Professional Documents
Culture Documents
Otto Cycle
Uploaded by
Saraju NandiCopyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Otto Cycle
Uploaded by
Saraju NandiCopyright:
Available Formats
Gas Power Cycles
Prof. U.S.P. Shet , Prof. T. Sundararajan and Prof. J.M . Mallikarjuna
4.4 Air Standard Otto Cycle:
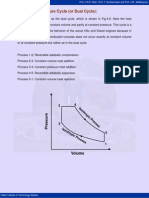
The air-standard-Otto cycle is the idealized cycle for the spark-ignition internal combustion engines. This cycle is shown above on p-v and T-s diagrams. The Otto cycle 1-2-3-4 consists of following four process:
Process 1-2: Reversible adiabatic compression of air. Process 2-3: Heat addition at constant volume. Process 3-4: Reversible adiabatic expansion of air. Process 4-1: Heat rejection at constant volume.
2 4
Volume
Indian Institute of Technology Madras
Gas Power Cycles
Prof. U.S.P. Shet , Prof. T. Sundararajan and Prof. J.M . Mallikarjuna
2 4 1
Entropy
Fig.4.4. Otto cycle on p-v and T-s diagrams
Air Standard Efficiency:
th = Net workdone Net heat added
Since processes 1-2 and 3-4 are adiabatic processes, the heat transfer during the cycle takes place only during processes 2-3 and 4-1 respectively. Therefore, thermal efficiency can be written as,
th =
Heat added - Heat rejected Heat added
Consider m kg of working fluid,
Heat added = mCV ( T3 - T2 ) Heat Rejected = mCV ( T4 - T1 )
th = mCV ( T3 - T2 ) - mCV ( T4 - T1 ) mCV ( T3 - T2 ) =1T4 - T1 T3 - T2
Indian Institute of Technology Madras
Gas Power Cycles
Prof. U.S.P. Shet , Prof. T. Sundararajan and Prof. J.M . Mallikarjuna
For the reversible adiabatic processes 3-4 and 1-2, we can write,
v T4 = 3 T3 v4
-1
V T and 1 = 2 T2 V1
-1
v 2 = v3 and v 4 = v1
V T4 T T -T = 1 = 4 1 = 2 T3 T2 T3 - T2 V1
1
th
The ratio
V T1 = 1- 2 = 1 T2 V1
-1
V1 is called as compression ratio, r. V2
th 1 =1- r
-1
From the above equation, it can be observed that the efficiency of the Otto cycle is mainly the function of compression ratio for the given ratio of Cp and Cv. If we plot the variations of the thermal efficiency with increase in compression ratio for different gases, the curves are obtained as shown in Fig.4.4.1. Beyond certain values of compression ratios, the increase in the thermal efficiency is very small, because the curve tends to be asymptotic. However, practically the compression ratio of petrol engines is restricted to maximum of 9 or 10 due to the phenomenon of knocking at high compression ratios.
Indian Institute of Technology Madras
Gas Power Cycles
Prof. U.S.P. Shet , Prof. T. Sundararajan and Prof. J.M . Mallikarjuna
=1.67
=1.40
=1.30
Compression ratio,r
Effect of CR and on efficiency for Otto cycle.
Fig.4.4.1. Variation of thermal efficiency with compression ratio
Mean Effective Pressure:
Generally, it is defined as the ratio of the net workdone to the displacement volume of the piston. Let us consider m kg of working substance.
