You are on page 1of 27
Unit 1: The Endocrine System I
In Greek, “Endon” means within, and "Krinein" means to secrete.
The endocrine system is one of the body's two regulatory systems, while the other being the nervous system.
The endocrine system consists of endocrine glands that secrete hormones.
What are hormones?
Hormones are chemical messengers that’re secreted from endocrine glands or tissues, and exert
physiological control in target cells.
Three types of hormones include:
Endocrine hormones that are secreted into the blood stream and transported to target cells where it
produces its physiological effect.
Autocrine hormones that are secreted into extracellular fluid and acts upon the cell that produced it.
Paracrine hormones that exerts its effects on cells near its secretion site, it acts within the organ
where it is produced.
Hormones are generally concerned with the control of metabolic functions such as the rates of chemical
reactions in cells, the transport of substances through plasma membranes, and other aspect of cellular
metabolism like growth or secretion
Hormones need physiological regulation:
Target cells must have receptor proteins
Combination of regulatory molecules and receptors must cause changes in the target cell
Must be a mechanism to turn sequence off – rapid removal or chemical inactivation
What are Hormones?
Interactions between hormones produce three different effects:
Synergistic effect is when hormones work together to produce an additive or complementary result
o Additive effect is when both hormones separately produce an increase
o Complementary effect is when both hormones do the same thing
Permissive effect is when it enhances the responsiveness of a target organ to the second hormone or
increases the activity of the second hormone.
Antagonistic effect is to antagonize the effects of other hormones
Hormones can produce their effects within seconds or even days, but then continue for weeks, months, or
even years.
Hormone effects may differ when taken at abnormally high, or pharmacological, concentrations
Hormones can either affect almost all cells, can be tissue specific, or affect only target cells.
A single gland may secrete multiple hormones, and a hormone can be secreted from more than one gland.
Although there are exceptions, each hormone is secreted by a specific cell type within the gland.
Some specialized neurons also act as endocrine glands by secreting chemical messages into the blood
rather than a narrow synaptic cleft, these are called neurohormones.
Hormones removed by the liver are converted by enzymatic reactions into less active products.
Second messengers are a component of signal transduction mechanism because extracellular hormones
are transduced into intracellular signals
These involve the activation: Adenylate Cyclase, Phospholipase C, Tyrosine Kinase, and cAMP
A substance that acts like a hormone but has not been purified and identified as a distinct chemical
compound is called a Factor.
Once it has been chemically characterized, it is called a hormone.
Different Types of Endocrine Hormones
Hormone molecules that affect the metabolism of target cells are often derived from precursor molecules.
Chemical categories of hormones include steroids, amines, polypeptides, and glycoproteins.
Hormones can be polar, which are water soluble, and nonpolar, which are water insoluble, and this regards their
action in target cells.
Non- polar hormones are also called Lipophilic hormones and can gain entrance into target cells
Polar hormones can’t pass through plasma membranes, these include include polypeptides, glycoproteins,
and catecholamine hormones.
Amine Hormones
Amine hormones are derived from amino acids such as Tyrosine and Tryptophan
Thyroxine and Triiodothyronine, which are both produced in the Thyroid Gland
Epinephrine and Norepinephrine, which are both produced in the Adrenal Medulla
Melatonin, which is produce in the Pineal Gland
Glycoproteins Hormone
Glycoproteins are proteins bound to one or more carbohydrate groups
Follicle Stimulating hormone (FSH), which is secreted by the Anterior Pituitary
Luteinizing hormone (LH), which is secreted by the Anterior Pituitary
Thyroid Stimulating Hormone (TSH), which is secreted by the Anterior Pituitary
Human Chorionic Gonadotropin (hCG), which is secreted by the Placenta.
These hormones cant be taken orally because they will be digested into inactive fragments
Intermediate Metabolism and Clearance of Hormones
Most protein hormones are secreted to peripheral circulation as a mix of multiple isoforms
These heterodimeric hormones consist of amino acid chains and N-linked glycosylation sites where
multiple oligosaccharide residues can be added
Glycosylation adds a high proportion of sialic acid-enriched carbohydrates and a lesser number of
sulphated residues to protein molecules.
Deglycosylation and proteolysis are major mechanisms of the inactivation and clearance of glycoprotein
hormones
The terminal sialic acid residues are typically removed by plasma glycosidases.
Hepatic cells then recognize the exposed hormone and peptide bonds between amino acids become
susceptible to enzymatic attack.
The less glycosylated/acidic isoforms of protein hormones exhibit a shorter half-live in systemic circulation
than their more acidic counterparts.
The differences in the degree of sialylation and sulphation of protein hormones are very important
determinants of their biological potency
The more acidic isoforms appear to be more potent stimulators of target organs
Peptide/Protein Hormones
These hormones are either small peptides (~ 3 A.A) or proteins (polypeptides)
They are made as larger hormones, called Preprohormones, on the ribosomes then cleaved to
prohormones by enzymes in the granular endoplasmic reticulum.
Prohormone are then packaged into secretory vesicles by the Golgi apparatus in which they are
cleaved to yield a specific hormone and other peptide chains – released by exocytosis
An example of two hormones coming from one prohormone are ACTH and MSH; because of this,
elevated levels may cause marked darkening of the skin.
An increase in the intracellular Ca concentration is the stimulus for exocytosis of some peptide hormones,
if not all, as it is for neurotransmitter release.
Source must be either extracellular or intracellular from the endoplasmic reticulum
The stimuli for the Ca influx may be a nerve, hormonal, or chemical/physical signal that directly or
indirectly cause some degree of depolarization of the cell membrane
Many of the peptide hormones also function as neurotransmitters such as the many hormones secreted by
endocrine cells of the gut which are also produced by neurons in the brain as neurotransmitters
Polypeptide Hormones include:
Calcitonin, from the Thyroid Gland
Parathyroid Hormone, from the Parathyroid Gland
Antidiuretic Hormone (ADH), from the Hypothalamus then the Posterior Pituitary
Adrenocorticotropic Hormone (ACTH), and Growth Hormone (GH), from the Anterior Pituitary
Insulin, Glucagon, and Somatostatin, from the Pancreas
Oxytocin and Prolactin, from the Posterior Pituitary
Steroid Hormones
Steroid hormones are derived from cholesterol by a series of enzymatic reactions following cleavage of the
cholesterol side chain that’s attached to the 5-C “D” ring to form Pregnenolone.
Steroid hormones are highly lipid soluble and can diffuse across plasma membranes and enter the blood.
Large quantities of steroid hormones are not stored, but cholesterol and precursors are.
