Professional Documents
Culture Documents
A2 Physics Practicals 56873669 Practical 12 The Relationship Between The Pressure and Temperature of A Gas
Uploaded by
Sachitra WijethungaOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
A2 Physics Practicals 56873669 Practical 12 The Relationship Between The Pressure and Temperature of A Gas
Uploaded by
Sachitra WijethungaCopyright:
Available Formats
Student 1 of 1
Practical 12 The relationship between the pressure and
temperature of a gas
Purpose Safety
The aim of this experiment is to investigate how Wear eye protection if your face is to be close to
the pressure of a gas changes when it is heated at the hot water.
a constant volume.
You will need:
• Round-bottomed flask • Length of rubber tubing • Water
• Temperature sensor and probe • Pressure sensor (or Bourdon gauge) • Ice
(or 0–100 °C thermometer) • Bunsen burner, tripod, gauze and mat • Eye protection
• Rubber bung with a short length of glass • Glass beaker
tube fitted through it
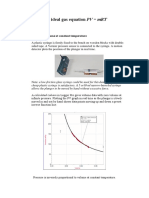
Experimental instructions
Set up the apparatus as shown in the diagram with some ice
in the water to cool it to near 0 °C.
Record the temperature of the water (effectively the
temperature of the air in the flask) and the pressure of the air
as shown on the Bourdon gauge.
Light the Bunsen burner and heat the water slowly. Record
the pressure and temperature of the air at 10-degree intervals
until the water temperature reaches 80 °C. (The temperature
air
sensor may record temperatures at equal time intervals.)
Bourdon
gauge
water
Analysis and conclusions
Plot a graph of the pressure of the trapped air ( y-axis)
heat against the temperature of the trapped air (x-axis). (Make
sure that the pressure you record is the pressure of the
trapped air, not just the excess above atmospheric pressure).
Figure 1: Investigating the relationship between It is assumed that the temperature of the trapped air will be
temperature and pressure the same as that of the water in the beaker.
Draw a second graph with the temperature axis showing minus 300 °C to plus 100 °C and
find the intercept on the pressure axis (when the gas has zero pressure). This should be at
absolute zero.
Record your value for absolute zero, suggesting any inaccuracies in your experiment and
how they might be reduced.
19
You might also like
- Chap01 Earth ScienceDocument24 pagesChap01 Earth Scienceopulithe100% (1)
- Pressure, Heat and Temperature - Physics for Kids - 5th Grade | Children's Physics BooksFrom EverandPressure, Heat and Temperature - Physics for Kids - 5th Grade | Children's Physics BooksNo ratings yet
- Practical 1 Momentum and Momentum Conservation - Large TrolleysDocument2 pagesPractical 1 Momentum and Momentum Conservation - Large TrolleysShafkat Sakeebur Rahman83% (6)
- Environmental Controls and Air Quality PropertiesDocument34 pagesEnvironmental Controls and Air Quality PropertiesXiaoyu ZhuNo ratings yet
- Refrigerator Air ConditionerDocument61 pagesRefrigerator Air ConditionerVijay GanapathyNo ratings yet
- Proselect PSTS11P52 and PSTS21P52Document1 pageProselect PSTS11P52 and PSTS21P52Ryan Murray50% (4)
- Lab Report Shell &tube Heat ExchangerDocument27 pagesLab Report Shell &tube Heat Exchangerfahmirased67% (3)
- Bell 429 Helicopter - High Resolution PanelDocument1 pageBell 429 Helicopter - High Resolution Panelguicordova0% (1)
- Lab 07 Enthalpy of Vaporization of WaterDocument4 pagesLab 07 Enthalpy of Vaporization of WaterVanesh NezhNo ratings yet
