Professional Documents
Culture Documents
HW 06 Solutions Spring 2012
Uploaded by
Wolfgang HoofmeisterOriginal Description:
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
HW 06 Solutions Spring 2012
Uploaded by
Wolfgang HoofmeisterCopyright:
Available Formats
EGR 334 Thermodynamics: Homework 06
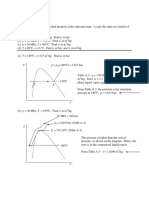
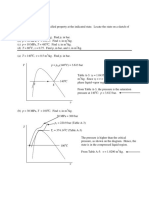
Prob 3: 5
Determine the phase of phases in a system consisting of H2O at the following conditions and sketch p-v and T-v
diagrams showing the location of each state.
a) p = 80 psi
T = 312.07 deg F.
b) p = 80 psi
T = 400 deg F.
c) p = 360 psi
T = 400 deg F.
d) p = 70 psi
T = 320 deg F.
e) 14.7 psi
T = 10 deg F.
----------------------------------------------------------------------------------------------------------------------------a) p = 80 psi
T = 312.07 deg F.
from Table A-3E: p = 80 and T = 312.07 are saturation values. Therefore this is somewhere along the liquidvapor line and exists as two phases. Quality is not given, so the spec. volume cannot be determined.
------------------------------------------------------------------------------------------------------------------------------b) p = 80 psi
T = 400 deg F.
from Table A-3E at p = 80 note that T=400 > 312.07 (saturation pressure) so the substance is vapor or
superheated.
from Table A-4E
p = 80 and T = 400 gives v = 6.22 ft 3/ lbm
----------------------------------------------------------------------------------------------------------------------------- -c) p = 360 psi
T = 400 deg F.
From table A-2E: at T = 400 note that p=360 > 247.1 (saturation pressure) so that the substance is condensed
liquid.
from table A-5E
p=500 psi and T = 400 is as close as you can get to p = 360 and 400.
therefore use v as approximately equal to 0.0186 ft3/lbm.
----------------------------------------------------------------------------------------------------------------------------- --------d) p = 70 psi
T = 320 deg F.
From table A-2E: at T=320 note that p= 70 < 89.60 (saturation pressure) so the substance is vapor or superheated.
from table A-4E
at p = 60
at p = 80
T
v
T
v
300 7.26
312.1 5.47
350 7.82
350
5.80
Using double interpolation:
at p=60:
at p = 80
v320deg 7.26 (320 300)
= 7.48
then at 70 psi:
7.82 7.26
350 300
v320deg 5.47 (320 312.1)
5.80 5.47
350 312.1
= 5.54
v 7.48 (70 60)
5.54 7.48
80 60
= 6.51 ft3/lbm.
---------------------------------------------------------------------------------------------------------------------------------------e) 14.7 psi
T = 10 deg F.
We know that water freezes at 32 deg F., so at 14.7 psi and 10 deg, the water is ice. At 10 deg the sat. spec. volume
of ice is 0.01744 ft3/lbm.
(c)
(a)
80
400
320
(b)
(c)
(b)
(d)
(a)
(d)
(e)
(e)
v
EGR 334 Thermodynamics: Homework 06
Prob 3:7
the following table lists temperature and specific volume of water vapor at two pressures.
p= 1.0 MPa
p=1.5 MPa
T (deg C)
v (m3/kg)
T (deg C)
200
0.2060
200
240
0.2275
240
280
0.2480
280
v (m3/kg)
0.1325
0.1483
0.1627
Data encounter in solving problems often do not fall exactly on the grid of values provided by property tables, and
linear interpolation between adjacent table entries becomes necessary. Using the data provided here, estimate
a) the specific volume at T=240 deg C, p = 1.25 MPa in m3/kg
b) the temperature at p = 1.5 MPa, v = 0.155 m3/kg in deg C.
c) the specific volume at T =220 deg C, p = 1.4 MPa in m3/kg
----------------------------------------------------------------------------------------------------------------------------a) the specific volume at T=240 deg C, p = 1.25 MPa in m3/kg
v va ( p pa )
vb va
pb pa
v 0.2275 (1.25 1.0)
0.1483 0.2275
0.1879 m3/kg
1.5 1.0
b) the temperature at p = 1.5 MPa, v = 0.155 m3/kg in deg C.