Net work done = m Cv {( T3 - T2 ) - ( T4 - T1 )}
Displacement Volume =
( V1
- V2 )
m R T1 r - 1 1 = V1 1 - = r P1 r
since, R = Cv ( - 1)
m C v ( - 1) T1 r - 1 P1 r
Indian Institute of Technology Madras
Gas Power Cycles
Prof. U.S.P. Shet , Prof. T. Sundararajan and Prof. J.M . Mallikarjuna
mep =
m C v ( T3 - T2 ) m C v ( -1) T1 P1
- ( T4 - T1 ) r - 1 r
1 p1 r - 1 T1 r - 1
-1
{( T3
- T2 ) - ( T4 - T1
)}
Now,
T2 = T1 ( r )
Let,
rp =
P3 T3 = = Pressure ratio P2 T2
P3 T2 = rp T2 = rp r -1 T1 P2
-1
T3 =
(for V = C)
So,
T4
1 = T3 r
= rp r
-1
1 T1 r
-1
= rp T1
mep =
P1 r ( r - 1) ( - 1)
{( r
r -1 - r -1
) - ( rp - 1)}
r -1 r - 1 - r - 1 p p = P1 r ( - 1) ( r - 1)
) (
r -1 - 1 r - 1 p mep = P1 r ( r - 1) ( - 1)
)(
Indian Institute of Technology Madras
You might also like
- Experimental Fluid Mechanics: The Commonwealth and International Library: Thermodynamics and Fluid Mechanics DivisionFrom EverandExperimental Fluid Mechanics: The Commonwealth and International Library: Thermodynamics and Fluid Mechanics DivisionRating: 4 out of 5 stars4/5 (2)
- Stabilization and Dynamic of Premixed Swirling Flames: Prevaporized, Stratified, Partially, and Fully Premixed RegimesFrom EverandStabilization and Dynamic of Premixed Swirling Flames: Prevaporized, Stratified, Partially, and Fully Premixed RegimesNo ratings yet
- Module 3 IC EnginesDocument70 pagesModule 3 IC EnginesAnkitNo ratings yet
- Pokhara University Service Commission Curriculum For Assistant Administrative (Lab) Level ExaminationDocument27 pagesPokhara University Service Commission Curriculum For Assistant Administrative (Lab) Level ExaminationHonnyes100% (1)
- Reviewer 5Document59 pagesReviewer 5KleberNo ratings yet
- Classroom Activities in Aerodynamics: © 2000 Dr. Charley Rodriguez, PH.DDocument21 pagesClassroom Activities in Aerodynamics: © 2000 Dr. Charley Rodriguez, PH.DAerocse EceitmechNo ratings yet
- Propeller FundamentalDocument7 pagesPropeller FundamentalSharath UpadhyaNo ratings yet
- Incompressible Flow Over Finite Wings-IntroDocument11 pagesIncompressible Flow Over Finite Wings-IntroJanuarSianiparNo ratings yet
- O Ring GB en PDFDocument215 pagesO Ring GB en PDFPéter BorbásNo ratings yet
- Experimental Stress AnalysisDocument194 pagesExperimental Stress AnalysisJEEVA KUMAR M100% (1)
- AF Structure ContDocument21 pagesAF Structure ContlongmilkNo ratings yet
- Mooring, Towing, TaxingDocument4 pagesMooring, Towing, TaxingRodriguez ArthursNo ratings yet
- Exercises: Flight Dynamics-I Prof. E.G. Tulapurkara Chapter-5Document1 pageExercises: Flight Dynamics-I Prof. E.G. Tulapurkara Chapter-5Chegrani AhmedNo ratings yet
- Aircraft Pneumatic SystemsDocument46 pagesAircraft Pneumatic SystemsSasiKumar100% (1)
- IC EnginesDocument14 pagesIC EnginesPradeep N B100% (1)
- Study Question! Powerplant Section (Gas Turbine)Document8 pagesStudy Question! Powerplant Section (Gas Turbine)IdrisNo ratings yet
- E5 Lab Sheet Measurement of Jet ForcesDocument9 pagesE5 Lab Sheet Measurement of Jet ForcesHoongNo ratings yet
- Club66 17.5 (Ice Protection)Document5 pagesClub66 17.5 (Ice Protection)Tharrmaselan manimaranNo ratings yet
- Wind Tunnel InroductionDocument38 pagesWind Tunnel Inroductionmich48chinNo ratings yet
- Radial TurbinesDocument46 pagesRadial TurbinesarorabbNo ratings yet
- Smoke Tunnel ReportDocument11 pagesSmoke Tunnel ReportRana HanzlaNo ratings yet
- Airframe Continuation!...Document11 pagesAirframe Continuation!...IdrisNo ratings yet
- YhhjjDocument52 pagesYhhjjSam CunananNo ratings yet
- Module 11.5.1 NotesDocument9 pagesModule 11.5.1 Notesmanu gargNo ratings yet
- Aeroplane Systems Objective Questions DATE:16/04/2016: 4) Class DDocument4 pagesAeroplane Systems Objective Questions DATE:16/04/2016: 4) Class DrashmiameNo ratings yet
