Professional Documents
Culture Documents
Biochemistry: Water and Organic Compounds: Modern Biology
Uploaded by
Ana AyalaOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Biochemistry: Water and Organic Compounds: Modern Biology
Uploaded by
Ana AyalaCopyright:
Available Formats
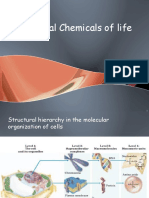
Biochemistry: Water and Organic Compounds
Modern Biology Chapter 3
Water
Formed by covalent bonds (atoms share electrons)
Atoms dont share equallywater is POLAR
Oxygen end is slightly negative (O is electron hog) Hydrogen ends are slightly positive POLARITY is why water is a good solvent
Dissolves lots of different compounds
Hydrogen Bonding in Water
Polarity causes water to be attracted to each other H-bond: attraction that holds water molecules together Responsible for cohesion (sticking together) that produces surface tension
Cohesion and adhesion (attraction between unlike substances) responsible for capillarity (water moves up through stems)
Water has to gain/lose lots of energy to change temp.
This energy initially goes into breaking H-bonds This is why a pot of water on the stove isnt hot right away
Carbon Compounds
Organic compounds
Contain carbon atoms covalently bonded to each other and to other elements (H, O, N, usually) Needs 8 to be stableshares its electrons with other atoms
Carbon has 4 electrons in its outer E-level
Able to form straight chains, branched chains, or rings
Examples of Carbon bonds
Propane
Ethylene (plant hormoneripens fruit)
2-methylpropane (used in refrigeration and petrochemical industry)
Glucose
Functional Groups
Cluster of atoms that influence the properties of the molecule they are attached to.
Ex: -OH (hydroxl group)
Alcohols contain hydroxyl groups Makes alcohols polar
Allows them to dissolve in water, have H-bonds like water
Carbon Molecules
Monomers
Condensation Rxn
Small and simple; single building block Made of repeated, linked monomers
Polymers
Links monomers together AKA dehydration synthesis Forms water
Macromolecule
Hydrolysis Rxn
Made of linked polymers
Uses water to break polymers apart
Condensation/Hydrolysis
ATP
Adenosine Triphosphate
Energy molecule used by the body Broken down into ADP (releases P) ADP + P ATP Continuous Cycle
Carbohydrates
Made of C, H, O Ratio of H to O is 2:1 Sugars! Used for quick energy
Monosaccharides
Building blocks of all carbs (CH2O)n is generic formula Simple sugars Most common are glucose, fructose, galactose
Isomers: same formula, different structure
Carbohydrates
Disaccharides
Formed from 2 monosaccharides
Condensation reaction Ex: sucrose
Polysaccharides
Formed from 3+ monosaccharides
Condensation reaction
Glycogen: storage in animals (used for energy) \ Starch: storage in plants Cellulose: in plant cell walls (gives them rigidity, crunch)
Lipids
Fats! Large, non-polar; do not dissolve in water Have more C, H than O Responsible for storing energy
Lipids
Fatty Acids
Building blocks of lipids Straight carbon chain with carboxyl group (COOH) on one end (makes that end polar) Carboxyl end head = hydrophilic
Water-Loving Water-Fearing Saturated: all C-C bonds are single bonds (is full of Hs) Unsaturated: contains some double bonds
Carbon chain tail = hydrophobic
Can be Saturated or Unsaturated
Lipids
Lipids
Triglycerides
Waxes
3 Fatty Acids attached to 1 Glycerol molecule
Saturated: solid at room temp (shortening) Unsaturated: liquid at room temp (oils, found in plant seeds/fruits)
Waterproof Form protective coating on plant leaves
Steroids
Phospholipids
4 fused carbon rings w/ functional groups Hormones
2 FAs attached to 1 Glycerol
Major component of cell membrane (lipid bilayer)
Cholesterol Testosterone Etc.