Tb Ta
vb va
280 240
T 240 (220 200)
258.6 oC
240 200
T Ta (v va )
c) the specific volume at T =220 deg C, p = 1.4 MPa in m3/kg:
need to do a double linear interpolation:
then
at p = 1.0
since 220 is halfway between 200 and 240
at p = 1.5
v v
v v
vaT 220 200 240
vbT 220 200 240
2
2
0.2060 0.2275
0.1325 0.1483
vaT 220
0.2168
vbT 220
0.1404
2
2
then:
vb va
pb pa
0.1404 0.2168
v 0.2168 (1.4 1.)
0.15568 m3/kg:
1.5 1.0
v va ( p pa )
EGR 334 Thermodynamics: Homework 06
Prob 3:10
For H20 determine the specified property at the indicated state. Locate the state on a sketch on the T-v diagram.
a) p=300 kPa,
v = 0.5 m3/kg,
find T in deg C.
b) p = 28 MPa,
T = 200 deg C, find v in m3/kg
c) p = 1 MPa,
T = 405 deg C,
Find v in m 3/kg
d) T = 100 deg C, x = 60 %,
find v in m 3/kg.
----------------------------------------------------------------------------------------------------------------------------a) p=300 kPa = 3 bar,
v = 0.5 m3/kg ,
find T in deg C.
Table A-3 at p = 300
and vapor.
vf = 0.0010732 < 0.5 < 0.6058 = v g (sat. vapor)so the substance is mixture of liquid
Therefore the temperature is the saturation temp for p=300 which is T = 133.6 deg C.
----------------------------------------------------------------------------------------------------------------------------- ----------b) = 28 MPa = 280 bar
T = 200 deg C, find v in m3/kg
Table A-2 at T = 200 p=280 > 15.54 (sat pressure) so the substance is compressed liquid.
using table A-5:
p = 250 bar
p = 300 bar
T= 200
T = 200
v = 0.0011344
v = 0.0011302
interpolating:
v 0.0011344 (280 250)
0.0011302 0.0011344
0.0011319 m3/kg
300 250
---------------------------------------------------------------------------------------------------------------------------------------- c) p = 1 MPa= 10 bar
T = 405 deg C,
Find v in m 3/kg
Table A-3 at 10 bar
Using table A-4: at
T= 405 > 179.9 (sat temp) so that the substance is vapor or superheated.
p = 10 bar
T
v
400
0.3066
440
0.3257
interpolating:
v 0.3066 (405 400)
0.3257 0.3066
0.3090 m3/kg
440 400
----------------------------------------------------------------------------------------------------------------------------- -------d) T = 100 deg C, x = 60 %,
find v in m3/kg.
Table A-2 at T = 100
using the quality:
vf =0.0010435
and vg = 1.673
v v f x (v g v f )
0.0010435 0.6(1.673 0.0010435) 1.004 m3/kg.
T
(c)
(a)
(b)
(d)
v
EGR 334 Thermodynamics: Homework 06
Prob 3: 29
Ammonia contained in a piston cylinder assembly initially saturated vapor at 0 deg F, undergoes an isothermal
process during which its volume (a) double and ( b) reduces by a half. For each case, fix the final state by giving
the quality or pressure in lbf/in2 as appropriate. Locate the initial and final states on sketch of the p-v and T-v
diagrams.
----------------------------------------------------------------------------------------------------------------------------For ammonia the tables A-13E, A-14E, and A-15E will be used.
a) Double the volume:
State 1: saturated vapor at 0 deg F.
Process 1-2: isothermal (T= constant) and v2 = 2 v1 ( volume doubles)
St1ate 2: substance becomes vaporNeed to use the superheated table: A-15E.
saturated vapor give p1 = 30.416 psi and v1 = vg = 9.1100 ft3/lbm
At State 1: T1 = 0 degrees
T2=0 and v2 = 2v1 = 2(9.11)= 18.2200 ft3/lbm
Using the super heated table A-15E
At state 2:
at p = 14 psi
T=0
v = 20.289
p = 16 psi
T=0
v = 17.701
interpolating:
p 14 (18.220 20.289)
16 14
15.60 psi
17.701 20.289
----------------------------------------------------------------------------------------------------------------------------- ----b) half the volume:
State 1: saturated vapor at 0 deg F.