- BOQ - Bill of Quantities and Cost Sheets For MEP ProjectsDocument21 pagesBOQ - Bill of Quantities and Cost Sheets For MEP ProjectsChaithanya100% (5)
- Practical 6 Centripetal Force - Whirling BungDocument1 pagePractical 6 Centripetal Force - Whirling BungShafkat Sakeebur Rahman100% (3)
- ASTM E1641 Decomposition Kinetics by ThermogravimetryDocument7 pagesASTM E1641 Decomposition Kinetics by ThermogravimetryandresjypNo ratings yet
- Relationship between gas pressure and temperatureDocument1 pageRelationship between gas pressure and temperatureMohammed HarebNo ratings yet
- Charles Law Example Experiment - 1Document5 pagesCharles Law Example Experiment - 1nettexts75% (4)
- The Zeroth Law of ThermodynamicsDocument15 pagesThe Zeroth Law of ThermodynamicsLuqman HaqeemNo ratings yet
- Temp AirflowDocument2 pagesTemp Airflowanon_551879060No ratings yet
- Lec 5-6Document17 pagesLec 5-6Muhammad Muzammil EjazNo ratings yet
- ChE Calc - Process VariablesDocument6 pagesChE Calc - Process VariablesJumarie CoresesNo ratings yet
- Fluid Mechanics: Topic 003: PressureDocument25 pagesFluid Mechanics: Topic 003: PressureairaNo ratings yet
- Chapter 1 - Properties of Humid Air - 2017 - Air Conditioning System DesignDocument28 pagesChapter 1 - Properties of Humid Air - 2017 - Air Conditioning System Designsasha.zach1024No ratings yet
- Lab - Pressure and TemperatureDocument4 pagesLab - Pressure and Temperatureapi-383619824No ratings yet
- Temperature and Pressure ConversionsDocument9 pagesTemperature and Pressure ConversionsSafeen Y HurmzNo ratings yet
- Boyle's Law Lab Pressure-Volume RelationshipDocument1 pageBoyle's Law Lab Pressure-Volume RelationshipFrançoise NicolasNo ratings yet
- Marcet Boiler ExperimentDocument3 pagesMarcet Boiler ExperimentGarfield SmithNo ratings yet
- Basic-Thermodynamics 4Document20 pagesBasic-Thermodynamics 4Bonsay 23No ratings yet
- Lab 6 Heat of VaporizationDocument6 pagesLab 6 Heat of Vaporizationapi-458764744No ratings yet
- CHE486 - Marcet BoilerDocument6 pagesCHE486 - Marcet BoilerHaikal SuhaimiNo ratings yet
- 1.0 Introduction Properties of AirDocument86 pages1.0 Introduction Properties of AirMuhd ShazanyNo ratings yet
- PsychrometricsDocument13 pagesPsychrometricssuirezNo ratings yet
- 1920 Level MS Core Physics BTG Questions Chapters 1-2 PDFDocument48 pages1920 Level MS Core Physics BTG Questions Chapters 1-2 PDFZhaoshen Zhai100% (2)
- Koç University Physical Chemistry Lab Experiments GuideDocument42 pagesKoç University Physical Chemistry Lab Experiments GuideIreneVeladoNo ratings yet
- ME 331 Refrigeration & Air Conditioning: M AsfandyarDocument30 pagesME 331 Refrigeration & Air Conditioning: M AsfandyarSuaid Tariq BalghariNo ratings yet
- Charles' LawDocument5 pagesCharles' LawSuganya BaabuNo ratings yet
- AP Chemistry - Vaporization Pressure LabDocument5 pagesAP Chemistry - Vaporization Pressure LabJonathan ChenNo ratings yet
- Pressure and Temperature Relationship of GasDocument25 pagesPressure and Temperature Relationship of GasAhmad HilmanNo ratings yet
- Exp 9 GRP 9 20110153Document12 pagesExp 9 GRP 9 20110153Rahul KumarNo ratings yet
- Relative Humidity PDFDocument3 pagesRelative Humidity PDFMariel CondesaNo ratings yet
- Thermodynamics Module 1.lesson2Document6 pagesThermodynamics Module 1.lesson2Jhelyne FloresNo ratings yet
- Vapor Pressure LabDocument3 pagesVapor Pressure Labzky_cardonaNo ratings yet
