Professional Documents
Culture Documents
Using Your Text Book and These Descriptions of Chemical Reactions, Complete This Worksheet Parts 1,2 and 3
Uploaded by
MuhammadOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Using Your Text Book and These Descriptions of Chemical Reactions, Complete This Worksheet Parts 1,2 and 3
Uploaded by
MuhammadCopyright:
Available Formats
CHEM 51.
CHEMICAL REACTIONS WORKSHEET (Chapter 5)
Using your text book and these descriptions of chemical reactions, complete this worksheet Parts 1,2 and 3.
DESCRIPTIONS:
1. COMBINATION REACTION: Two reactants combine to form a single product.
2. DECOMPOSITION REACTION: One reactant decomposes into two or more products.
3. COMBUSTION REACTION: A reactant (a carbon compound made of C, H and sometimes O) combines
with oxygen to give CO2 and H2O as the only products.
4. SINGLE DISPLACEMENT (REPLACEMENT) REACTION: Reactants are an element and a compound.
Products are an element and compound. The reactant element replaces a similar element in the compound and
the replaced element becomes the product element.
5. DOUBLE DISPLACEMENT (REPLACEMENT) REACTION. Two reactant compounds and two product
compounds. One of the product compounds has to be a covalent compound, a gas, or a solid (precipitate).
Balancing Combustion Reactions
1. Balance carbon first
2. Balance hydrogen second
3. Sum the number of oxygens from water and CO2 the right side.
3a) Subtract any oxygens in the carbon compound on the left (reactant) side of the equation from the total
number of oxygens on the right.
4. Put this number of oxygen atoms divided by 2 in front of O2 as the coefficient.
5. If the coefficient is divisible by 2 then divide and place that number in front of the oxygen as the coefficient.
5a) If the coefficient is not divisible by 2 then multiply the whole equation by 2 to clear the X/2 in the oxygen
coefficient.
6. Write the balanced equation.
Example 1. Write a balanced equation for the combustion of C 4H10
C4H10 + 13 O2 5H2O + 4CO2
2 Step 2 Step 1
Step 4
Step 3 Total oxygen = 5 (5H2O) + 8 (4CO2) = 13 oxygen
Step 5a Multiply the whole equation by 2:
2 [C4H10 + 13 O2 5H2O + 4 CO2] = 2C4H10 + 13 O2 10H2O + 8 CO2
2
Step 6
Example 2. Write a balanced equation for the combustion of C 4H10O
C4H10O + 6 O2 5H2O + 4CO2 balanced Step 6
Steps 4 & 5 Step 2 Step 1
Step 3 Total oxygen = 5 (5H2O) + 8 (4CO2) = 13 oxygen – 1 oxygen = 12 oxygen
Step 3a
PART 1. Match the following reactions with the type reaction (draw a line from one to the other):
CH4 (g) + O2 (g) CO2 (g) + H2O (l) combination reaction
CuSO4 (aq) + Al (s) Al2(SO4)3 (aq) + Cu (s) decomposition reaction
HgO (s) Hg (l) + O2 (g) combustion reaction
Fe (s) + O2 (g) Fe2O3 (s) single replacement reaction
NaOH (aq) + HNO3 (aq) NaNO3(aq) + H2O (l) double replacement reaction
PART 2. Rewrite each of the equations above so they are balanced.
PART 3. FOR EACH OF THE FOLLOWING WORD REACTIONS, WRITE A BALANCED EQUATION
AND GIVE THE NAME OF TYPE REACTION
1. Solid carbon reacts with oxygen gas to produce carbon dioxide gas.
2. Water reacts with sodium metal to produce hydrogen gas and aqueous sodium hydroxide
3. Aqueous aluminum chloride reacts with aqueous sodium hydroxide to produce solid aluminum hydroxide
and aqueous sodium chloride.