Process 1-2: isothermal (T= constant) and v2 = 0.5 v1 ( volume is halved)
St1ate 2: substance becomes liquid-vapor mixture.
saturated vapor give p1 = 30.416 psi and v1 = vg = 9.1100 ft3/lbm
At State 1: T1 = 0 degrees
T2=0 and v2 = 0.5v1 = 0.5(9.11)= 4.555 ft3/lbm
At state 2:
using
vf = 0.02419
and vg = 9.1100
then to find the quality:
v vg
v f vg
4.555 0.02419
.499 49.9%
9.1100 0.02419
since the substance is a two phase mixture, the pressure is the saturation pressure for T = 0 deg or p = 30.416 psi
2b
1
2a
2a
2b
You might also like
- Chapter 3 Problems 7th EditionDocument25 pagesChapter 3 Problems 7th Editionallswell0% (2)
- HW3 SolutionDocument14 pagesHW3 SolutionBryceNo ratings yet
- Toaz - Info Chapter 3 Problems 7th Edition PRDocument25 pagesToaz - Info Chapter 3 Problems 7th Edition PRFiras 01No ratings yet
- NEU Thermo HW 5 SolutionsDocument7 pagesNEU Thermo HW 5 SolutionschrisNo ratings yet
- Departure FunctionDocument6 pagesDeparture FunctionzidinhoNo ratings yet
- JuasDocument49 pagesJuasAndrés RodríguezNo ratings yet
- Chap3 4Document10 pagesChap3 4rpb109100% (1)
- 8.8 Calculation of Fugacity (Liquids) : Example S8.1 Vapor and Liquid Fugacities Using The Virial EquationDocument1 page8.8 Calculation of Fugacity (Liquids) : Example S8.1 Vapor and Liquid Fugacities Using The Virial EquationtruffeloveNo ratings yet
- Assignment 2 SolutionDocument5 pagesAssignment 2 SolutionFlynn John DseeNo ratings yet
- 8 16Document3 pages8 16ejans54No ratings yet
- Lecture - 3 - Heat and The First Law Example ProblemsDocument22 pagesLecture - 3 - Heat and The First Law Example ProblemsifiokNo ratings yet
- ENGSC 2333 - ThermodynamicsDocument47 pagesENGSC 2333 - ThermodynamicsDan McNo ratings yet
- ITK-233-2 - PVT Behavior of FluidDocument57 pagesITK-233-2 - PVT Behavior of FluidVinay GuptaNo ratings yet
- Thermodynamics Midterm 1Document11 pagesThermodynamics Midterm 1Andrew LuongNo ratings yet
- Chapter 15 Heat Exchanger NetworksDocument29 pagesChapter 15 Heat Exchanger NetworksRina Hapsarininggar0% (1)
- CHE243 Winter 2007 Assignment 02Document2 pagesCHE243 Winter 2007 Assignment 02Adam SchellNo ratings yet
- State T (F) P (Mpa) H (Btu/Lb) S (Btu/Lb F) : 1 - 280 0.032 218 2 Sat'D Vap - 280 0.032 339 2.35 3' 2 2.35 4 - 160 2 218 1.54Document2 pagesState T (F) P (Mpa) H (Btu/Lb) S (Btu/Lb F) : 1 - 280 0.032 218 2 Sat'D Vap - 280 0.032 339 2.35 3' 2 2.35 4 - 160 2 218 1.54Harry VirgoNo ratings yet
- ME 231 ch3Document5 pagesME 231 ch3Hassan MrowehNo ratings yet
- Problem SetDocument2 pagesProblem SetLORD BOY SILONGNo ratings yet
- Thermo HW SolutionsDocument35 pagesThermo HW SolutionsekantikdevoteeNo ratings yet
- Solution#2Document5 pagesSolution#2Mohammad W. JeeNo ratings yet
- Thermo 5th Chap10 P001Document29 pagesThermo 5th Chap10 P001welberTonetoMotaNo ratings yet
- AerE344L Lab 02 Instruction - 2017VDocument7 pagesAerE344L Lab 02 Instruction - 2017VThi KumarNo ratings yet
- HW1 SolutionDocument8 pagesHW1 SolutionFransiscus Leo ChandraNo ratings yet
- Tut. - No.1 - ME2121 (July 2011)Document6 pagesTut. - No.1 - ME2121 (July 2011)Divij SoodNo ratings yet
- HW 12 Solutions Spring 2012Document4 pagesHW 12 Solutions Spring 2012Pravallika KolliparaNo ratings yet
