Professional Documents
Culture Documents
4 Ligand Field Theory
Uploaded by
Yeraldith RojasOriginal Description:
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
4 Ligand Field Theory
Uploaded by
Yeraldith RojasCopyright:
Available Formats
Chemistry 3820 (Fall 2006)
MO Diagrams for O2 and N2
Drawing MO diagrams - always same number of MOs as AOs. - more electronegative element on the right - lower in energy. ------------------------------------------------------------------------------------------------------------------
O2
* 2p
2p
*2p
Paramagnetic Bond order of 2 (O=O)
2p
2p
2 MOs like this perpendicular to each other
2p *2s
larger E
2s
2s
2s O O2 O
------------------------------------------------------------------------------------------------------------------
N2
*2p
Diamagnetic Bond order of 3 (N= N)
*2p
2p
2p
2p
Very strong NN bond Unlike O2, the 2p and 2p orbitals are switched in energy (see next page)
smaller E
2p *2s 2s N N2 N
HOMO = 2p, LUMO = *2p
2s
2s
2006 - Dr. Paul G. Hayes University of Lethbridge
Chemistry 3820 (Fall 2006) Ordering of MOs in O2 is 2p , 2p, *2p , *2p but in N2 and CO, the order is 2p, 2p , *2p , *2p
Reason - O2 - larger E between s and p orbitals less orbital mixing - larger E because across the period (B,C,N,O,F) more protons added to the nucleus, which pulls the electrons in closer to the nucleus (lower in energy). This effect is felt more strongly by the s-orbitals than the p -orbitals, so the energy of the s-orbitals drops more rapidly than that of the p-orbitals. - N2 and CO - mixing between the 2p and the *2s orbitals raises the energy of the 2p above the 2p.
Changes in the energy of the MOs of homonuclear diatomic molecules in the 2 nd period
MO Diagram for CO
2006 - Dr. Paul G. Hayes University of Lethbridge
Chemistry 3820 (Fall 2006)
*2p
2p
*2p 2p
LUMO
2p
HOMO (slightly antibonding)
2p
2s
* 2s 2s C CO O
2s
- CO is isoelectronic with N2 (C has one less electron than N, O has one more) MO diagram is very similar to that of N2. - Diamagnetic, Bond order of 3 (C= O) - Important: HOMO = 2p (weakly antibonding), LUMO = *2p (strongly antibonding) - Since O is far more electronegative than C, would expect a large dipole moment with - on O. However, CO actually has only a small dipole moment (0.1 Debye) with the - on Carbon! Usually electronegativities are a good indication of the direction and magnitude of the dipole on a molecule, but especially in molecules where orbitals with antibonding character are occupied, things are not so straightforward.
2006 - Dr. Paul G. Hayes University of Lethbridge
Chemistry 3820 (Fall 2006)
Pictorial Representations of Possible Bonds
2006 - Dr. Paul G. Hayes University of Lethbridge
Chemistry 3820 (Fall 2006)
MO theory ~ Ligand Field Theory Constructing an MO diagram for an octahedral complex
Symmetry Adapted combinations of Ligand -orbitals in an Octahedral Complex
- any combination between t2g and ligands becomes non-bonding
In an octahedral environment, the metal orbitals (3d, 4s, 4p for a 1st row TM) divide by symmetry into 4 sets: s = a1g , p = t1u , axial d = eg , inter-axial d = t 2g. The orbitals of the six ligands can be combined to give six symmetry-adapted linear combinations which are of the correct symmetry to interact with the s, 3 x p and 2 x axial-d orbitals, but not the inter-axial d orbitals. The result is that 3 orbitals (the inter-axial d-orbitals) are non-bonding, while the rest (6 metal orbitals and 6 ligand orbitals) combine to form six bonding and six anti-bonding MOs. See next page. This is a much more correct approach than crystal field theory, but is not as easy to use.