- Version: 008 Date: Nov. 12, 1998 UpdatedDocument22 pagesVersion: 008 Date: Nov. 12, 1998 UpdatedDavidTauNo ratings yet
- MCL241 L24 RAC HVAC Psychrometry PDFDocument16 pagesMCL241 L24 RAC HVAC Psychrometry PDFAditi SamdarshiniNo ratings yet
- E4 - Marcet Boiler - NewDocument7 pagesE4 - Marcet Boiler - NewSalahuddin NorazmiNo ratings yet
- PAG 08.3 - Estimating Work Done by A Gas When Temperature IncreasesDocument3 pagesPAG 08.3 - Estimating Work Done by A Gas When Temperature IncreasesjmsonlNo ratings yet
- Exp 4 ChemistryDocument6 pagesExp 4 ChemistryMUHAMMAD ASYRAF BIN RAZALI MoeNo ratings yet
- Calculation of The Natural Unventilated Wet Bulb TemperatureDocument17 pagesCalculation of The Natural Unventilated Wet Bulb Temperaturevapaja2915No ratings yet
- Temperature Measurement: S. Estrella, and I. FrancoDocument4 pagesTemperature Measurement: S. Estrella, and I. FrancoIzy FrancoNo ratings yet
- 04 Gas LawsDocument12 pages04 Gas LawsmelissaNo ratings yet
- ME150P ELecture 1 (04 January 2011)Document115 pagesME150P ELecture 1 (04 January 2011)engrjayasis200% (1)
- Burlap and Soil Saver Products Jute Erosion Control Mesh JutDocument7 pagesBurlap and Soil Saver Products Jute Erosion Control Mesh Jutdineshv74No ratings yet
- Clausius Clap LabDocument4 pagesClausius Clap LabBunty MandaliaNo ratings yet
- Gas Law DemonstrationsDocument6 pagesGas Law Demonstrationsmayur mulikNo ratings yet
- Refrigeration Part 5 TemperatureDocument3 pagesRefrigeration Part 5 TemperaturemaloyNo ratings yet
- Gas LawsDocument13 pagesGas LawsVenkatNo ratings yet
- Marcet Boiler AppartusDocument13 pagesMarcet Boiler AppartusNIKHIL DHIMANNo ratings yet
- Trifon Ov Ivanov 2003Document5 pagesTrifon Ov Ivanov 2003ArnoldoNo ratings yet
- Boyle's Law Experiment: Last Date: 3rd Dec 2020 Sir Khurram Abbas Group 06 Experiment No: 06Document9 pagesBoyle's Law Experiment: Last Date: 3rd Dec 2020 Sir Khurram Abbas Group 06 Experiment No: 06Shayan AliNo ratings yet
- 13 DpconvDocument2 pages13 DpconvReena ThakurNo ratings yet
- Experiment 1-Vapour Pressure and Enthalpy of VaporisationDocument5 pagesExperiment 1-Vapour Pressure and Enthalpy of VaporisationStefani KavangoNo ratings yet
- Specific GravityDocument5 pagesSpecific GravityVenkiteshNo ratings yet
- Mechanical equivalent of heat experimentDocument3 pagesMechanical equivalent of heat experimentjwad masarwahNo ratings yet
- 03 CEHYDR40 - Fluid StaticsDocument34 pages03 CEHYDR40 - Fluid StaticsHarambe GorillaNo ratings yet
- Building Physics Assignment 1 Temperature and Pressure ConversionsDocument2 pagesBuilding Physics Assignment 1 Temperature and Pressure ConversionsROHIT SHRESTHANo ratings yet
- Phys 23 T2 The Constant Volume Air ThermometerDocument4 pagesPhys 23 T2 The Constant Volume Air ThermometerShashwat ChakrabortiNo ratings yet
- 1 - GasesDocument5 pages1 - GasesVon Joby RomeroNo ratings yet
- Lab 6Document6 pagesLab 6Brandon SookdeoNo ratings yet
- IDEAL GAS LAW LAB REPORTDocument5 pagesIDEAL GAS LAW LAB REPORTNor Ashikin Ismail50% (4)
- Weather Studies: The Commonwealth and International Library: Rural and Environmental Studies DivisionFrom EverandWeather Studies: The Commonwealth and International Library: Rural and Environmental Studies DivisionNo ratings yet
- A Treatise on Meteorological Instruments: Explanatory of Their Scientific Principles, Method of Construction, and Practical UtilityFrom EverandA Treatise on Meteorological Instruments: Explanatory of Their Scientific Principles, Method of Construction, and Practical UtilityNo ratings yet