4. Propane (C3H8) reacts with oxygen gas to produce carbon dioxide gas and water.
5. Hydrogen peroxide produces water and oxygen gas.
You might also like
- Practice Makes Perfect in Chemistry: Oxidation-ReductionFrom EverandPractice Makes Perfect in Chemistry: Oxidation-ReductionRating: 5 out of 5 stars5/5 (1)
- Practice Makes Perfect in Chemistry: Oxidation-Reduction with AnswersFrom EverandPractice Makes Perfect in Chemistry: Oxidation-Reduction with AnswersNo ratings yet
- Chapter - 8 - Chemical - Reactions - and - Equations 2Document13 pagesChapter - 8 - Chemical - Reactions - and - Equations 2b4398385No ratings yet
- CHAPTER 3: Reaction Stoichiometry (3.6-3.10)Document32 pagesCHAPTER 3: Reaction Stoichiometry (3.6-3.10)jalepeNo ratings yet
- AiTECH 4.3.-Chemical-Reactions-and-Chemical-EquationsDocument26 pagesAiTECH 4.3.-Chemical-Reactions-and-Chemical-EquationsRyan PazonNo ratings yet
- Chemistry 2 M1Document4 pagesChemistry 2 M1Rain AlmsNo ratings yet
- Unit 4R - Calculations and Chemical ReactionsDocument23 pagesUnit 4R - Calculations and Chemical ReactionsAjay0% (1)
- Chapter 8 Chemical Reactions and EquationsDocument13 pagesChapter 8 Chemical Reactions and EquationsJane JennerNo ratings yet
- Writing and Balancing Chemicak EquationDocument20 pagesWriting and Balancing Chemicak EquationJan Marlon GoritNo ratings yet
- Writing Balancing and Predicting Products of ChemicalDocument6 pagesWriting Balancing and Predicting Products of ChemicalRonald Anthony Gebilaguin BarrugaNo ratings yet
- 012.1 1b Chemical EquationDocument21 pages012.1 1b Chemical EquationC E R E B R ONo ratings yet
- Lab Act 4Document4 pagesLab Act 4Churrizo Islami100% (1)
- Lecture 2 StoichiometryDocument45 pagesLecture 2 StoichiometryKalinda MondeNo ratings yet
- CHM11-3 Balancing EquationsDocument35 pagesCHM11-3 Balancing EquationsBenmar N. OcolNo ratings yet
- Chemistry 1 Report NewDocument43 pagesChemistry 1 Report NewAlwenzel OsialNo ratings yet
- Worksheet 1 10.2Document2 pagesWorksheet 1 10.2Salman AlbastakiNo ratings yet
- CHEMICAL EQUATIONS Final VersionDocument33 pagesCHEMICAL EQUATIONS Final VersionFrancis Kirby BrutasNo ratings yet
- Chemical Equation and BalancingDocument23 pagesChemical Equation and BalancingJack Villarin100% (3)
- V22 - M4 DBA - Exam Review-Answer KeyDocument14 pagesV22 - M4 DBA - Exam Review-Answer Keypickles.squad11No ratings yet
- SK015 1.3 Stoichiometry StudentDocument73 pagesSK015 1.3 Stoichiometry StudentAazril71No ratings yet
- Chapter3 Mole ConceptDocument10 pagesChapter3 Mole Conceptmatyiman_123No ratings yet
- 8 Types of Chemical Reactions and Balancing EquationsDocument2 pages8 Types of Chemical Reactions and Balancing Equationssummer07No ratings yet
- 2324 Level L (Gr10 UAE-Gulf) Chemistry Chapter 4 NotesDocument6 pages2324 Level L (Gr10 UAE-Gulf) Chemistry Chapter 4 Noteslaithobeidat7981No ratings yet
- Chemical ReactionDocument91 pagesChemical ReactionGlebuNo ratings yet
- BLB chp3Document86 pagesBLB chp3Nora Zor-elNo ratings yet
- Module3notes - Answer KeyDocument31 pagesModule3notes - Answer Keymelissa.figueroamoralesNo ratings yet
- Stoichiometry and Mole Concept: Grade 11 Chemistry Notes 2016Document61 pagesStoichiometry and Mole Concept: Grade 11 Chemistry Notes 2016Joseph chabuNo ratings yet