2006 - Dr. Paul G. Hayes University of Lethbridge
Chemistry 3820 (Fall 2006)
MO energy levels for an octahedral complex (only -bonding considered)
t1u*
4p (t1u)
a1g*
4s (a1g) eg* O 3d (eg + t2g) t2g
(a 1g + t1u + e g) eg
TM
6L t1u a1g
The six bonding orbitals are filled with 12 electrons from the six ligands Orbitals shown in red (t2g and eg *) are the frontier orbitals where d-electrons reside (which is why TM MOs are often simplified to show only t 2g and eg * orbitals). 2006 - Dr. Paul G. Hayes University of Lethbridge
Chemistry 3820 (Fall 2006)
-bonding ligands (the above MO diagram does not take into account -bonding)
M L M L filled orbital -donor NH3 , CH3 -, H M L M L M L empty orbital
-donor Cl-, OH -, NR 2- , OH 2
-acceptor CO, NO+, CN -
For -acceptor ligands, the bonding is SYNERGIC: -donation to the metal strengthens -backbonding to the ligand, and -donation from the metal to the ligand strengthens the -donor component of bonding. This is because -donation leads to increased electron density on the metal, which allows increased -backdonation. Conversely, -backdonation reduces the amount of electron density on the metal, which allows more -donation from the ligand to the metal.
Cr(CO)6: Octahedral complex with good -acceptor ligands
-interaction M lobe of acceptor orbital (whole orbital not shown) C O M slightly antibonding HOMO of CO M axial d-orbitals C O C O -interaction
strongly antibonding * orbitals of CO
2006 - Dr. Paul G. Hayes University of Lethbridge
Chemistry 3820 (Fall 2006)
MO energy levels for an octahedral complex with -acceptor ligands (e.g. [Cr(CO)6])
t1u*
4p (t1u)
a1g*
4s (a1g)
t2g* eg*
* orbitals of CO (t2g)
3d (eg + t2g)
large O
t2g (a1g + t1u + eg) eg
Cr
6 CO t1u a 1g
-backdonation to CO from the t2g orbitals (which are non-bonding in the absence of -interactions between the metal and the ligands). The 3 t2g orbitals and 3 high lying * orbitals of the CO ligands form 3 bonding MOs and 3 antibonding MOs. Since the CO * orbitals are empty, the d -electrons occupy the bonding MO from this interaction. The result is (1) a very large o , so the e g* orbital is likely to remain empty. (2) the t2g orbital is strongly bonding (wants to be filled with 6 electrons) complexes of strong -acceptor ligands are the most likely to obey the 18 electron rule 2006 - Dr. Paul G. Hayes University of Lethbridge
Chemistry 3820 (Fall 2006)
Symmetry Adapted Linear Combinations of * orbitals in ML6 complexes
Q. Why are there only three ligand -acceptor orbitals shown in the MO diagram for CO when there are 6 ligands, each with two empty * orbitals ? A. The 12 * orbitals of the ligands can be combined to form 12 symmetry adapted linear combinations of atomic orbitals (3 x T1u , 3 x T2g , 3 x T1g and 3 x T2u). Only the three T2g linear combinations are of the correct symmetry to interact with the t2g orbitals (dxy , dxz, dyz) on the metal.
2006 - Dr. Paul G. Hayes University of Lethbridge
Chemistry 3820 (Fall 2006)
10
Simplified picture of how -acceptor and -donor interactions affect the MO diagram - Only the frontier orbitals are shown
t 2g *
eg*
eg * L large O
* (t2g)
t 2g TM complex with -bonding only
-acceptor ligands increase O
t2g [Cr(CO)6] with -backdonation from Cr to CO
eg *
eg * small O t 2g*
-donor ligands decrease O
t 2g
TM complex with -bonding only
t 2g TM complex with donation from Ligand to Metal
-donor ligands -donation from the ligands to the t2g orbitals the 3 t2g metal orbitals and 3 low lying, filled ligand orbitals of -symmetry form 3 bonding MOs and 3 antibonding MOs (thus t g is 2 lowered and o increases). Since the interacting ligand orbitals are full, these electrons occupy the bonding MO from this interaction, and the d-electrons occupy the antibonding MO. The result is (1) a small o (2) the t2g orbital is weakly antibonding 2006 - Dr. Paul G. Hayes University of Lethbridge