They exert their effects by entering the target cell and bind to the Nuclear Receptor Proteins where they
influence the target tissue by stimulating genetic transcription – this is called Genomic Action (~30min)
Sometimes action occurs too quickly, this is called Nongenomic action, and occurs in the cytoplasm where
it involves the activation of a second messenger system
Receptors for steroid hormones are in the cytoplasm or nucleus – depends on the target.
Cytoplasmic receptors bind to their hormone then translocate to the nucleus where the receptor complex
binds to a specific DNA sequence called Hormone Response Element
Hormone Response Elements contain two half sites, each with 6 nucleotide bases separated by 3
nucleotide spacer segments, where one steroid receptor that’s bound to a molecule of the hormone
attaches as a single unit to one half site, and the other one binds to the other half site.
This process is called Dimerization and this example is a Homodimer since it uses the same receptor
When a steroid hormone ligand binds to its nuclear receptor protein it changes the receptor protein
structure, causing a removal of heat shock proteins that prevent the receptor from binding to DNA, and the
recruitment of coactivators that prevent corepressors from binding to the receptor.
There are coactivators and corepressors that work on specific transcription factors because they don’t bind
to the DNA and promote inhibit abilities.
Hormones include:
Testosterone, from the Testes/Gonads
Estradiol/Estrogen and Progesterone, from the Ovaries/Gonads
Cortisol and Aldosterone, from the Adrenal Cortex
Hormone Secretion
Hormone secretion usually occurs in short bursts, with little or no release in between bursts.
Some hormone secretion follows a circadian rhythm, and other can be released at specific times
There are four main types of input to endocrine cells that control hormone secretion:
Changes in the plasma concentrations of mineral ions
Changes in the plasma concentrations of organic nutrients
Neurotransmitter release from neurons impinging on the endocrine cell
Another hormone acting the endocrine cell.
Control of Hormone Secretion by Negative Feedback
Endocrine gland tends to over secrete its hormone, so the hormone exerts more of its effect on the target
The target organ in turn is stimulated to perform its function more and more intensively
If too much function occurs, some factor associated with the function feeds back on the "control"
gland to reduce the hormone secretion – Negative Feedback Loop
If the target organ responds poorly to the stimulation, the hormone will continue to be released and the
levels in the blood will rise above what is considered normal, often leading to disease.
Hormone Transport and Delivery
Water-soluble hormones are carried in the plasma in a dissolved state to their target organ(s).
Steroid and thyroid hormones are water-insoluble and circulate in the blood bound to plasma proteins.
Some plasma proteins are specifically for the transport of the hormone
These hormones are active when taken orally.
Even though most lipid soluble hormones are bound, a small amount exists as free dissolved in the plasma.
It is only this free hormone that can cross capillary walls and affect its target organ
Its assumed that there are specific protein transporters that facilitate the passage of through the
capillary endothelium
Hormone Receptors
Hormones almost always combine with a receptor either on the plasma membrane or inside the cell.
This hormone/receptor complex sets up a cascade of reactions, with each stage becoming more
powerfully activated than the previous stage – called the Second Messenger System or Casacade
Hormone binding is highly specific and tend to bind to receptors with a high affinity (intermolecular force),
and a low capacity; saturating the receptors with hormone molecules.
The receptors for the peptide and catecholamine hormones are either in, or on, the surface of the plasma
membrane due to their hydrophilic/Lipophobic nature
The receptors for the steroid hormones are found in the cytoplasm or nucleus due to their lipophilic nature
Lipophilic hormone receptors are called Nuclear Hormone Receptors because they function within the cell
nucleus to activate genetic transcription – also function as transcription factors that must be activated.
Has two domains: A ligand (hormone)-binding domain, and a DNA binding domain
Receptor must be activated by binding to its ligand before it can bind to a specific region of the DNA
called the Hormone Response Element
Receptors from unknown hormone ligands are called Orphan receptors
Since receptors are proteins, they continually get broken down and new ones get manufactured.
Often the binding of the hormone to the receptors causes the number of receptors to decrease because of
the inactivation of some receptor molecules (hormone-receptor internalization) or because of decreased
production of the molecules – it desensitizes
Prolonged exposure to polypeptide hormones can desensitize the target causing less target response
due to the downregulation of receptors
To prevent this, many polypeptide and glycoprotein hormone are secreted in Pulsatile Secretion
In a few cases, the binding of the hormone induces the formation of more receptor molecules by the
protein manufacturing machinery of the target cell – increases sensitivity and response
This is called the priming effect, and it is caused by the upregulation of receptors
General Mechanism of Hormone Action
After hormone combines with the receptor, there are several things that can happen:
1. Change in membrane permeability – an opening or closure of membrane ion channels that cause a flux
of ions and changes in membrane potentials that may excite or inhibit the cell.
2. Activation and inactivation of intracellular enzymes – for example, Insulin binds with its membrane
receptor that’s on the outside producing a structural change that causes a portion to extend into the
cell and become an activated kinase that promotes phosphorylation inside the cell
o Adenyl Cyclase is an activated enzyme that occurs at the protrusion and causes the formation of
cAMP - a second messenger.
o Functions of activating or inactivating intracellular enzymes include initiating synthesis of
intracellular chemicals, and initiating secretion from cells.
3. Activation of genes – for example, when steroid and thyroid hormones bind with protein receptors
inside the cell they activate a specific locus on the DNA strands which then initiates the transcription of
specific genes to form mRNAs – other tropic hormones can enhance the translation of mRNA
Hormone-receptor complexes cause the dissociation of a subunit from a complex of G proteins that then
moves until it reaches the enzyme adenylate cyclase where it binds to and activates the enzyme that
catalyzes the following reaction - ATP cAMP + PPi
cAMP activates an inactive enzyme in the cytoplasm called Protein Kinase, which has a regulatory
subunit that it must attach to, so it can dissociate from the catalytic subunit and become active
The active kinase catalyzes the phosphorylation of different proteins in the target which causes some
enzymes to become active and others to become inactive
cAMP must be rapidly inactivated by Phosphodiesterase, which is an enzyme that hydrolyzes cAMP, so it
can function appropriately as a second messenger
cGMP, cyclic guanosine monophosphate, and Ca2+ can function as a or a part of a second messenger.
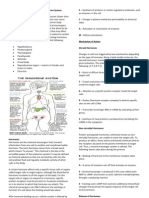
Pituitary Glands
The Pituitary, also called the Hypophysis, is a small gland at the base of the brain that’s attached to the
Hypothalamus by the Infundibulum, or the Hypophyseal stalk, in the region of diencephalon and it is divided into:
Anterior Pituitary, also called the Adenohypophysis – a glandular epithelium derived from Rathke’s Pouch
Pars Distalis – the rounded portion and the major endocrine part of the gland
Pars Tuberalis – a sheath of tissue that partially wraps around the infundibulum
Posterior Pituitary, also called the Neurohypophysis - nervous tissue and its called the Pars Nervosa
In contact with adenohypophysis and infundibulum – have neuroglia-like cells called Pituicytes
A zone between the two, called the Pars Intermedia, exists in animals, but not in adult human beings.