- A2 Physics Practicals 56873557 Practical 9 Effect of Length and Current On The Force On A Wire in A Magnetic FieldDocument2 pagesA2 Physics Practicals 56873557 Practical 9 Effect of Length and Current On The Force On A Wire in A Magnetic FieldSachitra Wijethunga100% (1)
- A2 Physics Practicals 56873595 Practical 10 Specific Heat Capacity of A LiquidDocument2 pagesA2 Physics Practicals 56873595 Practical 10 Specific Heat Capacity of A LiquidSachitra Wijethunga100% (2)
- Practical 8 Measuring The Charge Stored by A CapacitorDocument1 pagePractical 8 Measuring The Charge Stored by A CapacitorShafkat Sakeebur Rahman100% (1)
- Practical 11 Specific Heat Capacity of A SolidDocument2 pagesPractical 11 Specific Heat Capacity of A SolidShafkat Sakeebur RahmanNo ratings yet
- Practical 4 Rate of Change of Momentum Using A Linear Air TrackDocument2 pagesPractical 4 Rate of Change of Momentum Using A Linear Air TrackShafkat Sakeebur RahmanNo ratings yet
- A2 Physics Practicals 56873406 Practical 5 Rate of Change of Momentum Using A TrolleyDocument2 pagesA2 Physics Practicals 56873406 Practical 5 Rate of Change of Momentum Using A TrolleySachitra Wijethunga100% (1)
- GCE in Chemistry Supra Molecular Chemistry CSDocument10 pagesGCE in Chemistry Supra Molecular Chemistry CSSachitra WijethungaNo ratings yet
- Practical 7 Centripetal Force - Rotating TrolleyDocument1 pagePractical 7 Centripetal Force - Rotating TrolleyShafkat Sakeebur Rahman100% (2)
- Practical 2 Momentum and Momentum Conservation - Small TrolleysDocument1 pagePractical 2 Momentum and Momentum Conservation - Small TrolleysShafkat Sakeebur Rahman100% (3)
- A2 Physics Practicals 56873326 Practical 3 Momentum and Momentum Conservation Using A Linear Air TrackDocument2 pagesA2 Physics Practicals 56873326 Practical 3 Momentum and Momentum Conservation Using A Linear Air TrackSachitra Wijethunga0% (1)
- GCE in Chemistry Green Chemistry CSDocument13 pagesGCE in Chemistry Green Chemistry CSSachitra WijethungaNo ratings yet
- GCE in Chemistry Analysis of Chemicals A2 CSDocument16 pagesGCE in Chemistry Analysis of Chemicals A2 CSSachitra WijethungaNo ratings yet
- GCE in Chemistry Analysis of Chemicals As CSDocument14 pagesGCE in Chemistry Analysis of Chemicals As CSSachitra WijethungaNo ratings yet
- GCE Chemistry The Atomosphere Context Study EDEXCELDocument18 pagesGCE Chemistry The Atomosphere Context Study EDEXCELShuaib NaximNo ratings yet
- Edexcel Biology A2 2008 Jan MsDocument4 pagesEdexcel Biology A2 2008 Jan MsSachitra WijethungaNo ratings yet
- GCE in Chemistry Fuel CellsDocument13 pagesGCE in Chemistry Fuel CellsSachitra Wijethunga100% (1)
- Edexcel Biology A2 June 2009 MSDocument107 pagesEdexcel Biology A2 June 2009 MSSachitra WijethungaNo ratings yet
- Edexcel Biology A2 2011 Jan ERDocument42 pagesEdexcel Biology A2 2011 Jan ERSachitra Wijethunga100% (1)
- Edexcel Biology A2 June 2009 MSDocument107 pagesEdexcel Biology A2 June 2009 MSSachitra WijethungaNo ratings yet
- Edexcel Biology A2 2008 June MsDocument6 pagesEdexcel Biology A2 2008 June MsSachitra WijethungaNo ratings yet
- June 2007 - All UnitsDocument90 pagesJune 2007 - All Unitsapi-3726022No ratings yet
- Edexcel Biology A2 2010 June ERDocument10 pagesEdexcel Biology A2 2010 June ERSachitra WijethungaNo ratings yet
- 6bi07 Rep June 09Document9 pages6bi07 Rep June 09alevelscienceNo ratings yet