Chemistry 3820 (Fall 2006)
11
MO Diagram for a Tetrahedral Complex
e.g. [Ni(PPh3)4] (Ni0 , d10 , 18-electron complex)
t2*
4p (t 1u)
a 1*
4s (a1g) t2*
t ~ 4/9 o
3d (eg + t2g) e
(a1g + t1u + eg) a1
Ni
4 PPh 3
t2
2006 - Dr. Paul G. Hayes University of Lethbridge
Chemistry 3820 (Fall 2006)
12
Hard and Soft Acids and Bases
2006 - Dr. Paul G. Hayes University of Lethbridge
You might also like
- Shoe Dog: A Memoir by the Creator of NikeFrom EverandShoe Dog: A Memoir by the Creator of NikeRating: 4.5 out of 5 stars4.5/5 (537)
- Grit: The Power of Passion and PerseveranceFrom EverandGrit: The Power of Passion and PerseveranceRating: 4 out of 5 stars4/5 (587)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceFrom EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceRating: 4 out of 5 stars4/5 (894)
- The Yellow House: A Memoir (2019 National Book Award Winner)From EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Rating: 4 out of 5 stars4/5 (98)
- The Little Book of Hygge: Danish Secrets to Happy LivingFrom EverandThe Little Book of Hygge: Danish Secrets to Happy LivingRating: 3.5 out of 5 stars3.5/5 (399)
- On Fire: The (Burning) Case for a Green New DealFrom EverandOn Fire: The (Burning) Case for a Green New DealRating: 4 out of 5 stars4/5 (73)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeFrom EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeRating: 4 out of 5 stars4/5 (5794)
- Never Split the Difference: Negotiating As If Your Life Depended On ItFrom EverandNever Split the Difference: Negotiating As If Your Life Depended On ItRating: 4.5 out of 5 stars4.5/5 (838)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureFrom EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureRating: 4.5 out of 5 stars4.5/5 (474)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryFrom EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryRating: 3.5 out of 5 stars3.5/5 (231)
- The Emperor of All Maladies: A Biography of CancerFrom EverandThe Emperor of All Maladies: A Biography of CancerRating: 4.5 out of 5 stars4.5/5 (271)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreFrom EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreRating: 4 out of 5 stars4/5 (1090)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyFrom EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyRating: 3.5 out of 5 stars3.5/5 (2219)
- Team of Rivals: The Political Genius of Abraham LincolnFrom EverandTeam of Rivals: The Political Genius of Abraham LincolnRating: 4.5 out of 5 stars4.5/5 (234)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersFrom EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersRating: 4.5 out of 5 stars4.5/5 (344)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaFrom EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaRating: 4.5 out of 5 stars4.5/5 (265)
- The Unwinding: An Inner History of the New AmericaFrom EverandThe Unwinding: An Inner History of the New AmericaRating: 4 out of 5 stars4/5 (45)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)From EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Rating: 4.5 out of 5 stars4.5/5 (119)
- Her Body and Other Parties: StoriesFrom EverandHer Body and Other Parties: StoriesRating: 4 out of 5 stars4/5 (821)
- Espectro Infrarojo y de Raman de Complejos de Mesetileno Con CR, Mo y TuDocument12 pagesEspectro Infrarojo y de Raman de Complejos de Mesetileno Con CR, Mo y TuSofia BujosaNo ratings yet
- C DockerDocument14 pagesC Dockerharicoolguy111No ratings yet
- IMP Last Minute Revision Formulae Inorganic ChemistryDocument4 pagesIMP Last Minute Revision Formulae Inorganic ChemistryMakeshsvm2611No ratings yet
- Inter 2nd Year ChemistryDocument20 pagesInter 2nd Year ChemistryRsp Srinivas50% (4)