- (Chemical Engineering Laboratory 2) : Experiment 7: Marcet BoilerDocument13 pages(Chemical Engineering Laboratory 2) : Experiment 7: Marcet BoilerSaranya DeviNo ratings yet
- Assigment1 SolDocument5 pagesAssigment1 SolMahmood SamadpoorNo ratings yet
- Tutorial 1Document29 pagesTutorial 1pendrive80No ratings yet
- Homework 08Document7 pagesHomework 08BasilLeeNo ratings yet
- Chemistry 231 Tutorial Session # 5: The Following Questions Will Be Answered On The Tutorial SessionDocument8 pagesChemistry 231 Tutorial Session # 5: The Following Questions Will Be Answered On The Tutorial SessionFiqkyAkbarNo ratings yet
- Solutions To Exercise One - Fluid Statics and Manometers: Mechanical Engineering 390 Fluid MechanicsDocument5 pagesSolutions To Exercise One - Fluid Statics and Manometers: Mechanical Engineering 390 Fluid MechanicsKevin DevastianNo ratings yet
- Lec. 3 DR, MarwaDocument25 pagesLec. 3 DR, MarwaFathi ShokryNo ratings yet
- Problem Set 10 Key - Physical Chemistry For Engineers. (Book Work)Document8 pagesProblem Set 10 Key - Physical Chemistry For Engineers. (Book Work)krymxenNo ratings yet
- CH 3Document48 pagesCH 3hadeelNo ratings yet
- TermodinamicaDocument3 pagesTermodinamicaDiianaBm50% (2)
- Threlkeld 5 AnswersDocument11 pagesThrelkeld 5 AnswersJaycob Clavel100% (2)
- HW5Document6 pagesHW5YTK96No ratings yet
- PR Kelas Termodinamika - Reza WDocument8 pagesPR Kelas Termodinamika - Reza WReza WardhanaNo ratings yet
- ENGSC 2333 - ThermodynamicsDocument47 pagesENGSC 2333 - ThermodynamicsPravallika KolliparaNo ratings yet
- Chem e Thermo Chapter 1 SolnDocument5 pagesChem e Thermo Chapter 1 SolnBernice Go100% (1)
- Example 2:: PMMHGDocument15 pagesExample 2:: PMMHGJHuvieCLaireNo ratings yet
- Problemas CompletosDocument16 pagesProblemas CompletosakksNo ratings yet
- DistillationDocument23 pagesDistillationGueule D'angeNo ratings yet
- Assignment # 1: Date: August 21, 2015, Before 11:59 PMDocument3 pagesAssignment # 1: Date: August 21, 2015, Before 11:59 PMQil Aznan0% (1)
- 8-39E Oxygen Gas Is Compressed From A Specified Initial State To A Final Specified State. The Reversible WorkDocument8 pages8-39E Oxygen Gas Is Compressed From A Specified Initial State To A Final Specified State. The Reversible WorkAnonymous 5OI0ClBZNo ratings yet
- Power Plant Technology, El Wakil, Problem 8.14Document33 pagesPower Plant Technology, El Wakil, Problem 8.14Reynold Curampez100% (2)
- Thermodynamics Fundamentals For ApplicationsDocument7 pagesThermodynamics Fundamentals For ApplicationsMohamed Sayed AbdoNo ratings yet
- 38Document1 page38Edbert TulipasNo ratings yet
- Problem Set G 10.213 Solution Spring 2002 23) A Schematic of The Rankine Cycle and Representation On A T-S Diagram: Q 2 TDocument10 pagesProblem Set G 10.213 Solution Spring 2002 23) A Schematic of The Rankine Cycle and Representation On A T-S Diagram: Q 2 TRalph Segundo SalvadorNo ratings yet
- MAE113 HW1 SolutionDocument12 pagesMAE113 HW1 SolutionDeepak ChandrasekarNo ratings yet
- 4Document4 pages4Carla CoronaNo ratings yet
- Chapter 3Document5 pagesChapter 3luminaNo ratings yet
- ME2121 - ME2121E Slides Chapter 3 (2014)Document23 pagesME2121 - ME2121E Slides Chapter 3 (2014)FlancNo ratings yet