The Hypothalamus receives signals such as pain and olfactory from all sources in the nervous system.
Exciting and depressing thoughts also influence the Hypothalamus.
Receptors of the Hypothalamus monitor nutrients and ion concentrations in the blood.
Release of hormones from the Posterior Pituitary is controlled by signals that originate in the Hypothalamus.
The release of hormones from the Anterior Pituitary is controlled by hormones called Hypothalamic Releasing
and Inhibitors factors that are secreted by the Hypothalamus into the Hypothalamic-Hypophyseal portal vessels.
Hypothalamic Releasing/Inhibiting Factors
These factors are secreted from the neuron, picked up by the Hypothalamic-Hypophyseal portal system
and carried directly to the sinuses of the Anterior Pituitary gland.
These releasing/inhibiting factors of a non-reproductive nature include:
1. Thyroid-Stimulating Releasing hormone (TRH) - causes release of Thyroid-Stimulating hormone (TSH).
2. Corticotropin-Releasing hormone (CRH) - causes release of Adrenocorticotropin (ACTH).
3. Growth Hormone Releasing hormone (GHRH, Somatotropin) - causes release of Growth hormone (GH),
and Growth hormone inhibitory hormone (GHIH) – same hormone as Somatostatin
Posterior Pituitary
The Posterior pituitary is more of a storage organ.
Both posterior hormones, oxytocin and antidiuretic (ADH) hormones, are produced in neuron cell bodies of
the supraoptic and paraventricular nuclei in the hypothalamus.
They are then transported along axons of the hypothalamo-hypophyseal tract to the posterior
pituitary where they are stored and later released in response to neuroendocrine reflexes
ADH secretion is controlled by osmo-receptor neurons in the hypothalamus in response to increased
plasma osmolarity
Increased osmolarity stimulates an increased frequency of action potentials in the neurons that
produce ADH; this causes a greater opening of voltage gated Ca2+ channels at the axon terminals
which then produces a greater release of ADH by exocytosis
ADH secretion can also be inhibited by stretch receptors in the left atrium of the heart, which are
stimulated when there is a rise in blood volume.
Osmolarity is the concentration of a solution expressed as the total number of solute particles per liter.
Increase osmolarity = decrease in blood volume / more solutes
Decrease osmolarity = increase in blood volume / less solutes
Hormones Target Function
Antidiuretic Hormone (ADH) Kidneys Stimulates water retention
Oxytocin Uterus and Mammary Gland Stimulates uterine contraction and milk ejection
Antidiuretic Hormone (ADH)
The hormone Vasopressin regulates water excretion by the kidneys by increasing the permeability of
the collecting duct to water.
ADH is produced in Magnocellular neurons in the Supraoptic (80%) and Paraventricular (20%) nuclei of
the anterior hypothalamus.
Both of these nuclei have axonal extensions to the posterior pituitary where ADH is transported
with carrier proteins down these nerve endings where it is then released into the blood stream.
It is released from secretory granules in the nerve endings by Exocytosis in response to action
potentials in the neurons.
If osmolarity of the extracellular fluid increases above 300 mOsM, fluid is drawn from osmoreceptors in
the hypothalamus by osmosis decreasing the size of the osmoreceptor cell causing them to fire
This sends nerve signals to additional nerve cells in the Supraoptic nuclei, which then relay these
down the stalk to the posterior pituitary and causing ADH to be released.
A decrease in osmolarity of the extracellular fluid fails to generate the signal for ADH secretion.
There’s debate on the location of the osmoreceptors: some believe magnocellular neurons are the
receptors, evidence says there’s a synaptic afferent input into the supraoptic neurons, and its suggested
that they might be along the anteroventral region of the third ventricle near the median preotic nucleus
Minute amounts of ADH are secreted due to changes in osmolarity of only 1 mOsM.
Higher concentrations of ADH are secreted if there is at least a 10% change in blood volume, which
acts as a vasoconstrictor to restore blood pressure.
Secretion of Antidiuretic Hormone
When blood volume increases, receptors in the heart send signals to the hypothalamus to inhibit ADH.
Conversely, when blood volume is low, the receptors are not stimulated and ADH is secreted.
In addition, the right atrium of the heart contains receptors that are stretched by overfilling and send a
signal to the brain to inhibit ADH release.
Conversely, if the right atrium is under filled, there are fewer signals and more ADH is released to
produce vasoconstriction and to increase water reabsorption in the kidney.
In addition to the stretch receptors of the right atrium, the baroreceptors of the carotid, aortic, and
pulmonary vessels also participate in increasing ADH secretion.
The effect of ADH on the collecting duct is a graded response, so when ADH is low, little water is
reabsorbed in the collecting duct – urine with a low osmolarity (diluted) is voided.
Conversely, if hormone levels are high, a large amount of water is reabsorbed in the collecting duct -
urine with a high osmolarity (concentrated) is voided.
ADH is carried to the collecting duct in the blood of the Vasa Recta and combines with a receptor on the
basolateral membrane of the collecting duct cell.
This combination activates the processes that culminate in the insertion of water pores, also known
as Aquaporins, into the apical membrane so that water can move along its osmotic gradient.
The pores are stored in vesicles in the cytoplasm when ADH is not present.
In addition to osmotic stimuli, pain, nausea, surgical stress, emotions and drugs such as morphine,
nicotine and large doses of barbiturate increase ADH release; while alcohol depresses ADH release.
Diabetes Insipidus
According to the dictionary, Diabetes means increased urine production, also known as Polyuria.
Diabetes insipidus is a deficiency of ADH that causes polyuria and polydipsia, which is increased water drinking
In Diabetes Insipidus, the blood glucose levels are normal and there is no glucose in the urine.
Anterior Pituitary
Hormone secreted from the anterior pituitary are called trophic hormones because high concentrations of
cause targets to hypotrophy (degenerate); while low levels cause them to atrophy (waste away)
Some cells here are derived from the fetal pars intermedia and produce a large polypeptide called Pro-
opiomelanocortin (POMC) which is a prohormone whose products are beta-endorphine, MSH, and ACTH.
MSH, also known as Melanocyte-Stimulating Hormone, is a peptide hormone that is produced by the
pituitary gland, hypothalamus, and skin cells.
Axons don’t enter here, so hypothalamic control is achieved through hormonal regulation rather.
ACTH, TSH, and gonadotropins are controlled by negative feedback loops from the target gland hormones.
ACTH is inhibited by a rise in corticosteroid secretion, and TSH is inhibited by a rise in Thyroxine (T4).