- DocxDocument7 pagesDocxNazrizal FitriNo ratings yet
- BSC Chemistry Syllabus 01122015Document38 pagesBSC Chemistry Syllabus 01122015shyam sundarNo ratings yet
- Advanced Aspects SpectrosDocument548 pagesAdvanced Aspects SpectrosPraveen Mishra100% (3)
- NIT Agartala Assistant Professor Recruitment Exam SyllabusDocument46 pagesNIT Agartala Assistant Professor Recruitment Exam SyllabusAnupam AlokNo ratings yet
- Metal NitrosylDocument206 pagesMetal NitrosylSandipan SahaNo ratings yet
- Lab Report skt1013Document6 pagesLab Report skt1013Nur Wanyz SyazwanieNo ratings yet
- @iitwale Join JEE 215 Most Important Question With SolutionsDocument191 pages@iitwale Join JEE 215 Most Important Question With SolutionsSjNo ratings yet
- BS Chemistry IitbombayDocument35 pagesBS Chemistry IitbombaySwati GautamNo ratings yet
- Lec 28Document21 pagesLec 28Priyank DevpuraNo ratings yet
- Complexometric TitrationsDocument16 pagesComplexometric TitrationsRakesh Chawla0% (1)
- Chemistry QP4Document6 pagesChemistry QP4Jinendra UvarajNo ratings yet
- Chemistry EX - 2 Solution of Class 12th All Sheets RESONANCEDocument180 pagesChemistry EX - 2 Solution of Class 12th All Sheets RESONANCEGOURISH AGRAWALNo ratings yet
- Dehradun Public School ASSIGNMENT (2020-21) Subject-Chemistry (043) Class-Xii Unit1:Solid State I-Multiple Choice QuestionsDocument23 pagesDehradun Public School ASSIGNMENT (2020-21) Subject-Chemistry (043) Class-Xii Unit1:Solid State I-Multiple Choice QuestionsSarvesh Kumar SinghNo ratings yet
- The Effect of Curcumin On Metal IonsDocument2 pagesThe Effect of Curcumin On Metal IonsLai ChungyiNo ratings yet
- Synthesis and Characterization of Tungsten Carbonyl Complexes Containing ThioamidesDocument7 pagesSynthesis and Characterization of Tungsten Carbonyl Complexes Containing ThioamidesRasel MahfujNo ratings yet
- GATE Chemistry 2007Document21 pagesGATE Chemistry 2007prasant906No ratings yet
- Method of Continuous Variations - Applications of Job Plots To The Study of Molecular Associations in Organometallic ChemistryDocument17 pagesMethod of Continuous Variations - Applications of Job Plots To The Study of Molecular Associations in Organometallic ChemistryYosimayrobi Endo NavarroNo ratings yet
- Unit 5Document12 pagesUnit 5Sky NewNo ratings yet
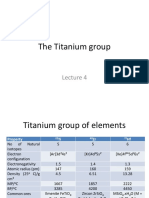
- Lecture 4 - Titanium GroupDocument31 pagesLecture 4 - Titanium Groupmalenya1No ratings yet
- D Block (2012 13)Document8 pagesD Block (2012 13)Anonymous 8VJhV1eI2y100% (1)
- Syntheses and Luminescence Behaviour of Dinuclear Copper Selenolate and Tellurolate Complexes. X-Ray Crystal Structures of (Cu (M-DPPM) (M-Seph) ) BF and (Cu (M-DPPM) (M-Teph) ) BFDocument2 pagesSyntheses and Luminescence Behaviour of Dinuclear Copper Selenolate and Tellurolate Complexes. X-Ray Crystal Structures of (Cu (M-DPPM) (M-Seph) ) BF and (Cu (M-DPPM) (M-Teph) ) BFArup KuntiNo ratings yet
- Chemistry BQP 2021Document16 pagesChemistry BQP 2021muhammadmansuri815No ratings yet
- Synthesis and Molecular Structure of Bis (2-Benzoylimino-Benzimidazolinato) Copper (II) - Dimethylformamide - A Metal-Containing Guanidine DerivativeDocument4 pagesSynthesis and Molecular Structure of Bis (2-Benzoylimino-Benzimidazolinato) Copper (II) - Dimethylformamide - A Metal-Containing Guanidine DerivativeJulissa Minaya AparicioNo ratings yet
- 9701 w15 QP 43 PDFDocument20 pages9701 w15 QP 43 PDFAl BeruniNo ratings yet
- CUNJCSSP 3 1 Question Paper CSIR UGC NET JRF December 2011 Chemical ScienceDocument42 pagesCUNJCSSP 3 1 Question Paper CSIR UGC NET JRF December 2011 Chemical Sciencehojibox903No ratings yet
- 2008 H2 Chemistry Preliminary Examinations Paper 2 Mark SchemeDocument10 pages2008 H2 Chemistry Preliminary Examinations Paper 2 Mark SchemeRonald McdonaldNo ratings yet