- Chemical Reaction and Equation 1Document36 pagesChemical Reaction and Equation 1sameer chaharNo ratings yet
- Guide To STPM PracticalDocument17 pagesGuide To STPM Practicalhex_a_gunNo ratings yet
- Chapter 6 Chemical ReactionsDocument18 pagesChapter 6 Chemical ReactionsApril Bartolome FloresNo ratings yet
- Ace Ahead Chemistry (CD-Rom) 1st (17.2.11)Document17 pagesAce Ahead Chemistry (CD-Rom) 1st (17.2.11)helenxiaochingNo ratings yet
- Gener AL Chemi Stry 1: Week 3Document11 pagesGener AL Chemi Stry 1: Week 3Faith AsdfNo ratings yet
- Gen Chem 1 Q1 M 5-6Document12 pagesGen Chem 1 Q1 M 5-6Joselito UbaldoNo ratings yet
- Worksheet #23 - Standard Enthalpies of FormationsDocument2 pagesWorksheet #23 - Standard Enthalpies of FormationsTanishq MainiNo ratings yet
- Chapter 6 Chemical ReactionsDocument18 pagesChapter 6 Chemical ReactionsMichael Conan MaglaqueNo ratings yet
- CHEMICAL EQUATIONS-NotesDocument31 pagesCHEMICAL EQUATIONS-NotesNOELIE IBACARRANo ratings yet
- Wolaita Sodo University: General Chemistry (Chem.1012) Chapter FourDocument56 pagesWolaita Sodo University: General Chemistry (Chem.1012) Chapter FourAbdulmajid AbdellaNo ratings yet
- Balancing Chemical EquationsDocument20 pagesBalancing Chemical EquationsQueen gwen IñegoNo ratings yet
- Lesson 5 Chemical Reaction and Its EquationDocument6 pagesLesson 5 Chemical Reaction and Its EquationscientistgenerosoNo ratings yet
- Chemistry Revision 2 For Test 2: Collision Theory and Rate of ReactionDocument7 pagesChemistry Revision 2 For Test 2: Collision Theory and Rate of ReactionDaniel BerryNo ratings yet
- Reaction Stoichiometry Limiting Reagent: EnglishDocument2 pagesReaction Stoichiometry Limiting Reagent: Englishjohn baptistNo ratings yet
- Chapter 8 Chemical Reactions and EquationsDocument10 pagesChapter 8 Chemical Reactions and EquationsgustafNo ratings yet
- Chemical ReactionsDocument57 pagesChemical ReactionsCassandra DadorNo ratings yet
- Lakhmir Singh Sol Class 10 Che Chapter 1Document32 pagesLakhmir Singh Sol Class 10 Che Chapter 1Devansh GuptaNo ratings yet
- Redox Reaction Balancing PPT 2024Document30 pagesRedox Reaction Balancing PPT 2024pearcevigilanteNo ratings yet
- Problem Set 7 SolutionDocument5 pagesProblem Set 7 SolutionRod De GuzmanNo ratings yet
- Chapter 20 - Modified by Rabeay - 2022Document76 pagesChapter 20 - Modified by Rabeay - 2022s-islam.safwatNo ratings yet
- Chemistry Revision 3 For Test 2: Collision Theory and Rate of ReactionDocument8 pagesChemistry Revision 3 For Test 2: Collision Theory and Rate of ReactionDaniel BerryNo ratings yet
- Answer The Following Questions For Chemistry ReviewDocument6 pagesAnswer The Following Questions For Chemistry ReviewDwyne Kaiser GallegoNo ratings yet
- Unit 4 - Chemical ReactionsDocument36 pagesUnit 4 - Chemical Reactionssyafr.e.424No ratings yet
- Unit 5 Notes Part 2Document5 pagesUnit 5 Notes Part 2kjhghassdNo ratings yet
- Balancing Equations 10Document4 pagesBalancing Equations 10Ignacio Jr. PaguyoNo ratings yet
- Modern Chemistry Chapter 8 Chemical EquationsDocument66 pagesModern Chemistry Chapter 8 Chemical EquationsanacercetNo ratings yet