The relationship between the anterior pituitary and the target is called the pituitary-gonad axis
This axis is stimulated by GnRH from the hypothalamus
This area can be stimulated by emotions due to the hypothalamus being able to receive neural input
from “higher brain centers”, and it can produce circadian rhythms
Psychological stress is known to activate the pituitary adrenal axis
Stressors produce an increase in CRH secretion from the hypothalamus, which in turn results in
elevated ACTH and corticosteroid secretion
Hormone Target Function
Growth Hormone (GH) Most Tissues Promotes protein synthesis and growth
Adrenocorticotropic (ACTH) Adrenal Cortex Increase secretion of glucocorticoids
Thyroid Stimulating Hormone Thyroid Increase secretion of thyroid hormones
Prolactin Mammary Gland Promotes milk production
Follicle Stimulating Hormone Gonads Promote gamete and estrogen production
Luteinizing Hormone (LH) Gonads Promote sex hormone secretion, ovulation and corpus lutem
formation in females, and testosterone production in males
Growth Hormone
Growth hormone (GH) causes growth of almost all tissues of the body that are capable of growing by increasing
cell size, mitosis, and specific differentiation of cells such as bone growth cells and early muscle cells.
Deficiencies in GH secretion may be congenital, or they may occur slowly, or suddenly.
The population of cells that produce GH constitute about 40-50% of endocrine cells of the anterior pituitary.
Growth hormone is under the control of two polypeptide hormones from the hypothalamus
Growth hormone-releasing hormone (GHRH) – stimulates secretion of growth hormone.
Growth Hormone inhibiting hormone (GHIH) – stimulates a withhold of GH release
Growth hormone has both anabolic and catabolic effects.
There is an age-related decline in GH secretion after the age of 20, accompanied by a decline in IGF-1.
May contribute to the increased adipose tissue and reduction in muscle mass that occur during aging.
Other effects of GH include:
Increases the rate of protein synthesis – similar to Insulin
Increases the mobilization of fatty acids from fat
Increases the use of the fatty acids for energy
Decreases the rate of glucose use in the body – due to decreased glycolysis; similar to Glucagon
Growth Hormone Effects – Stimulation of Protein Synthesis
Increased protein synthesis mechanism isnt known, but it does enhance transport of amino acids in the cell
In the absence of increased amino acids, GH still increases RNA translation.
GH also stimulates protein synthesis over 24-48hrs, by stimulating the transcription of DNA causing
increased quantities of RNA.
There is also a decrease in the breakdown of cellular protein maybe due to moving fatty acids from fat cells
which replaces protein as the source of energy
Growth Hormone Effects – Mobilization and Use of Fatty Acids
GH not only increases the mobilization of fatty acids from adipose tissue, but it also enhances the
conversion of fatty acids to acetyl-CoA.
Fat is utilized for energy preferentially over carbohydrates and proteins.
If there is an excessive quantity of GH and large amounts of fat are being mobilized
This excessive mobilization of fat from the adipose tissue causes a fatty liver
Can cause excess amounts of Acetoacetic Acid being formed in the liver which can cause a condition
called Ketosis to develop
Growth Hormone Effects – Decreased Utilization of Glucose
Decreased utilization of glucose is due to the increased mobilization and use of fatty acids for energy.
The fatty acids in turn form acetyl-CoA that initiates feedback effects to block the glycolytic
breakdown of glucose and glycogen.
Because the glucose and glycogen cannot easily be utilized for energy, the glucose that does enter
the cells is converted into glycogen and deposited – resulting in rapid saturation of glycogen
Within 30-60 minutes, blood glucose levels start to rise because the cells can no longer take up
glucose.
This type of hyperglycemia is not responsive to insulin therapy
The increase in blood glucose levels caused by the GH triggers the release of insulin from the pancreas.
In addition, GH directly stimulates the pancreas to secrete extra insulin.
The combination of these two forms of stimulations will eventually cause the Beta cells of the
pancreas to burn out – causes desensitization.
Growth Hormone Effects – Stimulation of Cartilage and Bone Growth
GH causes deposition of new cartilage by stimulating the maturation and mitosis of the chondrocytes in the
Epiphyseal Plates where the long bones grow.
The cartilage becomes calcified to bone and the shaft lengthens furthering the epiphyses more apart
The cartilage is progressively getting used up so there comes a time at which growth stops and bone
fusion of the shaft and the epiphysis occurs.
GH stimulates steoblasts in the Periosteum, the membrane on the surface of the bone and in some bone
cavities, deposit new bone on the surface of existing bone.
Simultaneously, osteoclasts in the bone remove old bone.
When the rate of deposition exceeds the removal process, the bones become thicker.
GH causes the liver and many other tissues to form and secrete Somatomedin-C or Insulin-like growth
factor 1(IGF-1).
IGF-1 is a paracrine hormone produced by many cells.
Its likely that GH stimulates the chondrocyte precursor cell and young chondrocytes in the epiphyseal
plates to differentiate into chondrocytes.
During differentiation, the chondrocytes begin to secrete IGF-1 and to become responsive to IGF-1
The IGF-1 then acts locally to stimulate the differentiating chondrocytes to undergo mitosis
Mediated by the somatomedins, IGF-1 and IGF-2, can secrete more cartilage matrix
Malnutrition during childhood inhibits the production of IGF-1 even though plasma levels of growth
hormone are normal.
Regulation of Growth Hormone Secretion
Rate of secretion of GH decreases after adolescence, but it does not cease all together.
In a child, the plasma concentration of GH is about 6 ng/ml and in the adult, its between 1.5-3 ng/ml.
Levels of GH fluctuate with the time of day, with the largest peak at 10pm-2am if the child is asleep
The rate of secretion can change very rapidly; factors such as stress, excitement, trauma and exercise can
cause these rapid changes.
Long term nutritional deficiencies such as hypoglycaemia, low concentration of fatty acids in the blood or
protein deficiency, will raise GH levels.
Secretion is regulated by Growth Hormone Releasing Hormone (GHRH) and Growth Hormone Inhibiting
Hormone (GHIH, SRIF), which are secreted by hypothalamus and transported to the Anterior Pituitary
through the Hypothalamic-Hypophyseal portal vessels.
The same area of the hypothalamus that secretes these is also sensitive to blood glucose levels.
Its likely that some of the same signals that initiate feeding also stimulate the secretion of GH.
Growth Hormone Disease – Gigantism
If the cells of the Anterior Pituitary become over active due to hyperplasia, which is the enlargement of an
organ or tissue, or due to a tumour, excess quantities of GH will be produced.
If this over-production occurs before the epiphyses of the long bones have fused with the shafts, the bones
continue to grow, and the individual will become taller than normal with extremely long bongs
They are usually hyperglycaemic and about 10% eventually develop Diabetes Mellitus.