- Edexcel Biology A2 2008 May QPDocument16 pagesEdexcel Biology A2 2008 May QPSachitra WijethungaNo ratings yet
- Edexcel Biology A2 2009 Jan QPDocument12 pagesEdexcel Biology A2 2009 Jan QPSachitra WijethungaNo ratings yet
- June 2007 - All UnitsDocument90 pagesJune 2007 - All Unitsapi-3726022No ratings yet
- Jan 2007 - All UnitsDocument31 pagesJan 2007 - All Unitsapi-3726022100% (2)
- Final Exam Gen - Chem 1Document7 pagesFinal Exam Gen - Chem 1Ricardo VillanuevaNo ratings yet
- Binkley Optimize Design For Distillation Feed PDFDocument7 pagesBinkley Optimize Design For Distillation Feed PDFRajendraNo ratings yet
- Nylon 66 Technical Data SheetDocument1 pageNylon 66 Technical Data SheetRajeev BujjiNo ratings yet
- APCI Process Analysis and OptimizationDocument10 pagesAPCI Process Analysis and OptimizationKunal KhandelwalNo ratings yet
- Case Studies in Thermal Engineering: Rubens E.N. Castro, Rita M.B. Alves, Claudio A.O. NascimentoDocument14 pagesCase Studies in Thermal Engineering: Rubens E.N. Castro, Rita M.B. Alves, Claudio A.O. NascimentoIstiqamah HarnamaNo ratings yet
- Epa Method Io3-1Document30 pagesEpa Method Io3-1Eddie MendozaNo ratings yet
- SavemyexamsDocument21 pagesSavemyexamsnitadarwina4No ratings yet
- Calculate Capacity of Room Air Conditioner: Room Detail Unit Electrical Appliances in The RoomDocument2 pagesCalculate Capacity of Room Air Conditioner: Room Detail Unit Electrical Appliances in The Roomzmei23No ratings yet
- Concepts in Condenser System (Vacuum Equipment)Document7 pagesConcepts in Condenser System (Vacuum Equipment)Mustafa DemircioğluNo ratings yet
- On The Definition of Propellant Force: SciencedirectDocument4 pagesOn The Definition of Propellant Force: SciencedirectMarcelo MartinezNo ratings yet
- Sta. Elena Integrated School 4 Periodical Examination in Science 7Document3 pagesSta. Elena Integrated School 4 Periodical Examination in Science 7Hazale AzasNo ratings yet
- BQ - KK3 Karak Bentong 1st Quote PDFDocument6 pagesBQ - KK3 Karak Bentong 1st Quote PDFAbu Harith HanzalahNo ratings yet
- Moist Heat Sterilization Highlights On Equivalent Time F0Document5 pagesMoist Heat Sterilization Highlights On Equivalent Time F0Rushyanth KR100% (1)
- HT Assignment 1 (2019)Document2 pagesHT Assignment 1 (2019)Mayank AgrawalNo ratings yet
- Fluent Tips and TricksDocument75 pagesFluent Tips and TricksAliAAdibNo ratings yet
- Scroll-Chillers Catalog High-ResDocument12 pagesScroll-Chillers Catalog High-ResubaidNo ratings yet
- Venkitaraj2019 (1) New NewDocument12 pagesVenkitaraj2019 (1) New Newramesh tNo ratings yet
- Heat Transfer Characteristics During Melting and Solidification of Phase Change Energy Storage ProcessDocument8 pagesHeat Transfer Characteristics During Melting and Solidification of Phase Change Energy Storage Processatulsharma74No ratings yet
- MHCCW Chilled Water Ceiling Fan Coil GuideDocument34 pagesMHCCW Chilled Water Ceiling Fan Coil GuideWESHIMNo ratings yet
- Rth6450d 5 1 1 Programmable Thermostat Installation ManualDocument64 pagesRth6450d 5 1 1 Programmable Thermostat Installation ManualITLHAPNNo ratings yet
- Folletos 27 2 2018 1Document2 pagesFolletos 27 2 2018 1Refrigeración EverestNo ratings yet
- Chemical Process Modeling in Modelica: September 2012Document9 pagesChemical Process Modeling in Modelica: September 2012EmBRONo ratings yet
- Wellbore Models GWELL, GWNACL, and HOLA User's Guide PDFDocument114 pagesWellbore Models GWELL, GWNACL, and HOLA User's Guide PDFWisnu ArdhiNo ratings yet