Proteins
Made of C, H, O, and N Building blocks are Amino Acids
Amino Acids (20) are almost identical
Difference is the R-group (functional group)
Di-peptide: chain of 2 Amino Acids Polypeptide: lots of AAs linked together Bond that holds AAs together is peptide bond
Proteins
Amino Acid Structure
Enzymes
Proteins Act as catalyst for many reactions that occur in the body Substrate: what the enzyme acts on Product(s): what is formed from the rxn
Enzymes
Are not used up during the reaction Are not changed during the reaction
Can do the job again and again Specific shape, must fit perfectly for rxn to occur
Lock and Key fit
Enzymes can be denatured (destroyed) when:
Change in temperature (too high) Change in pH
Nucleic Acids
Used by cells to store hereditary information
DNA
Deoxyribonucleic acid Ribonucleic acid
RNA
Made of building blocks called nucleotides
5-carbon sugar Phosphate group Nitrogen base
You might also like
- Molecules of LifeDocument38 pagesMolecules of LifeAmanda ArceoNo ratings yet
- BIO 22 MODULE 1 - Chemical Basis of LifeDocument14 pagesBIO 22 MODULE 1 - Chemical Basis of LifeBryan DGNo ratings yet
- Chapter 02 Winter 2020Document53 pagesChapter 02 Winter 2020LESLI RODRIGUEZ BENDEZUNo ratings yet
- Walang MakakaseethisDocument3 pagesWalang MakakaseethisAlissa YangNo ratings yet
- Biology Notes Unit 1Document40 pagesBiology Notes Unit 1MirzaAteeqAhmedBaigNo ratings yet
- AP Bio Unit 1 Study GuideDocument2 pagesAP Bio Unit 1 Study GuideEllie GriffinNo ratings yet
- AQA Module All Revision NotesDocument82 pagesAQA Module All Revision NotesRS JNo ratings yet
- Carbohydrates SlidesDocument75 pagesCarbohydrates SlidesShiva PratheekNo ratings yet
- As 2 Biological MoleculesDocument29 pagesAs 2 Biological MoleculesisaacNo ratings yet
- Biological Molecules: Reducing Sugars TestsDocument5 pagesBiological Molecules: Reducing Sugars TestsShinuk KangNo ratings yet
- Smackslidecom Powerpoint Temp 6032625eb534aDocument54 pagesSmackslidecom Powerpoint Temp 6032625eb534aQuang MinhNo ratings yet
- 3 Essential Chemicals of Life PPTDocument82 pages3 Essential Chemicals of Life PPTBhoni KumariNo ratings yet
- Biochemistry of CellsDocument79 pagesBiochemistry of CellsketakeeNo ratings yet
- AQA As-Level Biology Revision Notes - New SpecDocument38 pagesAQA As-Level Biology Revision Notes - New SpecjimboNo ratings yet
- Biology Revision NotesDocument42 pagesBiology Revision Noteshumza bhattiNo ratings yet
- Clase 2 Chemistry of CellDocument73 pagesClase 2 Chemistry of Cellediaz_956003No ratings yet
- Organic InorganicDocument51 pagesOrganic InorganicPatrick GoNo ratings yet
- H2 Biology - Notes On BiomoleculesDocument9 pagesH2 Biology - Notes On BiomoleculesSefLRho100% (1)
- Chemical Reactions Part II: Week 7: Organic ChemistryDocument24 pagesChemical Reactions Part II: Week 7: Organic Chemistrymia kris quiatchonNo ratings yet
- Bio Final Review Review Quiz and TestDocument15 pagesBio Final Review Review Quiz and TestLinda ChaoNo ratings yet
- AP Review: Chapters 2-5Document19 pagesAP Review: Chapters 2-5Min Cheol ShinNo ratings yet
- Molecular BiologyDocument21 pagesMolecular BiologyvaishnaviNo ratings yet
- A-level Biology Revision: Cheeky Revision ShortcutsFrom EverandA-level Biology Revision: Cheeky Revision ShortcutsRating: 5 out of 5 stars5/5 (5)
- BiochemistryDocument5 pagesBiochemistrynaruto710@wuNo ratings yet
- Lecture 2 - Introductory BiochemistryDocument15 pagesLecture 2 - Introductory BiochemistryJana-Tae KerrNo ratings yet
- Chemical Composition of The BodyDocument39 pagesChemical Composition of The BodyAyeshaNo ratings yet