Diabetes develops because the Beta cells of the islets of Langerhans in the pancreas become
degenerated, partially because they become over active due to the hyperglycemia and partially
because of a direct over-stimulating effect of GH on the islet cells
Growth Hormone Disease – Acromegaly
If a tumour occurs after the epiphyses has been fused, the long bones cannot continue to grow causing the
soft tissues continuing to grow and the thickness of bones to grow.
This results in enlargement of the hands and feet, in membranous bones in the cranium, the nose, as well
as the bones of the forehead, the Supraorbital ridges, the lower jawbone and portions of the vertebrae.
The result is a protruding jaw, sloping forehead, very large nose, feet and hands.
The growth of the vertebrae causes a hunched back, and organs such as tongue, liver and kidneys
become enlarged.
Growth Hormone Disease – Dwarfism
Most cases result from a generalized deficiency of Anterior Pituitary hormones (GH) during childhood.
The body develop in proportion to each other, but none of the parts achieve their "normal" size.
If there is a general deficiency of Anterior Pituitary hormones, the child doesn’t secrete sufficient amounts
of Gonadotropic hormones to attain puberty.
In about 1/3 of dwarfs, there is only a deficiency of GH and these individuals mature sexually.
Dwarfism is defined as adult height of less than 4f10in.
The African Pygmy has normal levels of GH, but there is a hereditary inability to form Somatomedin-
C/Insulin-like growth factors in response to G.
Levi-lorain dwarf also struggles with the release of these factors.
Laron dwarfism is when target cells are insensitive to GH because of a mutation in the gene the codes for
the GH receptor.
Most cases are caused by Achondroplasia which is a condition inherited as an autosomal dominant trait
that results from a mutated gene coding for Fibroblast Growth Factor receptor 3 (FGFR3)
When defective it causes impaired function and cell division of chondrocytes within the growing
portions of bones that grow by endochondral ossification
Thyroid Glands
The Thyroid gland is located below the Larynx on either side of the Trachea and connected anteriorly by a medial
mass of thyroid tissue called the Isthmus.
It secretes the amines Thyroxine (T4) and Triiodothyronine (T3), which are very important, and the peptide
Calcitonin
It’s the largest endocrine gland
Thyroid hormones stimulate the formation of many different types of mRNA, which within hours results in the
formation of hundreds on new types of proteins.
Because these hormones affect growth and metabolism, virtually every tissue in the body is affected.
Calcitonin inhibits the activity of osteoclasts, which promotes dissolution of calcium phosphate crystals of bone,
stimulates the urinary excretion of Ca+2 by the kidneys, lowers blood Ca+2, and antagonizes the effects of the
parathyroid hormone.
Its secreted by the parafollicular cells, also known as the C cells, and lack an iodide.
It has very little effect for adults, and is more important for young animals and humans.
The thyroid hormones are required for brain development in the fetus and the first few years after birth.
Although T4 is secreted in larger amounts (80%) than T3, many tissues, including the hormone's target cells,
convert most of the T4 into T3.
Because T3 and T4 are lipid soluble, they can cross the cytoplasmic membrane and bind to receptors attached to
the DNA genetic strands near them.
Structure and Physiology of the Thyroid Gland
The thyroid gland consists of many follicles which are spherical hollow sacs that consist of layers of simple
cuboidal follicular epithelial cells, called Thyrocytes, surrounding a colloid-filled space – synthesize T4
Interior of the follicles contain a colloid which is a protein rich fluid that is primarily a glycoprotein
called Thyroglobulin synthesized by the follicular cells and secreted into the lumen of the follicle.
Thyroglobulin contains about 70 tyrosine amino acids and that iodine binds to.
Thyroid hormones are formed, and stored within the thyroglobulin molecule in the follicular fluid.
Also contains parafollicular cells that secrete Calcitonin, or Thyrocalcitonin.
Most of the iodine ingested in food is transported from the intestine into the blood where it is converted
into an ionized form, iodide (I-).
Iodide then circulates in the blood to the thyroid gland where it’s actively transported into the
follicular epithelial cells in the colloid – called Iodide Trapping
The concentration of Iodide here may be 30x higher than in blood.
In the cells, iodide is oxidized by peroxidase to form iodine
Peroxidase is located at the apical border of the follicular cell where the thyroglobulin enters.
As iodine diffuses into the lumen, it binds to the tyrosine of the thyroglobulin with the help of an
iodinase enzyme - this is called Organification of the thyroglobulin.
Because this binding is assisted by the iodinase enzyme, the process is rapid.
But iodine does not combine with all the tyrosine’s, it combines one molecule at a time to form
Monoiodotyrosine (MIT), then Diiodotyrosine (DIT).
Over time, the DIT residues join to form the thyroid hormones.
If a MIT and a DIT couple, T3 is formed and if two DIT residues couple, T4 is formed.
The thyroid hormones are stored in large quantities in the follicular fluid, enough for several months.
At the time of release the hormones are enzymatically split from the thyroglobulin by proteases, then
diffuses through the base of the thyroid cell into surrounding capillaries.
Thyroid Hormone Effects – Bodily Mechanisms
Thyroid hormones are the most important determinant of the BMR for all sizes, ages and sexes.
The effects are due to the synthesis of proteins, some of which will act as enzymes.
They stimulate carbohydrate metabolism including the rate of glucose uptake by the cells,
Gluconeogenesis, gut absorption and insulin secretion.
Fat stores are rapidly depleted by these hormones because lipids are mobilized from the fat tissue,
therefore, increasing the free fatty acid concentration in the plasma.
Thyroid hormones accelerate the oxidation free fatty acids by the cells.
Though, cholesterol, phospholipids and triglycerides in the plasma decrease.
One of the mechanisms by which thyroid hormones decrease the plasma cholesterol concentration is to
increase the rate of cholesterol secretion in the bile.
It may increase the numbers of LDL receptors on the liver cells, leading to rapid removal of LDLs from
the plasma and subsequent secretion of the lipoprotein and cholesterol by the liver cells
Because vitamins are essential parts of some enzymes or coenzymes, thyroid hormones cause increased
requirements of vitamins.
Weight loss often occurs due to the increased metabolic rate, but it can be offset by increased appetite.
Increased metabolism increases the tissues requirements for oxygen, which causes a vasodilation
and increased blood flow to the metabolically active tissues.
Blood flow to the skin will increase in humans and animals that use the skin to thermoregulate.
To compensate for the increased blood flow, heart rate and force of contraction increases.
If there is excess thyroid hormone, the heart muscle becomes depressed because of the excessive
protein catabolism.
The heat producing, or calorigenic, effects of thyroxine are required for cold adaptions.
Though this is permissive, because T4 contributes to the adaptive thermogenesis as well as the BMR.