- Kevin Khein P. Biyong Bsme-2A 03-08-2021Document3 pagesKevin Khein P. Biyong Bsme-2A 03-08-2021Kevin Khein P. BiyongNo ratings yet
- PNEUMATICS AND AIR CIRCUITS UNDERSTANDING THE CASCADE VALVE AND PLC UNDERSTANDINGFrom EverandPNEUMATICS AND AIR CIRCUITS UNDERSTANDING THE CASCADE VALVE AND PLC UNDERSTANDINGNo ratings yet
- Voltage Induced in A CoilDocument12 pagesVoltage Induced in A CoilWolfgang HoofmeisterNo ratings yet
- Op ManuDocument6 pagesOp ManuWolfgang HoofmeisterNo ratings yet
- EM306 Thermodynamics II Homework 3: Student Name: Student IDDocument1 pageEM306 Thermodynamics II Homework 3: Student Name: Student IDWolfgang HoofmeisterNo ratings yet
- ME 321 - Winter 2002 Tutorial #4 Page 1 of 3Document3 pagesME 321 - Winter 2002 Tutorial #4 Page 1 of 3Wolfgang HoofmeisterNo ratings yet
- Solved Problems On Ch3Document4 pagesSolved Problems On Ch3Luis_Mauricio__1151100% (2)
- Kinematics & Dynamics of MachineryDocument43 pagesKinematics & Dynamics of MachineryWolfgang HoofmeisterNo ratings yet
- A-3 27Document3 pagesA-3 27Wolfgang HoofmeisterNo ratings yet
- Laplace AssignmentDocument1 pageLaplace AssignmentWolfgang HoofmeisterNo ratings yet
- Unrar SRCDocument1 pageUnrar SRCMax SuvilianNo ratings yet
- Calculation of Non-LinearityDocument2 pagesCalculation of Non-LinearityAlbertus Setya BudhiNo ratings yet
- Operation MNGDocument5 pagesOperation MNGWolfgang HoofmeisterNo ratings yet
- Operation ManagementslideDocument31 pagesOperation ManagementslideWolfgang HoofmeisterNo ratings yet
- Calculation of Non-LinearityDocument2 pagesCalculation of Non-LinearityAlbertus Setya BudhiNo ratings yet
- AutoCAD Drawing ManualDocument31 pagesAutoCAD Drawing ManualWolfgang HoofmeisterNo ratings yet
- Tech NoteDocument5 pagesTech NoteArslan AjmalNo ratings yet
- Hand Drawing ManualDocument13 pagesHand Drawing ManualWolfgang HoofmeisterNo ratings yet
- Verified Email Ids From IndoreDocument3 pagesVerified Email Ids From IndoreAnuraag GuptaaNo ratings yet
- Computing For Engineers: Lab Activity 6: Arrays ObjectivesDocument4 pagesComputing For Engineers: Lab Activity 6: Arrays ObjectivesWolfgang HoofmeisterNo ratings yet
- Hurricane Isaac Louisiana Mississippi River US Geological Survey Plaquemines Parish New Orleans Baton RougeDocument1 pageHurricane Isaac Louisiana Mississippi River US Geological Survey Plaquemines Parish New Orleans Baton RougeWolfgang HoofmeisterNo ratings yet
- HW 06 Solutions Spring 2012Document4 pagesHW 06 Solutions Spring 2012Wolfgang HoofmeisterNo ratings yet
- Assignment 3Document1 pageAssignment 3Wolfgang HoofmeisterNo ratings yet
- Fluid Flow Shell Momentum BalanceDocument16 pagesFluid Flow Shell Momentum BalanceMegan MundellNo ratings yet
- Unit 5 Testing of DC MachinesDocument23 pagesUnit 5 Testing of DC Machinesmastanamma.Y100% (1)
- Science 7Document4 pagesScience 7junar asentistaNo ratings yet
- Phys. 3341 Homework Solution Set #5: 1 Reif 3.3Document4 pagesPhys. 3341 Homework Solution Set #5: 1 Reif 3.3Daiver JuarezNo ratings yet
- Inductor Loss Calcs VishayDocument12 pagesInductor Loss Calcs VishayealbinNo ratings yet