- Biochemistry: Biological MoleculesDocument18 pagesBiochemistry: Biological MoleculesAnup Singh TanwarNo ratings yet
- Unit 1 Chemistry of LifeDocument9 pagesUnit 1 Chemistry of Liferaghad mohammedNo ratings yet
- Chapter 3 The Molecules of CellsDocument5 pagesChapter 3 The Molecules of Cellsmzunl25476No ratings yet
- Topic I: Introduction To AnatomyDocument3 pagesTopic I: Introduction To AnatomyCristenNo ratings yet
- General Biology All About BiomoleculesDocument6 pagesGeneral Biology All About BiomoleculeskimberlyfritzzunigaNo ratings yet
- Biochemistry: Water Carbohydrates Lipids ProteinsDocument16 pagesBiochemistry: Water Carbohydrates Lipids ProteinsryanNo ratings yet
- Worktext 1 - Biomol - Carbo & Lipids, PERALTADocument10 pagesWorktext 1 - Biomol - Carbo & Lipids, PERALTAKelsi Kyla PeraltaNo ratings yet
- A Level BiologyDocument522 pagesA Level BiologyJAMESNo ratings yet
- Biology 1308 NotesDocument21 pagesBiology 1308 NotesAshley CisnerosNo ratings yet
- Molecules of LifeDocument5 pagesMolecules of LifeEmmaNo ratings yet
- Biology 12 Biological Molecules Review KEYDocument5 pagesBiology 12 Biological Molecules Review KEYFeras TarawnehNo ratings yet
- The Chemical Basis of Life Ii: Organic MoleculesDocument47 pagesThe Chemical Basis of Life Ii: Organic Moleculesmeer0091No ratings yet
- Lecture 4, 5: Chemical Reactions Acids, Bases & SaltsDocument13 pagesLecture 4, 5: Chemical Reactions Acids, Bases & Saltstenzin bhutiNo ratings yet
- 26 Inorganic and Organic CompoundsDocument6 pages26 Inorganic and Organic CompoundsMark Allein AguinaldoNo ratings yet
- Biomolecules (KH, Lemak, Protein) - Topik 3 FBS 1Document47 pagesBiomolecules (KH, Lemak, Protein) - Topik 3 FBS 1Teofilus Dani PNo ratings yet
- Lectures by Tariq Alalwan, PH.D.: Biology, 12/e Mader & WindelspechtDocument74 pagesLectures by Tariq Alalwan, PH.D.: Biology, 12/e Mader & WindelspechtSulaiman Dawood Saif AbdullaNo ratings yet
- General Biology - Chap. 03+Document51 pagesGeneral Biology - Chap. 03+정서진No ratings yet
- Bio Molecules PPT For P AP BiologyDocument34 pagesBio Molecules PPT For P AP BiologyDivineDoctorNo ratings yet
- Chemical Basis of LifeDocument38 pagesChemical Basis of LifeFernadez RodisonNo ratings yet
- Biological Molecules: When X y It's Simple So CX (H20) y When X Does Not y It's Simple So CX (H20) yDocument15 pagesBiological Molecules: When X y It's Simple So CX (H20) y When X Does Not y It's Simple So CX (H20) yteeeNo ratings yet
- Chapter 4 - Biological MoleculesDocument28 pagesChapter 4 - Biological MoleculesshammmssNo ratings yet
- Atoms and MoleculesDocument5 pagesAtoms and MoleculesRelaisa CimafrancaNo ratings yet
- Lecture 2 - Introductory BiochemistryDocument17 pagesLecture 2 - Introductory BiochemistryKevonSingh1No ratings yet
- Biological MoleculesDocument11 pagesBiological MoleculesYasasmi GunasekeraNo ratings yet
- Week 3 Carbohydrates 2Document26 pagesWeek 3 Carbohydrates 2sabeera.ahmadNo ratings yet
- Anatomy and Physiology ChemistryDocument49 pagesAnatomy and Physiology ChemistryNoahSullivanNo ratings yet
- Biology Notes (Lipids)Document5 pagesBiology Notes (Lipids)Teo Jia Ming Nickolas100% (1)
- All Biology LessonsDocument217 pagesAll Biology Lessonsadliabuwade3No ratings yet
- Biochemistry: Additional Support Materials I.E. Animations, Quizzes, Pictures, WorksheetsDocument26 pagesBiochemistry: Additional Support Materials I.E. Animations, Quizzes, Pictures, WorksheetsnikkikeswaniNo ratings yet
- Biology As + A2 CombinedDocument253 pagesBiology As + A2 CombinedgalaxyreaderNo ratings yet