BMR has two components, one that’s independent of T4 action and one that is regulated by T4
Thyroid Hormone Effects - Cellular Metabolic Activity
Primary function is to increase the metabolic activities of almost all the tissues in the body.
Using food energy is increased and increased protein synthesis is matched by increased protein catabolism
Mental processes are excited, and the activity of most endocrine glands are stimulated.
Increased metabolism occurs because of an increase in the number and size of the mitochondria.
Total membrane surface area of the mitochondria increases in proportion to increased metabolic rate
The thyroid hormones also increase the activity of Na+/ K+ ATPase, which increases Na+ and K+ transport
This activity utilizes energy and increases the amount of protein produced in the body.
Regulation of Thyroid Hormone Secretion
Thyroid hormone receptor proteins are in the nucleus bound to DNA even in absence of their ligand.
Thyroid hormone response element of DNA has two half sites, but its receptor only binds to one.
The other is for Vitamin D derivative, 9-cis-retinoic acid, which works together to form a heterodimer.
In the absence of T3, the receptors recruit corepressors that inhibit genetic transcription
If T3 is there, corepressors are removed and degraded by proteasomes, & coactivators are recruited
T3 uses nonspecific binding proteins in the cytoplasm as stepping stones to enter the nucleus, where
T3 binds to the ligand-binding domain of the receptor and causes the receptor to change shape
Feedback mechanisms operate through the hypothalamus and anterior pituitary to control secretion
TSH, or Thyrotropin, increases the secretion of T4/T3 by increasing proteolysis of the thyroglobulin.
TSH also increases the activity of the iodide pump which increases the "Iodide Trapping" in the
glandular cells - increases the size, secretory activity, and number of thyroid cells.
Anterior pituitary is controlled by Thyrotropin-Releasing hormone (TRH) which increases TSH secretion
Cold exposure is a potent stimulators of TRH releases and therefore TSH release.
Increased plasma levels of thyroid hormones decrease the secretion of TSH by inhibition of the
anterior pituitary – proposed that they reduce the number of TRH receptors on cells that secret TSH.
Thyroid Hormone Diseases – Hyperthyroidism
In Hyperthyroidism, the thyroid gland is enlarged and appears as a large lump over the larynx.
The patient exhibits symptoms of:
Heat intolerance, and increased sweating
Mild to extreme weight loss, and varying degrees of diarrhea
Muscular weakness, and extreme fatigue – but inability to sleep
Nervousness, and tremoring of the hands
Some develop a degree of protrusion of the eyeballs - Exophthalmia
The enlargement of the thyroid gland is due Hyperplasia of the follicular epithelial cells of the follicles.
As the number of cells increase, the rate of secretion of thyroglobulin increases so the formation
of thyroid hormones also increases.
The changes in the thyroid gland are like those seen in excesses of TSH, but TSH levels are usually zero
due to negative feedback.
Instead, Thyroid Stimulating Immunoglobulins that mimic the actions of TSH are believed to be
the cause - an autoimmune condition.
Occasionally, hyperthyroidism results from a thyroid tumour that results in proliferation of the thyroid
cells and the secretion of large quantities of thyroid hormones.
Due to the high level of secretion, negative feedback will inhibit the normal thyroid cells and only
the tumour cells secrete at an excessive rate.
Thyroid Hormone Diseases – Hypothyroidism
In hypothyroidism, the thyroid may be small due to deterioration and fibrosis, which results in reduced
secretion of hormones - an autoimmune condition where antibodies destroy the thyroid gland
Often starts out as an inflammation that subsequently progresses to destruction and fibrosis.
However, the thyroid may be enlarged (Goitre) due to a deficiency of iodine in the diet.
Lack of iodine prevents production of T4 and T3, but doesn’t stop the formation of thyroglobulin.
The production of TSH by the anterior pituitary is not inhibited through negative feedback and TSH
causes the thyroid cells to secrete tremendous amounts of thyroglobulin into the follicles, causing the
gland grows larger and larger.
Because thyroid hormones levels determine basal metabolic rate, low levels result in the patient being
tired all the time even though they are sleeping 14-18 hours per day.
If this occurs during the first 6 months after birth, it may result in Cretinism which may cause the person
to suffer from severe mental retardation.
Symptoms include:
Extreme muscular weakness
Slow heart rate, decreased cardiac output, and decreased blood volume
Decreased ability to adapt to cold stress
Sometimes, increased weight gain, constipation, mental sluggishness, depressed hair growth,
scariness of the skin, development of a froglike husky voice
In advanced cases, Edema, that is particularly evident in the throat area (Myxedema), in which
mucoproteins and fluid accumulate in the subcutaneous connective tissues and viscera.
Causes swelling of the hands, feet, face, and tissue around the eyes and also slows physical
and mental activity which may eventually result in myxedema coma.
Calcitonin
Calcitonin, which is the last hormone from the thyroid gland, has the antagonizes of parathyroid
hormone and reduces plasma concentrations of calcium.
Its stimulated by an increase in plasma calcium
There is a second hormonal feedback mechanism for controlling calcium, but calcitonin works
more rapidly than the parathyroid feedback system
In some young animals, calcitonin decreases blood calcium very rapidly.
It functions by decreasing the absorptive activities and the formation of the osteoclasts, which favours
the deposition of calcium in the bones.
The calcitonin mechanism acts primarily in the short-term, yet it is not activated by elevated calcium
levels after a milk meal.