- Ws 8 Stoichiometric Calculations With Chemical ReactionsDocument2 pagesWs 8 Stoichiometric Calculations With Chemical Reactionsrdt2wwbmgkNo ratings yet
- CHEM101: General Chemistry: Chemical Reactions and Reaction StoichiometryDocument60 pagesCHEM101: General Chemistry: Chemical Reactions and Reaction Stoichiometrybarre PenroseNo ratings yet
- RedoxreactionDocument11 pagesRedoxreactionWindows AjsNo ratings yet
- Writing and Balancing Chemical Equations: Equation. Consider As An Example The Reaction Between One Methane Molecule (CHDocument5 pagesWriting and Balancing Chemical Equations: Equation. Consider As An Example The Reaction Between One Methane Molecule (CHMegan CabahugNo ratings yet
- Asexual Reproduction Worksheet: Name: . DateDocument4 pagesAsexual Reproduction Worksheet: Name: . DateMuhammadNo ratings yet
- Do Not Allow "Gravity". N MG: G GravitationalDocument7 pagesDo Not Allow "Gravity". N MG: G GravitationalMuhammadNo ratings yet
- Cells Worksheet: Name: . DateDocument5 pagesCells Worksheet: Name: . DateMuhammadNo ratings yet
- MULTIPLE CHOICE. Choose The One Alternative That Best Completes The Statement or Answers The QuestionDocument2 pagesMULTIPLE CHOICE. Choose The One Alternative That Best Completes The Statement or Answers The QuestionMuhammadNo ratings yet
- 0972 w19 GT PDFDocument1 page0972 w19 GT PDFMuhammadNo ratings yet
- ThermalDocument9 pagesThermalMuhammadNo ratings yet
- P1 HomeworkDocument5 pagesP1 HomeworkMuhammad0% (1)
- Measures of Simple Harmonic Motion: Term Example S UnitDocument1 pageMeasures of Simple Harmonic Motion: Term Example S UnitMuhammadNo ratings yet
- Worksheet Grade 8Document1 pageWorksheet Grade 8MuhammadNo ratings yet
- Chemistry For The IB MYP 4&5: by Concept: Unit PlannerDocument11 pagesChemistry For The IB MYP 4&5: by Concept: Unit PlannerMuhammad80% (5)
- Teacher Notes and Answers 11 Vibrations and WavesDocument3 pagesTeacher Notes and Answers 11 Vibrations and WavesMuhammadNo ratings yet
- Quiz Grade 10 OctoberDocument2 pagesQuiz Grade 10 OctoberMuhammadNo ratings yet
- Vibrations and Waves: Additional Practice ADocument4 pagesVibrations and Waves: Additional Practice AMuhammadNo ratings yet
- Answer Key: Holt PhysicsDocument8 pagesAnswer Key: Holt PhysicsMuhammadNo ratings yet
- Of Water (HDocument7 pagesOf Water (HMuhammadNo ratings yet
- S 2 in One Orbital P 6 in Three Orbital D 10 in Five Orbital F 14 in Seven OrbitalDocument6 pagesS 2 in One Orbital P 6 in Three Orbital D 10 in Five Orbital F 14 in Seven OrbitalMuhammadNo ratings yet
- Ag + C H ODocument9 pagesAg + C H OMuhammadNo ratings yet
- The Activation Energy of The Forward Reaction Is Shown by A. B. C. D. EDocument7 pagesThe Activation Energy of The Forward Reaction Is Shown by A. B. C. D. EMuhammadNo ratings yet
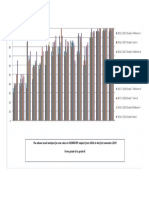
- The Above Result Analysis For One Class in CHEMISTRY Subject From 2016 To The First Semester 2019 From Grade 6 To Grade 8Document1 pageThe Above Result Analysis For One Class in CHEMISTRY Subject From 2016 To The First Semester 2019 From Grade 6 To Grade 8MuhammadNo ratings yet
- Apqp - DR SHFTDocument60 pagesApqp - DR SHFTrajesh5000100% (2)
- Sjoblom, J. - Handbook of Emulsion Technology PDFDocument731 pagesSjoblom, J. - Handbook of Emulsion Technology PDFdcharlies92% (13)