- Prof. Dr. Niranjan D. Chatterjee (Auth.) - Applied Mineralogical Thermodynamics - Selected Topics-Springer-Verlag Berlin Heidelberg (1991) PDFDocument331 pagesProf. Dr. Niranjan D. Chatterjee (Auth.) - Applied Mineralogical Thermodynamics - Selected Topics-Springer-Verlag Berlin Heidelberg (1991) PDFJoão Grilo0% (1)
- Impulse MomentumDocument11 pagesImpulse Momentumdaa ampsNo ratings yet
- SF6 Abatement in A Packed Bed Plasma Reactor: Role of Zirconia Size and Optimization Using RSMDocument12 pagesSF6 Abatement in A Packed Bed Plasma Reactor: Role of Zirconia Size and Optimization Using RSMClaudia TrujilloNo ratings yet
- CT-LTI System Analysis: Approach #IDocument18 pagesCT-LTI System Analysis: Approach #IBAYAT MOBIN BAMIMNo ratings yet
- 10.1 - Resistance QP PDFDocument16 pages10.1 - Resistance QP PDFMuhammad Saif AmirNo ratings yet
- Preview of 13.10-13.15: Kinetics of Particles: Energy and Momentum MethodsDocument21 pagesPreview of 13.10-13.15: Kinetics of Particles: Energy and Momentum MethodsAnonymous ANo ratings yet
- Important NoticeDocument57 pagesImportant NoticeSahil KaundunNo ratings yet
- Structure of Atoms: NEET/JEE: 2020-21Document2 pagesStructure of Atoms: NEET/JEE: 2020-21shantinath123gmailcoNo ratings yet
- EM 67 - Recommended Practices For A Bump TestDocument4 pagesEM 67 - Recommended Practices For A Bump TestChad Hunt100% (2)
- Problem Sheet - 2 Topic: Stress Submission Date: 27/08/2019Document3 pagesProblem Sheet - 2 Topic: Stress Submission Date: 27/08/2019Beesam Ramesh KumarNo ratings yet
- Convolution WebDocument7 pagesConvolution WebtesNo ratings yet
- J Jenvrad 2016 04 008 PDFDocument5 pagesJ Jenvrad 2016 04 008 PDFNguyễnKhươngDuyNo ratings yet
- The Language of Quantum MechanicsDocument7 pagesThe Language of Quantum Mechanicslaura HernandezNo ratings yet
- Ansys CFX Intro 12.0 1st-Edition - WorkshopsDocument312 pagesAnsys CFX Intro 12.0 1st-Edition - Workshopskdennis_nguyenNo ratings yet
- Jee Mains Qs1Document11 pagesJee Mains Qs1Ajay KumarNo ratings yet
- Methods and Practice in Cantilever Retaining Wall DesignDocument14 pagesMethods and Practice in Cantilever Retaining Wall DesignArdalan100% (2)
- CHAPTER 5 - STATES OF MATTER (S) Edit20152016Document12 pagesCHAPTER 5 - STATES OF MATTER (S) Edit20152016PAKK20622P Syarifah Nor Izzah binti Syed Abd HamidNo ratings yet
- 300+TOP CURRENT ELECTRICITY Multiple Choice Questions & AnswersDocument59 pages300+TOP CURRENT ELECTRICITY Multiple Choice Questions & Answersczds6594No ratings yet
- Chemical Bonding Notes For ExamDocument4 pagesChemical Bonding Notes For ExamMermz96No ratings yet
- Poster PresentationDocument1 pagePoster PresentationKarli KingNo ratings yet
- Lateral Earth PressureDocument55 pagesLateral Earth PressureRakesh SheteNo ratings yet
- A Study of The Eigenvalue Analysis Capabilities ofDocument9 pagesA Study of The Eigenvalue Analysis Capabilities ofNedim HadžiaganovićNo ratings yet
- New Mathod To Evaluate The Uplift Cap of Belled Pile in Sandy Soil PDFDocument11 pagesNew Mathod To Evaluate The Uplift Cap of Belled Pile in Sandy Soil PDFM TaufikNo ratings yet
- A Novel Micro Gripper With Parallel Movement of Gripping ArmsDocument4 pagesA Novel Micro Gripper With Parallel Movement of Gripping ArmssohailNo ratings yet