If the thyroid gland is removed and calcitonin is not secreted, the long-term plasma calcium ion
concentration is not significantly affected
It appears that the parathyroid hormone is the most important hormone regulation calcium ion
concentrations in the plasma
General Overview of All Endocrine Glands
Gland Hormone Major Functions
Adrenal Medulla Adrenaline & Noradrenaline (amines) Stimulates metabolism; affect CV system; stress response
Thyroid Thyroxine (T4) & Triiodothyronine (T3) (Amines) Organic metabolism, brain development & functions,
Calcitonin (peptide) growth; plasma calcium levels
Parathyroid Parathyroid hormone (peptide) Plasma calcium & phosphate levels
Hypothalamus Inhibiting/Releasing hormones, & Vasopressin Secretion of hormones by the anterior pituitary
(ADH) (peptide)
Anterior Growth Hormone (somatotropin), Thyroid- Growth; secretion of insulin-like growth factors, organic
Pituitary Stimulating hormone (thyrotropin), metabolism, Thyroid gland stimulation, Adrenal cortex
Adrenocorticotropic hormone (corticotropin)
(proteins)
Posterior Vasopressin (peptide) produced in hypothalamus Water excretion by the kidneys; blood pressure
Pituitary but released here
Pancreas Insulin, Glucagon, Somatostatin, Pancreatic Organic metabolism; plasma glucose levels
polypeptides (polypeptides)
Kidney 1,25-dihydrovitamin D3 Erythrocyte production; calcium absorption by intestine
Pineal gland Melatonin (amine) Body rhythms
Thymus Thymosin (thymopoietin) (peptide) T-lymphocytes function
GI Tract Gastrin, Secretin, Cholecystokinin, Somatostatin GI tract activity, liver & pancreas; Glucose-dependent
(peptides) insulin-tropic
Heart Atrial Natiuretic factor (ANF) (peptide) Sodium excretion by kidneys; blood pressure regulation
Adrenal Cortex Cortisol Organic metabolism; response to stresses; immune
Aldosterone (steroids) system; Na, K & H ion secretion by the kidney
You might also like
- Hormone: For Other Uses, SeeDocument5 pagesHormone: For Other Uses, SeeEki Ryan SetiowatiNo ratings yet
- Endrocrinology - PPTX Version 1Document40 pagesEndrocrinology - PPTX Version 1Abishek BhadraNo ratings yet
- EndocrinologyDocument35 pagesEndocrinologyNasif HussainNo ratings yet
- Hormones, ClassificationDocument10 pagesHormones, ClassificationMenoNo ratings yet
- Introduction - CC3: Mechanism of Hormonal ActionDocument40 pagesIntroduction - CC3: Mechanism of Hormonal ActionAl-hadad AndromacheNo ratings yet
- Hormone: For Other Uses, SeeDocument4 pagesHormone: For Other Uses, SeePrasanna Nair ParameswaranNo ratings yet
- Introduction To Endocrinology: Dr. Jehad Al-ShuneigatDocument21 pagesIntroduction To Endocrinology: Dr. Jehad Al-Shuneigatyousef sarairehNo ratings yet
- Study of The Endocrine SystemDocument225 pagesStudy of The Endocrine SystemHuzaifa RashidNo ratings yet
- Chemistry ProjectDocument23 pagesChemistry Projecttwinklelee123450% (2)
- What Are HormonesDocument16 pagesWhat Are HormonesHarry RoyNo ratings yet
- Biochemistry of HormonDocument16 pagesBiochemistry of HormonMuhamad SdeqNo ratings yet
- The Endocrine System Part 1Document64 pagesThe Endocrine System Part 1rizwanbasNo ratings yet
- Adrenocorticotropic Hormone (ACTH) From The Anterior Pituitary Gland Specifically StimulatesDocument2 pagesAdrenocorticotropic Hormone (ACTH) From The Anterior Pituitary Gland Specifically StimulatesStjepan ErženNo ratings yet
- HORMONESDocument9 pagesHORMONESSARA M SNo ratings yet
- Endocrine SystemDocument41 pagesEndocrine SystemMerrin EncarnacionNo ratings yet
- 4 Endocrine 1Document18 pages4 Endocrine 1ShenNo ratings yet
- Hormones 859645Document34 pagesHormones 859645Musharaf RehmanNo ratings yet
- 6 Group Phsiology.....Document14 pages6 Group Phsiology.....Ayat ChaudaryNo ratings yet
- Anatomy and Physiology-Endocrine SystemDocument5 pagesAnatomy and Physiology-Endocrine SystemEixid Enna YeLikNo ratings yet
- Biokim Pak AgusDocument6 pagesBiokim Pak AgusJOSEPHINE 102020111No ratings yet
- Classification of Hormones: By. Dr. Luna PhukanDocument13 pagesClassification of Hormones: By. Dr. Luna Phukanacrehell8No ratings yet
- Biology ProjectDocument27 pagesBiology Projectravifact0% (2)
- Endocrine System OutlineDocument17 pagesEndocrine System OutlineEsthermae PanadenNo ratings yet
- EndocrinologyDocument74 pagesEndocrinologysyedahaleemajunaidsNo ratings yet
- Endocrinology 1st TrimesterDocument16 pagesEndocrinology 1st TrimesteriwennieNo ratings yet
- Introduction To HormonesDocument26 pagesIntroduction To HormonesHanif JundanaNo ratings yet
- The Endocrine System: A System of Chemical CommunicationDocument7 pagesThe Endocrine System: A System of Chemical CommunicationKaleem UllahNo ratings yet
- Endocrine SystemDocument3 pagesEndocrine SystemRosario ArroyoNo ratings yet
- E N D o C R I N o L o G Y: Dr. Victoria G. Giango Chair, Dept. of PhysiologyDocument23 pagesE N D o C R I N o L o G Y: Dr. Victoria G. Giango Chair, Dept. of Physiologyanon-271548No ratings yet
- HORMONESDocument65 pagesHORMONESsoumya palavalasaNo ratings yet
- Lec 8 - Hormones PDFDocument13 pagesLec 8 - Hormones PDFrajeshNo ratings yet
- The Endocrine System: B. Pimentel, M.D. University of Makati College of NursingDocument54 pagesThe Endocrine System: B. Pimentel, M.D. University of Makati College of Nursingapi-19824701100% (1)
- HormonesDocument10 pagesHormonesAmjad AlmousawiNo ratings yet
- The Endocrine SystemDocument41 pagesThe Endocrine SystemОксана КрасильниковаNo ratings yet
- Endocrine Glands & Their Functions: Anatomy & Physiology II Georgia Perimeter College K. Donaldson, InstructorDocument55 pagesEndocrine Glands & Their Functions: Anatomy & Physiology II Georgia Perimeter College K. Donaldson, InstructorIsaacJ22No ratings yet
- Hormonal Regulation of ExerciseDocument11 pagesHormonal Regulation of ExerciseMozil Fadzil KamarudinNo ratings yet
- 1 - Endocrine 1 (Introduction) - MedicineDocument36 pages1 - Endocrine 1 (Introduction) - MedicineBHUWAN BASKOTANo ratings yet