- DMX5533 Tma01 2017 2018Document7 pagesDMX5533 Tma01 2017 2018Shenal FernandoNo ratings yet
- AC Knowedge 3 146questions +answersDocument20 pagesAC Knowedge 3 146questions +answersmoik ojooNo ratings yet
- Fuel Injection & TurbochargingDocument4 pagesFuel Injection & Turbocharging6 avinashNo ratings yet
- Chapter 4 - Airfoil and Geometry SelectionDocument75 pagesChapter 4 - Airfoil and Geometry SelectionSaad MasoodNo ratings yet
- MECH 554 Project II Spring2021Document5 pagesMECH 554 Project II Spring2021sannyalNo ratings yet
- Check of MLG Torque Links For Excessive PlayDocument37 pagesCheck of MLG Torque Links For Excessive PlayColin Shane MamalesNo ratings yet
- Airframe Maintenance and Repair - QBDocument5 pagesAirframe Maintenance and Repair - QBandrw1987No ratings yet
- Jawaban PropulsiDocument35 pagesJawaban PropulsiyandaNo ratings yet
- Total Question OutDocument31 pagesTotal Question OutJoe Ho100% (2)
- Forces and MomentsDocument13 pagesForces and Momentsanon_287164994No ratings yet
- Rockets and Missiles Question BankDocument5 pagesRockets and Missiles Question BankkannanNo ratings yet
- Axial Flow Turbines - Dr.K.M.ParammasivamDocument22 pagesAxial Flow Turbines - Dr.K.M.Parammasivammaran2326No ratings yet
- Aero 7sem HADocument96 pagesAero 7sem HAKumarGuglothNo ratings yet
- 4.3 ServomechanismsDocument65 pages4.3 ServomechanismsYousef Asmar100% (1)
- Data Collection and Preliminary 3 View DesignDocument5 pagesData Collection and Preliminary 3 View DesignBrijesh PatelNo ratings yet
- Aerodynamic Performance Analysis of A Non Planar C-Wing Using Experimental and Numerical ToolsDocument28 pagesAerodynamic Performance Analysis of A Non Planar C-Wing Using Experimental and Numerical ToolsGiang Nguyễn TrườngNo ratings yet
- EME Notes IC EnginesDocument11 pagesEME Notes IC EnginesKarthik NerellaNo ratings yet
- GATE Aero-Propulsion by IGCDocument256 pagesGATE Aero-Propulsion by IGCParth PatelNo ratings yet
- Neral Capítulo.1 PDFDocument8 pagesNeral Capítulo.1 PDFemhidalgoNo ratings yet
- AE8302-Elements of Aeronautical Engineering: Reg. NoDocument2 pagesAE8302-Elements of Aeronautical Engineering: Reg. NoViswanath ViswaNo ratings yet
- M 17Document5 pagesM 17amhm2000100% (1)
- Iii Mech Vi Sem QB 2013 2014 Even PDFDocument137 pagesIii Mech Vi Sem QB 2013 2014 Even PDFSiva RamanNo ratings yet
- Essays Module 7Document3 pagesEssays Module 7Mohd Shah100% (2)
- Fluid Mechanics Chapter 6Document34 pagesFluid Mechanics Chapter 6Ricky Mak100% (1)
- PTC B1.1 Notes - Sub Module 17.1 (Fundamentals)Document44 pagesPTC B1.1 Notes - Sub Module 17.1 (Fundamentals)DrAmir AhmedNo ratings yet
- Airframe and Powerplant ReviewerDocument45 pagesAirframe and Powerplant ReviewerBobby SereNo ratings yet
- Nptel - Ac.in Courses IIT-MADRAS Applied Thermodynamics Module 4 6 AsdcDocument4 pagesNptel - Ac.in Courses IIT-MADRAS Applied Thermodynamics Module 4 6 AsdcSanjeev PrakashNo ratings yet
- Limited Pressure CycleDocument4 pagesLimited Pressure Cyclecasu19No ratings yet
- Stirling CycleDocument3 pagesStirling CycleRon QuerubinNo ratings yet
- Stirling CycleDocument6 pagesStirling CycleChristian_Joe__9147No ratings yet
- 2 Carnot CycleDocument6 pages2 Carnot CycleSamujawal Kumar SumanNo ratings yet
- IC Engines 2012 Edition Theory & QuestionsDocument178 pagesIC Engines 2012 Edition Theory & Questionskumarrohit91No ratings yet
- Lecture Notes OnDocument200 pagesLecture Notes Onananth k r100% (3)
- U3 New Packages: Special For OctoberDocument6 pagesU3 New Packages: Special For OctoberSaraju NandiNo ratings yet
- Alternatives To Petroleum-Based EnergyDocument9 pagesAlternatives To Petroleum-Based EnergySaraju NandiNo ratings yet