- Ultra Petronne Interior Supply Corp.: Manufacturer of Light Metal FramesDocument1 pageUltra Petronne Interior Supply Corp.: Manufacturer of Light Metal Framesjun vincint geleraNo ratings yet
- 1998-10 The Computer Paper - Ontario EditionDocument156 pages1998-10 The Computer Paper - Ontario Editionthecomputerpaper100% (1)
- Autodesk Inventor - Sheet Metal Punch IfeaturesDocument6 pagesAutodesk Inventor - Sheet Metal Punch IfeaturesNdianabasi UdonkangNo ratings yet
- Parallel Port Programming (PART 1) With CDocument13 pagesParallel Port Programming (PART 1) With ChertzoliNo ratings yet
- A Sample of Wet Soil Has A Volume of 0Document8 pagesA Sample of Wet Soil Has A Volume of 0eph0% (1)
- Aeroshell Fluid 41 PdsDocument2 pagesAeroshell Fluid 41 Pdsja.moreno930No ratings yet
- EM230 BR BM3 Fitting InstructionsDocument2 pagesEM230 BR BM3 Fitting InstructionsRoman BabaevNo ratings yet
- Nuevo CvuDocument1 pageNuevo CvuJesús GonzálezNo ratings yet
- 2013 11 DUPLEX EasyDocument21 pages2013 11 DUPLEX EasyAnonymous HC0tWeNo ratings yet
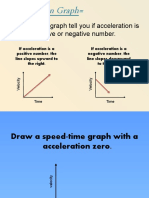
- Acceleration GrpahDocument14 pagesAcceleration GrpahRAFAEL TORRESNo ratings yet
- Simatic EKB Install 2012-03-08Document2 pagesSimatic EKB Install 2012-03-08Oton SilvaNo ratings yet
- Biomasse-HKW Sandreuth E NQDocument8 pagesBiomasse-HKW Sandreuth E NQValeriu StanNo ratings yet
- Le22a1321 AocDocument130 pagesLe22a1321 AocEchefisEchefisNo ratings yet
- " " Reach: Ordering InformationDocument8 pages" " Reach: Ordering InformationTrong TranNo ratings yet
- ED ProcessDocument9 pagesED ProcesskhanasifalamNo ratings yet
- AirStash Manual A01 PDFDocument15 pagesAirStash Manual A01 PDFArcangelo Di TanoNo ratings yet
- Huawei ACU2 Wireless Access Controller DatasheetDocument12 pagesHuawei ACU2 Wireless Access Controller Datasheetdexater007No ratings yet
- Transmicion de CF 500, CF 600 PDFDocument337 pagesTransmicion de CF 500, CF 600 PDFgusspeSNo ratings yet
- Report On Automatic Phase Changer: Submitted ByDocument32 pagesReport On Automatic Phase Changer: Submitted ByAndrea JohnsonNo ratings yet
- Concept of Circulation in A Free Vortex FlowDocument55 pagesConcept of Circulation in A Free Vortex FlowAnil KumarNo ratings yet
- High Integrity Aluminum (GENERAL)Document140 pagesHigh Integrity Aluminum (GENERAL)Kmilo Giraldo100% (3)
- The Difference Between The Internet and World Wide WebDocument3 pagesThe Difference Between The Internet and World Wide WebSonal Jain100% (1)
- Report On HACCP ManualDocument72 pagesReport On HACCP ManualRizwan KhanNo ratings yet
- Electronic Dice Using LEDs Electrical Engineering ProjectDocument16 pagesElectronic Dice Using LEDs Electrical Engineering Projectprakhar agarwal33% (3)
- Marine Lubricants Equivalents List: Sae 50 TBN 57 Sae 50 TBN 55 Sae 40 TBN 60 Sae 50 TBN 50 Sae 50 TBN 55Document3 pagesMarine Lubricants Equivalents List: Sae 50 TBN 57 Sae 50 TBN 55 Sae 40 TBN 60 Sae 50 TBN 50 Sae 50 TBN 55spyros_peiraiasNo ratings yet
- Experiment 5 DACDocument3 pagesExperiment 5 DACABHISHEK SHARMANo ratings yet
- Islamic Law - WaterDocument12 pagesIslamic Law - WaterAnum FaheemNo ratings yet
- Justine J. Beroy: Career ObjectivesDocument5 pagesJustine J. Beroy: Career ObjectivesJustine BeroyNo ratings yet