- ENDOCRINE SYSTEM - Anatomy and PhysiologyDocument10 pagesENDOCRINE SYSTEM - Anatomy and PhysiologyJay Crishnan Morales CajandingNo ratings yet
- Hormones:: Signaling MoleculesDocument20 pagesHormones:: Signaling MoleculesSangeeta DwivediNo ratings yet
- Coordination of Body Functions by Chemical MessengerDocument6 pagesCoordination of Body Functions by Chemical MessengerHorain FatimaNo ratings yet
- Endocrine System OverviewDocument11 pagesEndocrine System OverviewwasimsafdarNo ratings yet
- Endocrine System: EndocrinologyDocument10 pagesEndocrine System: EndocrinologyAnya IgnacioNo ratings yet
- Basic of Endocrine: Dr. Anis Irmawati, DRG., MkesDocument31 pagesBasic of Endocrine: Dr. Anis Irmawati, DRG., MkesBima Ewando KabanNo ratings yet
- Biochemistry Assignment Submitted To: Dr. Noureen Submitted byDocument15 pagesBiochemistry Assignment Submitted To: Dr. Noureen Submitted bySalman KhanNo ratings yet
- SGD Physiology Endocrine and MetabolismDocument7 pagesSGD Physiology Endocrine and MetabolismTinesh RajahNo ratings yet
- Reproductive SystemDocument114 pagesReproductive SystemIsha NagleNo ratings yet
- DocumentDocument45 pagesDocumentPrajwal PatilNo ratings yet
- UntitledDocument12 pagesUntitledDerrick kinyaNo ratings yet
- NOTES Endocrinology ToxicologyDocument52 pagesNOTES Endocrinology ToxicologyRenz Gerard AmorNo ratings yet
- Endocrine System PDFDocument38 pagesEndocrine System PDFUlfat NiazyNo ratings yet
- Endo Lect - OverviewDocument24 pagesEndo Lect - OverviewdoctorrfarrukhNo ratings yet
- Clinton Hackney Anatomy & Physiology II BctcsDocument65 pagesClinton Hackney Anatomy & Physiology II BctcsJack HolthouseNo ratings yet
- The Endocrine System: Hormone Classification and Mechanisms of Intercellular CommunicationDocument82 pagesThe Endocrine System: Hormone Classification and Mechanisms of Intercellular Communicationعم رNo ratings yet
- Endocrine Physiology - Part 1Document40 pagesEndocrine Physiology - Part 1Terrence Beniasi CharumbiraNo ratings yet
- Care of Clients With Problem On EndocrineDocument48 pagesCare of Clients With Problem On EndocrineAngel CauilanNo ratings yet
- Endocrine SystemDocument95 pagesEndocrine SystemUmar Ilyasu JibrilNo ratings yet
- Endocrine GlandDocument4 pagesEndocrine Glandakash_zizouNo ratings yet
- Biochemistry of Hormones (Part 1)Document31 pagesBiochemistry of Hormones (Part 1)arun231187No ratings yet
- EndocrinologyDocument223 pagesEndocrinologyMr mk RollinsNo ratings yet
- Endocrine System: A Tutorial Study GuideFrom EverandEndocrine System: A Tutorial Study GuideRating: 5 out of 5 stars5/5 (1)
- 39 Prep GuideDocument73 pages39 Prep Guidedaljit chahalNo ratings yet
- IAL Biology SB2 Assessment 7CDocument4 pagesIAL Biology SB2 Assessment 7CsalmaNo ratings yet
- Nutrition Noteworthy, 4 (1) Garcia, Marcela EsperanzaDocument7 pagesNutrition Noteworthy, 4 (1) Garcia, Marcela EsperanzadidikekoNo ratings yet
- Excretion Worksheet 1 AnswersDocument3 pagesExcretion Worksheet 1 Answersjjnaseem100% (3)
- Endocrine Anatomy 4Document16 pagesEndocrine Anatomy 4Snehal JayaramNo ratings yet
- DI - Patho, Signs, Causes, PharmacologyDocument1 pageDI - Patho, Signs, Causes, PharmacologyVishalNo ratings yet
- STAAR EOC 2017 Test Biology PDFDocument48 pagesSTAAR EOC 2017 Test Biology PDFAnonymous WZXek8GNo ratings yet
- Dysnatremia in The ICU: Milap Pokaharel and Clay A. BlockDocument13 pagesDysnatremia in The ICU: Milap Pokaharel and Clay A. BlockJonathan Gustavo MenaNo ratings yet
- MEdical Surgical REVEWERDocument86 pagesMEdical Surgical REVEWERennaedNo ratings yet
- The Endocrine System The Endocrine System: © 2018 Pearson Education, Ltd. 1Document12 pagesThe Endocrine System The Endocrine System: © 2018 Pearson Education, Ltd. 1lourd nabNo ratings yet
- Endocrine DisordersDocument77 pagesEndocrine Disordersahmad100% (2)
- Basic Concepts of Fluid and Electrolyte TherapyDocument136 pagesBasic Concepts of Fluid and Electrolyte Therapymuhammad ajiNo ratings yet
- 83 Siadh2 PDFDocument8 pages83 Siadh2 PDFAnonymous QNYOxtHNo ratings yet
- Organ Systems Work TogetherDocument15 pagesOrgan Systems Work TogetherShekaina Faith Cuizon LozadaNo ratings yet
- IVMS Physiology and Pathophysiology Flash FactsDocument4,648 pagesIVMS Physiology and Pathophysiology Flash FactsMarc Imhotep Cray, M.D.No ratings yet
- NCM 116 Endocrine DisordersDocument106 pagesNCM 116 Endocrine DisordersAnthony Seth AguilandoNo ratings yet
- DI Diagnosis and TreatmentDocument2 pagesDI Diagnosis and TreatmentLuiciaNo ratings yet
- UrosepsisDocument22 pagesUrosepsisWita Ferani KartikaNo ratings yet
- Rot 3 - Darunday SicDocument9 pagesRot 3 - Darunday SicEzra Miguel DarundayNo ratings yet
- LECTURE 4 - Major Intra - and Extracellular ElectrolytesDocument66 pagesLECTURE 4 - Major Intra - and Extracellular ElectrolytesGamotkoto PharmacyNo ratings yet
- The Porn Circuit - Covenant Eye - DesconocidoDocument47 pagesThe Porn Circuit - Covenant Eye - DesconocidoirvmacNo ratings yet
- Renal Siadh Di CSWDocument54 pagesRenal Siadh Di CSWRizka Nurul Firdaus100% (1)
- Water Balance and Regulation of OsmolalityDocument35 pagesWater Balance and Regulation of OsmolalityMaria InolynNo ratings yet
- Metheny - Fluid and Electrolyte Balance 4th EdDocument227 pagesMetheny - Fluid and Electrolyte Balance 4th Edfirst aid kit100% (1)
- Pediatric Nephrology, 7th EditionDocument65 pagesPediatric Nephrology, 7th EditionDavien Utoyo100% (1)
- Type Drug ChartDocument3 pagesType Drug ChartKarina Rodriguez100% (3)
- Endocrine WorksheetDocument7 pagesEndocrine WorksheetBrenda Zaragoza63% (8)
- NCMB316 LEC Questions for SIADH and Diabetes InsipidusDocument18 pagesNCMB316 LEC Questions for SIADH and Diabetes InsipidusRegiever Salvante100% (1)
- Select All That Apply SATADocument58 pagesSelect All That Apply SATANicholas TagleNo ratings yet
- Assessment and Management of Patients With Endocrine DisordersDocument78 pagesAssessment and Management of Patients With Endocrine Disordershenny1620100% (1)