- Notes+4 +ATP,+Water+and+Inorganic+IonsDocument5 pagesNotes+4 +ATP,+Water+and+Inorganic+IonsSyeda Wardah NoorNo ratings yet
- Anatomy and Physiology For Students: A College Level Study Guide for Life Science and Allied Health MajorsFrom EverandAnatomy and Physiology For Students: A College Level Study Guide for Life Science and Allied Health MajorsNo ratings yet
- Organic Chemistry Past Papers by SKNDocument5 pagesOrganic Chemistry Past Papers by SKNMuhammad IshaqueNo ratings yet
- NAPLEX Random Notes 68 PagesDocument51 pagesNAPLEX Random Notes 68 PagesZehra Amir100% (4)
- Summary Ubat 112Document31 pagesSummary Ubat 112weehanNo ratings yet
- 20 Amino AcidsDocument1 page20 Amino AcidsJoseph PaguioNo ratings yet
- Nucleotide MetabolismDocument89 pagesNucleotide Metabolismfeby ary annasNo ratings yet
- CHAPTER 2-3 LipidDocument20 pagesCHAPTER 2-3 LipidDaniel IsmailNo ratings yet
- Aryl Halides: By:-Dr - Harpreet Kaur Pggcg11 CHDDocument21 pagesAryl Halides: By:-Dr - Harpreet Kaur Pggcg11 CHDyeateshwarriorNo ratings yet
- Answer KeyDocument64 pagesAnswer Keyamit jainNo ratings yet
- AlkaloidsDocument21 pagesAlkaloidssanjiv_m20100% (1)
- Glycolysis and Difference Between Aerobic and AnaeDocument12 pagesGlycolysis and Difference Between Aerobic and Anaepc usageNo ratings yet
- CRODA Surfactants and Alkoxylated Polyols Overview FEB 2013Document8 pagesCRODA Surfactants and Alkoxylated Polyols Overview FEB 2013Maria Eugenia CiveiraNo ratings yet
- Chapter 25 Vocabulary - Organic ChemistryDocument2 pagesChapter 25 Vocabulary - Organic ChemistryEvilasio CostaNo ratings yet
- W Ts 8 U1 P2Document308 pagesW Ts 8 U1 P2Cem ÜstündağNo ratings yet
- BMS1 - K10 - The Role of B Vitamins As An Adjuvant AnalgesicDocument45 pagesBMS1 - K10 - The Role of B Vitamins As An Adjuvant AnalgesicAndreNo ratings yet
- Consumos Medicamentos 2020Document568 pagesConsumos Medicamentos 2020LUCERO LOPEZNo ratings yet
- Waktu Tunggu Hasil Lab MutuDocument42 pagesWaktu Tunggu Hasil Lab MutuDwinoorNo ratings yet
- Karlshamns Handbook Vegetable Oils and Fats 2007Document255 pagesKarlshamns Handbook Vegetable Oils and Fats 2007Nicolas Benavidez86% (7)
- Antihyperlipidemic AgentsDocument71 pagesAntihyperlipidemic AgentsFirda100% (1)
- Cell Membrane WSDocument2 pagesCell Membrane WSDana FransenNo ratings yet
- Chemistry of Opioid Analgesics PHA 4220 - Neurology PharmacotherapeuticsDocument13 pagesChemistry of Opioid Analgesics PHA 4220 - Neurology PharmacotherapeuticsethanalbertNo ratings yet
- CYP450 Enzymes in Drug DiscoveryDocument35 pagesCYP450 Enzymes in Drug DiscoveryDiego E MarinaNo ratings yet
- Carbohydrates NotesDocument28 pagesCarbohydrates Noteschaitanyaa baliNo ratings yet
- Lipids 2018 PDFDocument102 pagesLipids 2018 PDFHyvieNo ratings yet
- Synthesis of Pyrimidine DerivativesDocument32 pagesSynthesis of Pyrimidine DerivativesJ-Paul DétoNo ratings yet
- Rice Bran Oil - Natures Gift To MankindDocument2 pagesRice Bran Oil - Natures Gift To Mankindlehmanwolf100% (1)
- CH 243. AromaticityDocument63 pagesCH 243. Aromaticityjob omyNo ratings yet
- Systematic NomenclatureDocument12 pagesSystematic NomenclatureSanjib Mal100% (1)
- 3 Parts of A NucleotideDocument2 pages3 Parts of A NucleotideGermaeGonzalesNo ratings yet
- BylawsDocument136 pagesBylawsdannyfiguracion45No ratings yet
- Hey 4-N/i'ethylenedio Yphenyl Sopropyl E.: 4-MethylenedioxyphenylisopropylamineDocument2 pagesHey 4-N/i'ethylenedio Yphenyl Sopropyl E.: 4-MethylenedioxyphenylisopropylamineAnonymous FigYuONxuuNo ratings yet