- Alternatives To Petroleum-Based EnergyDocument9 pagesAlternatives To Petroleum-Based EnergySaraju NandiNo ratings yet
- 7 Limited Pressure CycleDocument4 pages7 Limited Pressure CyclecaptainhassNo ratings yet
- BearingDocument1 pageBearingSaraju NandiNo ratings yet
- BearingDocument1 pageBearingSaraju NandiNo ratings yet
- Fundamental of Mechanical Engineering & Mechatronics MCQsDocument36 pagesFundamental of Mechanical Engineering & Mechatronics MCQsAshish VermaNo ratings yet
- V.Senthil Me WWW - Chennaituitions.in: Page 1 of 6Document6 pagesV.Senthil Me WWW - Chennaituitions.in: Page 1 of 6saranyakathiravan100% (1)
- 43 Austrian Chemistry Olympiad National Competition Theoretical Tasks 2017-05-25Document35 pages43 Austrian Chemistry Olympiad National Competition Theoretical Tasks 2017-05-25Quốc NguyễnNo ratings yet
- Formula Notes Ic Engine 28 01 20 1 65Document12 pagesFormula Notes Ic Engine 28 01 20 1 65OpenFOAMNo ratings yet
- Chapter 1 Introduction To Internal Combustion EngineDocument34 pagesChapter 1 Introduction To Internal Combustion EngineYeet server YesNo ratings yet
- Thermodynamic CyclesDocument9 pagesThermodynamic CyclesPilar Guzmán MartínezNo ratings yet
- Thermodynamics Lecture PDFDocument11 pagesThermodynamics Lecture PDFGenerale, Rey marck C.No ratings yet
- Mech Engg Question Bank - I Sem For MechanicalDocument124 pagesMech Engg Question Bank - I Sem For Mechanicalanon_102708056No ratings yet
- Gas Engines FundamentalsDocument64 pagesGas Engines FundamentalsAjmal Ansary100% (3)
- Combustion Plate 1Document26 pagesCombustion Plate 1Marc Bitang100% (3)
- Model Papers For 2nd Year AutoDocument40 pagesModel Papers For 2nd Year AutoOmar Rashdi83% (6)
- Chapter 3 Gas Power CyclesDocument21 pagesChapter 3 Gas Power CyclesHoàng KhôiNo ratings yet
- GTZ - Engines For Biogas - 1988Document164 pagesGTZ - Engines For Biogas - 1988Elias JourdyNo ratings yet
- Air Standard CyclesDocument23 pagesAir Standard CyclesNik HenNo ratings yet
- Mechanical Technical Interview Questions For ThermodynamicsDocument5 pagesMechanical Technical Interview Questions For Thermodynamicsjames saiji singh100% (1)
- Clil MeccanicaDocument5 pagesClil MeccanicaNiclae CobahmatencoNo ratings yet
- Assignment 3Document2 pagesAssignment 3younkushanks0% (1)
- Kocaeli University Internal Combustion Engines SPRING 2021-2022 Homework 5Document4 pagesKocaeli University Internal Combustion Engines SPRING 2021-2022 Homework 5nebghostNo ratings yet
- Power and Industrial Plant Engineering By:AlcorconDocument126 pagesPower and Industrial Plant Engineering By:AlcorconNelson Naval Cabingas77% (13)
- Stirling EngineDocument11 pagesStirling EngineMario MikulandraNo ratings yet
- Internal Combustion Engines: Cycle Analysis (L03)Document18 pagesInternal Combustion Engines: Cycle Analysis (L03)Shizza MianNo ratings yet
- I C Engine TerminologyDocument47 pagesI C Engine TerminologyShubham SharmaNo ratings yet
- 11 Gas Power CycleDocument20 pages11 Gas Power CycleRishabh KaushikNo ratings yet
- Introduction To ThermodynamicsDocument130 pagesIntroduction To ThermodynamicsJaimin Joshi0% (1)
- Fajril Ar Rahman 100401014Document4 pagesFajril Ar Rahman 100401014Fajril Ar-rahmanNo ratings yet
- Activity 1 in PpeDocument4 pagesActivity 1 in PpeJenny Mae Pomeda100% (1)
- Etlab - View Result - Exam 3 - ThermodynamicsDocument6 pagesEtlab - View Result - Exam 3 - ThermodynamicsMevin JacobNo ratings yet
- r7210305 ThermodynamicsDocument4 pagesr7210305 ThermodynamicssivabharathamurthyNo ratings yet
- 4 5902085539233794318Document366 pages4 5902085539233794318Angel Jesús Agapito Cortez100% (6)
- Thermodynamic Cycle PresentationDocument29 pagesThermodynamic Cycle PresentationAnonymous Oh1pxYX30% (1)